 | |
| Clinical data | |
|---|---|
| Trade names | Several |
| Other names | ST-1191; McN-A-2673-11 |
|
Routes of administration | By mouth |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
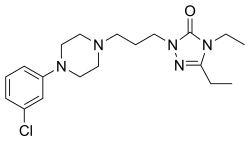
| Formula | C19H28ClN5O |
| Molar mass | 377.92 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Etoperidone, associated with several brand names, is an atypical antidepressant which was developed in the 1970s and either is no longer marketed or was never marketed. [1] [2] [3] It is a phenylpiperazine related to trazodone and nefazodone in chemical structure and is a serotonin antagonist and reuptake inhibitor (SARI) similarly to them. [4]
Medical uses
Etoperidone was used or was intended for use as an antidepressant in the treatment of depression. [1] [5]
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | 890 | Human | [7] |
| NET | 20,000 | Human | [7] |
| DAT | 52,000 | Human | [7] |
| 5-HT1A | 85 | Human | [8] |
| 5-HT2A | 36 | Human | [8] |
| 5-HT2C | ? | ? | ? |
| α1 | 38 | Human | [8] |
| α2 | 570 | Human | [8] |
| D2 | 2,300 | Human | [8] |
| H1 | 3,100 | Human | [8] |
| mACh | >35,000 | Human | [8] [9] |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
Etoperidone is as an antagonist of several receptors in the following order of potency: 5-HT2A receptor (36 nM) > α1-adrenergic receptor (38 nM) > 5-HT1A receptor (85 nM) (may be a partial agonist) > α2-adrenergic receptor (570 nM); [8] it has only very weak or negligible affinity for blocking the following receptors: D2 receptor (2,300 nM) > H1 receptor (3,100 nM) > mACh receptors (>35,000 nM). [8] In addition to its receptor blockade, etoperidone also has weak affinity for the monoamine transporters as well: serotonin transporter (890 nM) > norepinephrine transporter (20,000 nM) > dopamine transporter (52,000 nM). [7]
Pharmacokinetics
Etoperidone is metabolized in part to meta-chlorophenylpiperazine (mCPP), which likely accounts for its serotonergic effects. [10] [11]
Chemistry
Etoperidone is a phenylpiperazine and is chemically related to nefazodone and trazodone. [3] [12] [13]
History
Etoperidone was discovered by scientists at Angelini, who also discovered trazodone. [14] Its development names have included ST-1191 and McN-A-2673-11. [15] [1] The INN etoperidone was proposed in 1976 and recommended in 1977. [16] [17] The drug was given brand names in Spain (Centren (Esteve) and Depraser (Lepori)) and Italy (Staff (Sigma Tau)) [1] and was also given the brand names Axiomin and Etonin, [15] but it is not entirely clear if it was actually marketed; the Pharmaceutical Manufacturing Encyclopedia provides no dates for commercial introduction. [18] According to Micromedex's Index Nominum: International Drug Directory, etoperidone was indeed previously marketed in Spain and Italy. [1]
Society and culture
Generic names
Etoperidone is the generic name of the drug and its INN, while etoperidone hydrochloride is its USAN. [15] [1] [5]
Brand names
Etoperidone has been associated with the brand names Axiomin, Centren, Depraser, Etonin, and Staff. [1] [15] [18]
Research
Etoperidone has been studied in dementia and found to be about as effective as thioridazine. [19]
See also
References
- ^ a b c d e f g Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. p. 421. ISBN 978-3-88763-075-1.
- ^ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia. Elsevier. pp. 1533–. ISBN 978-0-8155-1856-3.
- ^ a b Akritopoulou-Zanze I (16 August 2012). "Arylpiperazine-Based 5-HT1A Receptor Partial Agonists and 5-HT2A Antagonists for the Treatment of Autism, Depression, Anxiety, Psychosis, and Schizophrenia". In Lamberth C, Dinges J (eds.). Bioactive Heterocyclic Compound Classes: Pharmaceuticals. John Wiley & Sons. pp. 81–97. doi: 10.1002/9783527664450.ch6. ISBN 978-3-527-66447-4.
