 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
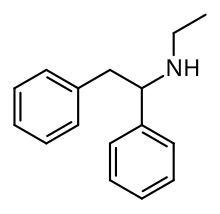
| Formula | C16H19N |
| Molar mass | 225.335 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Ephenidine (also known as NEDPA and EPE) is a dissociative anesthetic that has been sold online as a designer drug. [1] [2] It is illegal in some countries as a structural isomer of the banned opioid drug lefetamine, but has been sold in countries where it is not yet banned. [3] [4]
Pharmacology
Pharmacodynamics
Ephenidine and related diarylethylamines have been studied in vitro as treatments for neurotoxic injuries, and are antagonists of the NMDA receptor (Ki = 66.4 nM for ephenidine). [5] [6] [7] [8] [9] Ephenidine also possesses weaker affinity for dopamine and norepinephrine transporters (379 nM and 841 nM, respectively) as well as σ1R (629 nM) and σ2R (722 nM) binding sites. [10]
Pharmacokinetics
Metabolism
Ephenidine's metabolic pathway consists of N-oxidation, N-dealkylation, mono- and bis-hydroxylation of the benzyl ring, and hydroxylation of the phenyl ring only after N-dealkylation. The dihydroxy metabolites were conjugated by methylation of one hydroxy group, and hydroxy metabolites by glucuronidation or sulfation. [3] [11]
Chemistry
Ephenidine reacts with reagent testing kits to give a semi-unique array of colors which can be used to aid its identification.
| Reagent | Reaction color |
|---|---|
| Marquis | Orange > Brown |
| Mandelin | Green |
| Liebermann | Deep red > Brown (fast) |
| Froehde | Light Yellow |
Society and culture
Sweden's public health agency suggested that ephenidine be classified as a hazardous substance on 1 June, 2015. Due to that suggestion, ephenidine became a scheduled substance, in Sweden, as of 18 August, 2015. [13]
In 2016, Canada added MT-45 and "its salts, derivatives, isomers and analogues" to the Schedule I controlled substance list, and explicitly included ephenidine. [14] Possession without legal authority can result in maximum 7 years imprisonment.
See also
- AD-1211
- βk-Ephenidine
- Diphenidine
- Fluorolintane
- Lanicemine
- Methoxphenidine (MXP)
- MT-45
- NPDPA
- Propylphenidine
- Remacemide
- UWA-001
References
- ^ Morris H, Wallach J (July–August 2014). "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis. 6 (7–8): 614–32. doi: 10.1002/dta.1620. PMID 24678061.
- ^ Meyer MR, Orschiedt T, Maurer HH (February 2013). "Michaelis-Menten kinetic analysis of drugs of abuse to estimate their affinity to human P-glycoprotein". Toxicology Letters. 217 (2): 137–42. doi: 10.1016/j.toxlet.2012.12.012. PMID 23273999.
- ^ a b Wink CS, Meyer GM, Wissenbach DK, Jacobsen-Bauer A, Meyer MR, Maurer HH (October 2014). "Lefetamine-derived designer drugs N-ethyl-1,2-diphenylethylamine (NEDPA) and N-iso-propyl-1,2-diphenylethylamine (NPDPA): metabolism and detectability in rat urine using GC-MS, LC-MSn and LC-HR-MS/MS". Drug Testing and Analysis. 6 (10): 1038–48. doi: 10.1002/dta.1621. PMID 24591097.
- ^ Wink CS, Meyer GM, Meyer MR, Maurer HH (November 2015). "Toxicokinetics of lefetamine and derived diphenylethylamine designer drugs-Contribution of human cytochrome P450 isozymes to their main phase I metabolic steps". Toxicology Letters. 238 (3): 39–44. doi: 10.1016/j.toxlet.2015.08.012. PMID 26276083.
- ^ Gray NM, Cheng BK (6 April 1994). "Patent EP 0346791 - 1,2-diarylethylamines for treatment of neurotoxic injury". G.D. Searle, LLC. Archived from the original on 20 September 2018. Retrieved 11 August 2016 – via SureChEMBL.
- ^ Berger ML, Schweifer A, Rebernik P, Hammerschmidt F (May 2009). "NMDA receptor affinities of 1,2-diphenylethylamine and 1-(1,2-diphenylethyl)piperidine enantiomers and of related compounds". Bioorganic & Medicinal Chemistry. 17 (9): 3456–62. doi: 10.1016/j.bmc.2009.03.025. PMID 19345586.
