 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Sufrexal |
| Other names | R41468; R-41468; R-41,468 |
| AHFS/ Drugs.com | International Drug Names |
|
Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 50% [1] [2] |
| Protein binding | 95.0% (mainly albumin [2] |
| Elimination half-life | 10–29 hours [3] [1] [2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.070.598 |
| Chemical and physical data | |
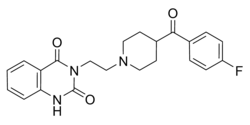
| Formula | C22H22FN3O3 |
| Molar mass | 395.434 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Ketanserin ( INN, USAN, BAN) (brand name Sufrexal; former developmental code name R41468) is a drug used clinically as an antihypertensive agent and in scientific research to study the serotonergic system; specifically, the 5-HT2 receptor family. [4] It was discovered at Janssen Pharmaceutica in 1980. [5] [6] It is not available in the United States. [1]
Uses
Medical uses
Ketanserin is classified as an antihypertensive by the World Health Organization [7] and the National Institute of Health. [8]
It has been used to reverse pulmonary hypertension caused by protamine (which in turn was administered to reverse the effects of heparin overdose). [9]
The reduction in hypertension is not associated with reflex tachycardia. [10]
It has been used in cardiac surgery. [11]
A 2000 Cochrane Review found that, compared to placebo, ketanserin did not provide significant relief for people suffering from Raynaud's phenomenon attacks in the setting of progressive systemic sclerosis (an autoimmune disorder). While the frequency of the attacks was unaffected by ketanserin, there was a reduction in the duration of the individual attacks. However, due to the significant adverse effect burden, the authors concluded that ketanserin's utility for this indication is likely unbeneficial. [12]
Ketanserin is a selective 5-HT2A receptor antagonist that was initially developed as an anti-hypertensive medicine. However, now the drug is available as a topical gel formulation for treating wounds, burns, ulcers, and anal fissures. Its action is through the acceleration of epithelialization.
Research uses
With tritium (3H) radioactively labeled ketanserin is used as a radioligand for serotonin 5-HT2 receptors, e.g. in receptor binding assays and autoradiography. [13] This radio-labeling has enabled the study of serotonin 5-HT2A receptor distribution in the human brain. [14]
An autoradiography study of the human cerebellum has found an increasing binding of 3H-ketanserin with age (from below 50 femtomol per milligram tissue at around 30 years of age to over 100 above 75 years). [15] The same research team found no significant correlation with age in their homogenate binding study.
Ketanserin has also been used with carbon (11C) radioactively labeled NNC112 in order to image cortical D1 receptors without contamination by 5-HT2 receptors. [16]
Increasing research into the use of psychedelics as antidepressants has seen ketanserin used to both block the hallucinogenic experience, and to disentangle the specific cognitive effects of 5-HT2A activation. [17]
Pharmacology
| Target | Affinity (Ki) | Ref(s) |
|---|---|---|
| α1A-adrenergic | 6.3 nM | [19] |
| α1B-adrenergic | 6.3 nM | [19] |
| α1D-adrenergic | 16 nM | [19] |
| α2A-adrenergic | 372 nM (HT29) | [18] |
| α2B-adrenergic | 199 nM | [18] |
| α2C-adrenergic | 159 nM (opossum) | [18] |
| 5-HT1A | 1,044–>10,000 nM | [19] [18] |
| 5-HT1B | 2,515–6,300 nM | [19] [18] |
| 5-HT1D | 32–>10,000 nM | [19] [20] [21] |
| 5-HT1E | >10,000 nM | [18] |
| 5-HT1F | 1.25–>10,000 nM | [18] |
| 5-HT2A | 0.20–9.8 nM | [19] [18] |
| 5-HT2B | 200–3,236 nM | [19] [18] |
| 5-HT2C | 17–186 nM | [19] [18] |
| 5-HT3 | >10,000 nM (rodent) | [18] |
| 5-HT4L | 1,000 nM (rat) | [18] |
| 5-HT5A | 20,000 nM | [19] [18] |
| 5-HT5B | 1,000–1,585 nM (rodent) | [18] |
| 5-HT6 | 2,800 nM | [18] |
| 5-HT7 | 320–1,334 nM | [19] [18] |
| D1 | 190–464 nM | [18] |
| D2 | >10,000 nM | [18] |
| D3 | ? | |
| D4 | 148 nM (canine) | [18] |
| D5 | 2,500 nM | [19] [18] |
| H1 | 1.79 nM | [18] |
| DAT | >10,000 nM | [18] |
| VMAT1 | 1,600 nM | [19] |
| VMAT2 | 500 nM | [19] |
Pharmacodynamics
Ketanserin is a high-affinity non-selective antagonist of 5-HT2 receptors in rodents, [18] [22] [20] In addition to the 5-HT2 receptors, ketanserin is also a high affinity antagonist for the H1 receptor. [23] It has also been found to block the vesicular monoamine transporter 2 (VMAT2). [24] [25]
Pharmacokinetics
The bioavailability of ketanserin is 50%. [1] [2] The plasma protein binding of ketanserin is 95.0% and it is mainly bound to albumin. [2] The elimination half-life of ketanserin is 10 to 29 hours. [3] [1]
Synthesis

Either 3-(2-Chloroethyl)quinazoline-2,4(1H,3H)-dione [5081-87-8] (1a), or alternatively 2,3-dihydro-[1,3]oxazolo[2,3-b]quinazolin-5-one [52727-44-3] (1b) can be used as starting material. Attachment of the sidechain to 4-(4-Fluorobenzoyl)piperidine [56346-57-7] (2) completes synthesis synthesis of Ketanserin (3).