- ^ Morrison-Valfre M (23 August 2016). Foundations of Mental Health Care - E-Book. Elsevier Health Sciences. pp. 245–. ISBN 978-0-323-37104-9.
- ^ a b Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 117–. ISBN 978-94-011-4439-1.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ a b c d Tatsumi M, Groshan K, Blakely RD, Richelson E (1997). "Pharmacological profile of antidepressants and related compounds at human monoamine transporters". Eur. J. Pharmacol. 340 (2–3): 249–58. doi: 10.1016/s0014-2999(97)01393-9. PMID 9537821.
- ^ a b c d e f g h i Cusack B, Nelson A, Richelson E (1994). "Binding of antidepressants to human brain receptors: focus on newer generation compounds". Psychopharmacology. 114 (4): 559–65. doi: 10.1007/bf02244985. PMID 7855217. S2CID 21236268.
- ^ Stanton T, Bolden-Watson C, Cusack B, Richelson E (1993). "Antagonism of the five cloned human muscarinic cholinergic receptors expressed in CHO-K1 cells by antidepressants and antihistaminics". Biochem. Pharmacol. 45 (11): 2352–4. doi: 10.1016/0006-2952(93)90211-e. PMID 8100134.
- ^ Garattini S (1985). "Active drug metabolites. An overview of their relevance in clinical pharmacokinetics". Clinical Pharmacokinetics. 10 (3): 216–227. doi: 10.2165/00003088-198510030-00002. PMID 2861928. S2CID 21305772.
- ^ Raffa RB, Shank RP, Vaught JL (1992). "Etoperidone, trazodone and MCPP: in vitro and in vivo identification of serotonin 5-HT1A (antagonistic) activity". Psychopharmacology. 108 (3): 320–326. doi: 10.1007/BF02245118. PMID 1387963. S2CID 24965789.
- ^ Dörwald FZ, ed. (2012). "46. Arylalkylamines". Lead optimization for medicinal chemists : pharmacokinetic properties of functional groups and organic compounds. Weinheim: Wiley-VCH. ISBN 9783527645640.
- ^ Eison MS, Taylor DB, Riblet LA (1987). "Atypical Psychotropic Agents". In Williams M, Malick JB (eds.). Drug Discovery and Development. Springer Science & Business Media. p. 390. ISBN 9781461248286.
- ^ Silvestrini B (1986). "Trazodone and the mental pain hypothesis of depression". Neuropsychobiology. 15 (Suppl 1): 2–9. doi: 10.1159/000118270. PMID 3014372.
- ^ a b c d Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 527–. ISBN 978-1-4757-2085-3.
- ^ "Proposed INN List 36" (PDF). Supplement to the WHO Chronicle. 30 (9). 1976.
- ^ "Recommended INN List 17" (PDF). Supplement to the WHO Chronicle. 31 (10). 1977.
- ^ a b McPherson EM (2007). Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Burlington: Elsevier. p. 1533. ISBN 978-0-8155-1856-3.
- ^ Kirchner V, Kelly CA, Harvey RJ (2001). "Thioridazine for dementia". The Cochrane Database of Systematic Reviews (3): CD000464. doi: 10.1002/14651858.CD000464. PMC 7034526. PMID 11686961.