- ^ Wallach J, Kavanagh PV, McLaughlin G, Morris N, Power JD, Elliott SP, et al. (May 2015). "Preparation and characterization of the 'research chemical' diphenidine, its pyrrolidine analogue, and their 2,2-diphenylethyl isomers" (PDF). Drug Testing and Analysis. 7 (5): 358–67. doi: 10.1002/dta.1689. PMID 25044512. Archived (PDF) from the original on 2020-03-07. Retrieved 2021-05-31.
- ^ Thurkauf A, Monn J, Mattson MV, Jacobson AE, Rice KC (1989). "Structural and conformational aspects of the binding of aryl-alkyl amines to the phencyclidine binding site" (PDF). NIDA Research Monograph. 95: 51–6. PMID 2561843. Archived from the original (PDF) on 2017-08-23. Retrieved 2016-08-11.
- ^ Goodson LH, Wiegand CJ, Splitter JS (November 1946). "Analgesics; n-alkylated-1,2-diphenylethylamines prepared by the Leuckart reaction". Journal of the American Chemical Society. 68 (11): 2174–2175. doi: 10.1021/ja01215a018. PMID 21002222.
- ^ Kang H, Park P, Bortolotto ZA, Brandt SD, Colestock T, Wallach J, et al. (January 2017). "Ephenidine: A new psychoactive agent with ketamine-like NMDA receptor antagonist properties". Neuropharmacology. 112 (Pt A): 144–149. doi: 10.1016/j.neuropharm.2016.08.004. PMC 5084681. PMID 27520396.
- ^ Wink CS, Meyer GM, Zapp J, Maurer HH (February 2015). "Lefetamine, a controlled drug and pharmaceutical lead of new designer drugs: synthesis, metabolism, and detectability in urine and human liver preparations using GC-MS, LC-MS(n), and LC-high resolution-MS/MS". Analytical and Bioanalytical Chemistry. 407 (6): 1545–57. doi: 10.1007/s00216-014-8414-3. PMID 25577353. S2CID 26014465.
- ^ "Ephenidine reaction results with various reagent tests". Reagent Tests UK. 17 January 2016. Archived from the original on 20 September 2018. Retrieved 23 January 2016.
- ^ "23 nya ämnen kan klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. 1 June 2015. Archived from the original on 17 June 2019. Retrieved 15 August 2016.
- ^ Arsenault D (1 June 2016). "Regulations Amending the Food and Drug Regulations (Parts G and J — Lefetamine, AH-7921, MT-45 and W-18)". Canada Gazette. 150 (11). Archived from the original on 2017-12-02. Retrieved 2016-11-17.
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C16H19N |
| Molar mass | 225.335 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Ephenidine (also known as NEDPA and EPE) is a dissociative anesthetic that has been sold online as a designer drug. [1] [2] It is illegal in some countries as a structural isomer of the banned opioid drug lefetamine, but has been sold in countries where it is not yet banned. [3] [4]
Pharmacology
Pharmacodynamics
Ephenidine and related diarylethylamines have been studied in vitro as treatments for neurotoxic injuries, and are antagonists of the NMDA receptor (Ki = 66.4 nM for ephenidine). [5] [6] [7] [8] [9] Ephenidine also possesses weaker affinity for dopamine and norepinephrine transporters (379 nM and 841 nM, respectively) as well as σ1R (629 nM) and σ2R (722 nM) binding sites. [10]
Pharmacokinetics
Metabolism
Ephenidine's metabolic pathway consists of N-oxidation, N-dealkylation, mono- and bis-hydroxylation of the benzyl ring, and hydroxylation of the phenyl ring only after N-dealkylation. The dihydroxy metabolites were conjugated by methylation of one hydroxy group, and hydroxy metabolites by glucuronidation or sulfation. [3] [11]
Chemistry
Ephenidine reacts with reagent testing kits to give a semi-unique array of colors which can be used to aid its identification.
| Reagent | Reaction color |
|---|---|
| Marquis | Orange > Brown |
| Mandelin | Green |
| Liebermann | Deep red > Brown (fast) |
| Froehde | Light Yellow |
Society and culture
Sweden's public health agency suggested that ephenidine be classified as a hazardous substance on 1 June, 2015. Due to that suggestion, ephenidine became a scheduled substance, in Sweden, as of 18 August, 2015. [13]
In 2016, Canada added MT-45 and "its salts, derivatives, isomers and analogues" to the Schedule I controlled substance list, and explicitly included ephenidine. [14] Possession without legal authority can result in maximum 7 years imprisonment.
See also
- AD-1211
- βk-Ephenidine
- Diphenidine
- Fluorolintane
- Lanicemine
- Methoxphenidine (MXP)
- MT-45
- NPDPA
- Propylphenidine
- Remacemide
- UWA-001
References
- ^ Morris H, Wallach J (July–August 2014). "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis. 6 (7–8): 614–32. doi: 10.1002/dta.1620. PMID 24678061.