See also
References
- ^ a b c d e Wolverton SE (8 March 2007). Comprehensive Dermatologic Drug Therapy (2 ed.). Elsevier Health Sciences. ISBN 978-1-4377-2070-9.
- ^ a b c d e Hideya Saitō, Masaru Minami, eds. (1992). Antihypertensive Drugs Today. VSP. pp. 191–. ISBN 978-90-6764-140-1. OCLC 231351327.
- ^ a b Cold GE, Dahl BL (11 November 2013). Topics in Neuroanaesthesia and Neurointensive Care: Experimental and Clinical Studies upon Cerebral Circulation, Metabolism and Intracranial Pressure. Springer Science & Business Media. pp. 193–. ISBN 978-3-662-04845-0. OCLC 1076237896.
- ^ O'Donnell J, Ahuja GD (2005). Drug Injury: Liability, Analysis, and Prevention. Lawyers & Judges Publishing Company. pp. 304–. ISBN 978-0-913875-27-8.
- ^ Healy D (1 July 2009). The Creation of Psychopharmacology. Harvard University Press. pp. 252–253. ISBN 978-0-674-03845-5.
- ^ Schwartz H (August 1989). Breakthrough: the discovery of modern medicines at Janssen. Skyline Pub. Group. p. 74. ISBN 978-1-56019-100-1.
- ^ ATC/DDD Index
- ^ Ketanserin
- ^ van der Starre PJ, Solinas C (1996). "Ketanserin in the treatment of protamine-induced pulmonary hypertension". Texas Heart Institute Journal. 23 (4): 301–304. PMC 325377. PMID 8969033.
- ^ Hodsman NB, Colvin JR, Kenny GN (May 1989). "Effect of ketanserin on sodium nitroprusside requirements, arterial pressure control and heart rate following coronary artery bypass surgery". British Journal of Anaesthesia. 62 (5): 527–531. doi: 10.1093/bja/62.5.527. PMID 2786422.
- ^ Elbers PW, Ozdemir A, van Iterson M, van Dongen EP, Ince C (February 2009). "Microcirculatory Imaging in Cardiac Anesthesia: Ketanserin Reduces Blood Pressure But Not Perfused Capillary Density". Journal of Cardiothoracic and Vascular Anesthesia. 23 (1): 95–101. doi: 10.1053/j.jvca.2008.09.013. PMID 19058975.
- ^ Pope J, Fenlon D, Thompson A, Shea B, Furst D, Wells G, Silman A (2000). "Ketanserin for Raynaud's phenomenon in progressive systemic sclerosis". The Cochrane Database of Systematic Reviews. 1998 (2): CD000954. doi: 10.1002/14651858.CD000954. PMC 7032891. PMID 10796396.
- ^ Eickhoff SB, Schleicher A, Scheperjans F, Palomero-Gallagher N, Zilles K (February 2007). "Analysis of neurotransmitter receptor distribution patterns in the cerebral cortex". NeuroImage. 34 (4): 1317–1330. doi: 10.1016/j.neuroimage.2006.11.016. PMID 17182260. S2CID 23363050.
- ^ Pazos A, Probst A, Palacios JM (April 1987). "Serotonin receptors in the human brain--IV. Autoradiographic mapping of serotonin-2 receptors". Neuroscience. 21 (1): 123–139. doi: 10.1016/0306-4522(87)90327-7. PMID 3601071. S2CID 23711420.
- ^ Eastwood SL, Burnet PW, Gittins R, Baker K, Harrison PJ (November 2001). "Expression of serotonin 5-HT(2A) receptors in the human cerebellum and alterations in schizophrenia". Synapse. 42 (2): 104–114. doi: 10.1002/syn.1106. PMID 11574947. S2CID 40304220.
- ^ Catafau AM, Searle GE, Bullich S, Gunn RN, Rabiner EA, Herance R, et al. (May 2010). "Imaging cortical dopamine D1 receptors using [11C]NNC112 and ketanserin blockade of the 5-HT 2A receptors". Journal of Cerebral Blood Flow and Metabolism. 30 (5): 985–993. doi: 10.1038/jcbfm.2009.269. PMC 2949183. PMID 20029452.
- ^ Quednow BB, Kometer M, Geyer MA, Vollenweider FX (February 2012). "Psilocybin-induced deficits in automatic and controlled inhibition are attenuated by ketanserin in healthy human volunteers". Neuropsychopharmacology. 37 (3): 630–640. doi: 10.1038/npp.2011.228. PMC 3260978. PMID 21956447.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x NIMH Psychoactive Drug Screening Program
- ^ a b c d e f g h i j k l m n o "Ketanserin Ligand page". IUPHAR/BPS Guide to PHARMACOLOGY.
- ^ a b Meneses A (11 March 2014). The Role of 5-HT Systems on Memory and Dysfunctional Memory: Emergent Targets for Memory Formation and Memory Alterations. Elsevier Science. pp. 23–. ISBN 978-0-12-801083-9.
- ^ Olivier B, van Wijngaarden I, Soudijn W (10 July 1997). Serotonin Receptors and their Ligands. Elsevier. pp. 118–. ISBN 978-0-08-054111-2.
- ^ Creed-Carson M, Oraha A, Nobrega JN (June 2011). "Effects of 5-HT(2A) and 5-HT(2C) receptor antagonists on acute and chronic dyskinetic effects induced by haloperidol in rats". Behavioural Brain Research. 219 (2): 273–279. doi: 10.1016/j.bbr.2011.01.025. PMID 21262266. S2CID 205882443.
- ^ Coyne CP (9 January 2008). Comparative Diagnostic Pharmacology: Clinical and Research Applications in Living-System Models. John Wiley & Sons. pp. 104–. ISBN 978-0-470-34429-3.
- ^ Muller CP, Jacobs B (30 December 2009). Handbook of the Behavioral Neurobiology of Serotonin. Academic Press. pp. 592–. ISBN 978-0-08-087817-1.
- ^ Henry JP, Sagné C, Botton D, Isambert MF, Gasnier B (1998). Molecular Pharmacology of the Vesicular Monoamine Transporter. Advances in Pharmacology. Vol. 42. San Diego, Calif.: Academic Press. pp. 236–9 (237). doi: 10.1016/s1054-3589(08)60736-x. ISBN 978-0-08-058134-7. PMID 9327887.
- ^ US 4335127, Vandenberk J, Kennis L, Van der Aa M, Van Heertum A, issued 1982, assigned to Janssen Pharmaceutica, N.V.
- ^ EP 0098499, Signorini R, Verga A, issued 1984, assigned to Ravizza SpA
- ^ CN 106866625, Shiwen R, et al., issued 2017, assigned to Shanghai Ding Ya Pharmaceutical Chemistry Science And Technology Ltd)
- ^ Fakhraian H, Heydary M (January 2014). "Reinvestigation of the Synthesis of Ketanserin (5) and its Hydrochloride Salt (5. HCl) via 3-(2-Chloroethyl)-2, 4-(1H, 3H)-quinazolinedione (2) or Dihydro-5H-oxazole (2, 3-b) quinazolin-5-one (1)". Journal of Heterocyclic Chemistry. 51 (1): 151–156. doi: 10.1002/jhet.1897..