- ^ Meyer MR, Orschiedt T, Maurer HH (February 2013). "Michaelis-Menten kinetic analysis of drugs of abuse to estimate their affinity to human P-glycoprotein". Toxicology Letters. 217 (2): 137–42. doi: 10.1016/j.toxlet.2012.12.012. PMID 23273999.
- ^ a b Wink CS, Meyer GM, Wissenbach DK, Jacobsen-Bauer A, Meyer MR, Maurer HH (October 2014). "Lefetamine-derived designer drugs N-ethyl-1,2-diphenylethylamine (NEDPA) and N-iso-propyl-1,2-diphenylethylamine (NPDPA): metabolism and detectability in rat urine using GC-MS, LC-MSn and LC-HR-MS/MS". Drug Testing and Analysis. 6 (10): 1038–48. doi: 10.1002/dta.1621. PMID 24591097.
- ^ Wink CS, Meyer GM, Meyer MR, Maurer HH (November 2015). "Toxicokinetics of lefetamine and derived diphenylethylamine designer drugs-Contribution of human cytochrome P450 isozymes to their main phase I metabolic steps". Toxicology Letters. 238 (3): 39–44. doi: 10.1016/j.toxlet.2015.08.012. PMID 26276083.
- ^ Gray NM, Cheng BK (6 April 1994). "Patent EP 0346791 - 1,2-diarylethylamines for treatment of neurotoxic injury". G.D. Searle, LLC. Archived from the original on 20 September 2018. Retrieved 11 August 2016 – via SureChEMBL.
- ^ Berger ML, Schweifer A, Rebernik P, Hammerschmidt F (May 2009). "NMDA receptor affinities of 1,2-diphenylethylamine and 1-(1,2-diphenylethyl)piperidine enantiomers and of related compounds". Bioorganic & Medicinal Chemistry. 17 (9): 3456–62. doi: 10.1016/j.bmc.2009.03.025. PMID 19345586.
- ^ Wallach J, Kavanagh PV, McLaughlin G, Morris N, Power JD, Elliott SP, et al. (May 2015). "Preparation and characterization of the 'research chemical' diphenidine, its pyrrolidine analogue, and their 2,2-diphenylethyl isomers" (PDF). Drug Testing and Analysis. 7 (5): 358–67. doi: 10.1002/dta.1689. PMID 25044512. Archived (PDF) from the original on 2020-03-07. Retrieved 2021-05-31.
- ^ Thurkauf A, Monn J, Mattson MV, Jacobson AE, Rice KC (1989). "Structural and conformational aspects of the binding of aryl-alkyl amines to the phencyclidine binding site" (PDF). NIDA Research Monograph. 95: 51–6. PMID 2561843. Archived from the original (PDF) on 2017-08-23. Retrieved 2016-08-11.
- ^ Goodson LH, Wiegand CJ, Splitter JS (November 1946). "Analgesics; n-alkylated-1,2-diphenylethylamines prepared by the Leuckart reaction". Journal of the American Chemical Society. 68 (11): 2174–2175. doi: 10.1021/ja01215a018. PMID 21002222.
- ^ Kang H, Park P, Bortolotto ZA, Brandt SD, Colestock T, Wallach J, et al. (January 2017). "Ephenidine: A new psychoactive agent with ketamine-like NMDA receptor antagonist properties". Neuropharmacology. 112 (Pt A): 144–149. doi: 10.1016/j.neuropharm.2016.08.004. PMC 5084681. PMID 27520396.
- ^ Wink CS, Meyer GM, Zapp J, Maurer HH (February 2015). "Lefetamine, a controlled drug and pharmaceutical lead of new designer drugs: synthesis, metabolism, and detectability in urine and human liver preparations using GC-MS, LC-MS(n), and LC-high resolution-MS/MS". Analytical and Bioanalytical Chemistry. 407 (6): 1545–57. doi: 10.1007/s00216-014-8414-3. PMID 25577353. S2CID 26014465.
- ^ "Ephenidine reaction results with various reagent tests". Reagent Tests UK. 17 January 2016. Archived from the original on 20 September 2018. Retrieved 23 January 2016.
- ^ "23 nya ämnen kan klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. 1 June 2015. Archived from the original on 17 June 2019. Retrieved 15 August 2016.
- ^ Arsenault D (1 June 2016). "Regulations Amending the Food and Drug Regulations (Parts G and J — Lefetamine, AH-7921, MT-45 and W-18)". Canada Gazette. 150 (11). Archived from the original on 2017-12-02. Retrieved 2016-11-17.