- ^ Herndon JL, Ismaiel A, Ingher SP, Teitler M, Glennon RA (December 1992). "Ketanserin analogues: structure-affinity relationships for 5-HT2 and 5-HT1C serotonin receptor binding". Journal of Medicinal Chemistry. 35 (26): 4903–10. doi: 10.1021/jm00104a017. PMID 1479590.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Sufrexal |
| Other names | R41468; R-41468; R-41,468 |
| AHFS/ Drugs.com | International Drug Names |
|
Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 50% [1] [2] |
| Protein binding | 95.0% (mainly albumin [2] |
| Elimination half-life | 10–29 hours [3] [1] [2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.070.598 |
| Chemical and physical data | |
| Formula | C22H22FN3O3 |
| Molar mass | 395.434 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Ketanserin ( INN, USAN, BAN) (brand name Sufrexal; former developmental code name R41468) is a drug used clinically as an antihypertensive agent and in scientific research to study the serotonergic system; specifically, the 5-HT2 receptor family. [4] It was discovered at Janssen Pharmaceutica in 1980. [5] [6] It is not available in the United States. [1]
Uses
Medical uses
Ketanserin is classified as an antihypertensive by the World Health Organization [7] and the National Institute of Health. [8]
It has been used to reverse pulmonary hypertension caused by protamine (which in turn was administered to reverse the effects of heparin overdose). [9]
The reduction in hypertension is not associated with reflex tachycardia. [10]
It has been used in cardiac surgery. [11]
A 2000 Cochrane Review found that, compared to placebo, ketanserin did not provide significant relief for people suffering from Raynaud's phenomenon attacks in the setting of progressive systemic sclerosis (an autoimmune disorder). While the frequency of the attacks was unaffected by ketanserin, there was a reduction in the duration of the individual attacks. However, due to the significant adverse effect burden, the authors concluded that ketanserin's utility for this indication is likely unbeneficial. [12]
Ketanserin is a selective 5-HT2A receptor antagonist that was initially developed as an anti-hypertensive medicine. However, now the drug is available as a topical gel formulation for treating wounds, burns, ulcers, and anal fissures. Its action is through the acceleration of epithelialization.
Research uses
With tritium (3H) radioactively labeled ketanserin is used as a radioligand for serotonin 5-HT2 receptors, e.g. in receptor binding assays and autoradiography. [13] This radio-labeling has enabled the study of serotonin 5-HT2A receptor distribution in the human brain. [14]
An autoradiography study of the human cerebellum has found an increasing binding of 3H-ketanserin with age (from below 50 femtomol per milligram tissue at around 30 years of age to over 100 above 75 years). [15] The same research team found no significant correlation with age in their homogenate binding study.
Ketanserin has also been used with carbon (11C) radioactively labeled NNC112 in order to image cortical D1 receptors without contamination by 5-HT2 receptors. [16]
Increasing research into the use of psychedelics as antidepressants has seen ketanserin used to both block the hallucinogenic experience, and to disentangle the specific cognitive effects of 5-HT2A activation. [17]
Pharmacology
| Target | Affinity (Ki) | Ref(s) |
|---|---|---|
| α1A-adrenergic | 6.3 nM | [19] |
| α1B-adrenergic | 6.3 nM | [19] |
| α1D-adrenergic | 16 nM | [19] |
| α2A-adrenergic | 372 nM (HT29) | [18] |
| α2B-adrenergic | 199 nM | [18] |
| α2C-adrenergic | 159 nM (opossum) | [18] |
| 5-HT1A | 1,044–>10,000 nM | [19] [18] |
| 5-HT1B | 2,515–6,300 nM | [19] [18] |
| 5-HT1D | 32–>10,000 nM | [19] [20] [21] |
| 5-HT1E | >10,000 nM | [18] |
| 5-HT1F | 1.25–>10,000 nM | [18] |
| 5-HT2A | 0.20–9.8 nM | [19] [18] |
| 5-HT2B | 200–3,236 nM | [19] [18] |
| 5-HT2C | 17–186 nM | [19] [18] |
| 5-HT3 | >10,000 nM (rodent) | [18] |
| 5-HT4L | 1,000 nM (rat) | [18] |
| 5-HT5A | 20,000 nM | [19] [18] |
| 5-HT5B | 1,000–1,585 nM (rodent) | [18] |
| 5-HT6 | 2,800 nM | [18] |
| 5-HT7 | 320–1,334 nM | [19] [18] |
| D1 | 190–464 nM | [18] |
| D2 | >10,000 nM | [18] |
| D3 | ? | |
| D4 | 148 nM (canine) | [18] |
| D5 | 2,500 nM | [19] [18] |
| H1 | 1.79 nM | [18] |
| DAT | >10,000 nM | [18] |
| VMAT1 | 1,600 nM | [19] |
| VMAT2 | 500 nM | [19] |
Pharmacodynamics
Ketanserin is a high-affinity non-selective antagonist of 5-HT2 receptors in rodents, [18] [22] [20] In addition to the 5-HT2 receptors, ketanserin is also a high affinity antagonist for the H1 receptor. [23] It has also been found to block the vesicular monoamine transporter 2 (VMAT2). [24] [25]
Pharmacokinetics
The bioavailability of ketanserin is 50%. [1] [2] The plasma protein binding of ketanserin is 95.0% and it is mainly bound to albumin. [2] The elimination half-life of ketanserin is 10 to 29 hours. [3] [1]
Synthesis

Either 3-(2-Chloroethyl)quinazoline-2,4(1H,3H)-dione [5081-87-8] (1a), or alternatively 2,3-dihydro-[1,3]oxazolo[2,3-b]quinazolin-5-one [52727-44-3] (1b) can be used as starting material. Attachment of the sidechain to 4-(4-Fluorobenzoyl)piperidine [56346-57-7] (2) completes synthesis synthesis of Ketanserin (3).
See also
References
- ^ a b c d e Wolverton SE (8 March 2007). Comprehensive Dermatologic Drug Therapy (2 ed.). Elsevier Health Sciences. ISBN 978-1-4377-2070-9.
- ^ a b c d e Hideya Saitō, Masaru Minami, eds. (1992). Antihypertensive Drugs Today. VSP. pp. 191–. ISBN 978-90-6764-140-1. OCLC 231351327.
- ^ a b Cold GE, Dahl BL (11 November 2013). Topics in Neuroanaesthesia and Neurointensive Care: Experimental and Clinical Studies upon Cerebral Circulation, Metabolism and Intracranial Pressure. Springer Science & Business Media. pp. 193–. ISBN 978-3-662-04845-0. OCLC 1076237896.
- ^ O'Donnell J, Ahuja GD (2005). Drug Injury: Liability, Analysis, and Prevention. Lawyers & Judges Publishing Company. pp. 304–. ISBN 978-0-913875-27-8.
- ^ Healy D (1 July 2009). The Creation of Psychopharmacology. Harvard University Press. pp. 252–253. ISBN 978-0-674-03845-5.
- ^ Schwartz H (August 1989). Breakthrough: the discovery of modern medicines at Janssen. Skyline Pub. Group. p. 74. ISBN 978-1-56019-100-1.
- ^ ATC/DDD Index
- ^ Ketanserin
- ^ van der Starre PJ, Solinas C (1996). "Ketanserin in the treatment of protamine-induced pulmonary hypertension". Texas Heart Institute Journal. 23 (4): 301–304. PMC 325377. PMID 8969033.
- ^ Hodsman NB, Colvin JR, Kenny GN (May 1989). "Effect of ketanserin on sodium nitroprusside requirements, arterial pressure control and heart rate following coronary artery bypass surgery". British Journal of Anaesthesia. 62 (5): 527–531. doi: 10.1093/bja/62.5.527. PMID 2786422.
- ^ Elbers PW, Ozdemir A, van Iterson M, van Dongen EP, Ince C (February 2009). "Microcirculatory Imaging in Cardiac Anesthesia: Ketanserin Reduces Blood Pressure But Not Perfused Capillary Density". Journal of Cardiothoracic and Vascular Anesthesia. 23 (1): 95–101. doi: 10.1053/j.jvca.2008.09.013. PMID 19058975.
- ^ Pope J, Fenlon D, Thompson A, Shea B, Furst D, Wells G, Silman A (2000). "Ketanserin for Raynaud's phenomenon in progressive systemic sclerosis". The Cochrane Database of Systematic Reviews. 1998 (2): CD000954. doi: 10.1002/14651858.CD000954. PMC 7032891. PMID 10796396.
- ^ Eickhoff SB, Schleicher A, Scheperjans F, Palomero-Gallagher N, Zilles K (February 2007). "Analysis of neurotransmitter receptor distribution patterns in the cerebral cortex". NeuroImage. 34 (4): 1317–1330. doi: 10.1016/j.neuroimage.2006.11.016. PMID 17182260. S2CID 23363050.
- ^ Pazos A, Probst A, Palacios JM (April 1987). "Serotonin receptors in the human brain--IV. Autoradiographic mapping of serotonin-2 receptors". Neuroscience. 21 (1): 123–139. doi: 10.1016/0306-4522(87)90327-7. PMID 3601071. S2CID 23711420.
- ^ Eastwood SL, Burnet PW, Gittins R, Baker K, Harrison PJ (November 2001). "Expression of serotonin 5-HT(2A) receptors in the human cerebellum and alterations in schizophrenia". Synapse. 42 (2): 104–114. doi: 10.1002/syn.1106. PMID 11574947. S2CID 40304220.
- ^ Catafau AM, Searle GE, Bullich S, Gunn RN, Rabiner EA, Herance R, et al. (May 2010). "Imaging cortical dopamine D1 receptors using [11C]NNC112 and ketanserin blockade of the 5-HT 2A receptors". Journal of Cerebral Blood Flow and Metabolism. 30 (5): 985–993. doi: 10.1038/jcbfm.2009.269. PMC 2949183. PMID 20029452.
- ^ Quednow BB, Kometer M, Geyer MA, Vollenweider FX (February 2012). "Psilocybin-induced deficits in automatic and controlled inhibition are attenuated by ketanserin in healthy human volunteers". Neuropsychopharmacology. 37 (3): 630–640. doi: 10.1038/npp.2011.228. PMC 3260978. PMID 21956447.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x NIMH Psychoactive Drug Screening Program
- ^ a b c d e f g h i j k l m n o "Ketanserin Ligand page". IUPHAR/BPS Guide to PHARMACOLOGY.
- ^ a b Meneses A (11 March 2014). The Role of 5-HT Systems on Memory and Dysfunctional Memory: Emergent Targets for Memory Formation and Memory Alterations. Elsevier Science. pp. 23–. ISBN 978-0-12-801083-9.
- ^ Olivier B, van Wijngaarden I, Soudijn W (10 July 1997). Serotonin Receptors and their Ligands. Elsevier. pp. 118–. ISBN 978-0-08-054111-2.
- ^ Creed-Carson M, Oraha A, Nobrega JN (June 2011). "Effects of 5-HT(2A) and 5-HT(2C) receptor antagonists on acute and chronic dyskinetic effects induced by haloperidol in rats". Behavioural Brain Research. 219 (2): 273–279. doi: 10.1016/j.bbr.2011.01.025. PMID 21262266. S2CID 205882443.
- ^ Coyne CP (9 January 2008). Comparative Diagnostic Pharmacology: Clinical and Research Applications in Living-System Models. John Wiley & Sons. pp. 104–. ISBN 978-0-470-34429-3.
- ^ Muller CP, Jacobs B (30 December 2009). Handbook of the Behavioral Neurobiology of Serotonin. Academic Press. pp. 592–. ISBN 978-0-08-087817-1.
- ^ Henry JP, Sagné C, Botton D, Isambert MF, Gasnier B (1998). Molecular Pharmacology of the Vesicular Monoamine Transporter. Advances in Pharmacology. Vol. 42. San Diego, Calif.: Academic Press. pp. 236–9 (237). doi: 10.1016/s1054-3589(08)60736-x. ISBN 978-0-08-058134-7. PMID 9327887.
- ^ US 4335127, Vandenberk J, Kennis L, Van der Aa M, Van Heertum A, issued 1982, assigned to Janssen Pharmaceutica, N.V.
- ^ EP 0098499, Signorini R, Verga A, issued 1984, assigned to Ravizza SpA
- ^ CN 106866625, Shiwen R, et al., issued 2017, assigned to Shanghai Ding Ya Pharmaceutical Chemistry Science And Technology Ltd)
- ^ Fakhraian H, Heydary M (January 2014). "Reinvestigation of the Synthesis of Ketanserin (5) and its Hydrochloride Salt (5. HCl) via 3-(2-Chloroethyl)-2, 4-(1H, 3H)-quinazolinedione (2) or Dihydro-5H-oxazole (2, 3-b) quinazolin-5-one (1)". Journal of Heterocyclic Chemistry. 51 (1): 151–156. doi: 10.1002/jhet.1897..
- ^ Herndon JL, Ismaiel A, Ingher SP, Teitler M, Glennon RA (December 1992). "Ketanserin analogues: structure-affinity relationships for 5-HT2 and 5-HT1C serotonin receptor binding". Journal of Medicinal Chemistry. 35 (26): 4903–10. doi: 10.1021/jm00104a017. PMID 1479590.