External links
-
 Media related to
Etoperidone at Wikimedia Commons
Media related to
Etoperidone at Wikimedia Commons
 | |
| Clinical data | |
|---|---|
| Trade names | Several |
| Other names | ST-1191; McN-A-2673-11 |
|
Routes of administration | By mouth |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C19H28ClN5O |
| Molar mass | 377.92 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Etoperidone, associated with several brand names, is an atypical antidepressant which was developed in the 1970s and either is no longer marketed or was never marketed. [1] [2] [3] It is a phenylpiperazine related to trazodone and nefazodone in chemical structure and is a serotonin antagonist and reuptake inhibitor (SARI) similarly to them. [4]
Medical uses
Etoperidone was used or was intended for use as an antidepressant in the treatment of depression. [1] [5]
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | 890 | Human | [7] |
| NET | 20,000 | Human | [7] |
| DAT | 52,000 | Human | [7] |
| 5-HT1A | 85 | Human | [8] |
| 5-HT2A | 36 | Human | [8] |
| 5-HT2C | ? | ? | ? |
| α1 | 38 | Human | [8] |
| α2 | 570 | Human | [8] |
| D2 | 2,300 | Human | [8] |
| H1 | 3,100 | Human | [8] |
| mACh | >35,000 | Human | [8] [9] |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
Etoperidone is as an antagonist of several receptors in the following order of potency: 5-HT2A receptor (36 nM) > α1-adrenergic receptor (38 nM) > 5-HT1A receptor (85 nM) (may be a partial agonist) > α2-adrenergic receptor (570 nM); [8] it has only very weak or negligible affinity for blocking the following receptors: D2 receptor (2,300 nM) > H1 receptor (3,100 nM) > mACh receptors (>35,000 nM). [8] In addition to its receptor blockade, etoperidone also has weak affinity for the monoamine transporters as well: serotonin transporter (890 nM) > norepinephrine transporter (20,000 nM) > dopamine transporter (52,000 nM). [7]
Pharmacokinetics
Etoperidone is metabolized in part to meta-chlorophenylpiperazine (mCPP), which likely accounts for its serotonergic effects. [10] [11]
Chemistry
Etoperidone is a phenylpiperazine and is chemically related to nefazodone and trazodone. [3] [12] [13]
History
Etoperidone was discovered by scientists at Angelini, who also discovered trazodone. [14] Its development names have included ST-1191 and McN-A-2673-11. [15] [1] The INN etoperidone was proposed in 1976 and recommended in 1977. [16] [17] The drug was given brand names in Spain (Centren (Esteve) and Depraser (Lepori)) and Italy (Staff (Sigma Tau)) [1] and was also given the brand names Axiomin and Etonin, [15] but it is not entirely clear if it was actually marketed; the Pharmaceutical Manufacturing Encyclopedia provides no dates for commercial introduction. [18] According to Micromedex's Index Nominum: International Drug Directory, etoperidone was indeed previously marketed in Spain and Italy. [1]
Society and culture
Generic names
Etoperidone is the generic name of the drug and its INN, while etoperidone hydrochloride is its USAN. [15] [1] [5]
Brand names
Etoperidone has been associated with the brand names Axiomin, Centren, Depraser, Etonin, and Staff. [1] [15] [18]
Research
Etoperidone has been studied in dementia and found to be about as effective as thioridazine. [19]
See also
References
- ^ a b c d e f g Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. p. 421. ISBN 978-3-88763-075-1.
- ^ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia. Elsevier. pp. 1533–. ISBN 978-0-8155-1856-3.
- ^ a b Akritopoulou-Zanze I (16 August 2012). "Arylpiperazine-Based 5-HT1A Receptor Partial Agonists and 5-HT2A Antagonists for the Treatment of Autism, Depression, Anxiety, Psychosis, and Schizophrenia". In Lamberth C, Dinges J (eds.). Bioactive Heterocyclic Compound Classes: Pharmaceuticals. John Wiley & Sons. pp. 81–97. doi: 10.1002/9783527664450.ch6. ISBN 978-3-527-66447-4.
- ^ Morrison-Valfre M (23 August 2016). Foundations of Mental Health Care - E-Book. Elsevier Health Sciences. pp. 245–. ISBN 978-0-323-37104-9.
- ^ a b Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 117–. ISBN 978-94-011-4439-1.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ a b c d Tatsumi M, Groshan K, Blakely RD, Richelson E (1997). "Pharmacological profile of antidepressants and related compounds at human monoamine transporters". Eur. J. Pharmacol. 340 (2–3): 249–58. doi: 10.1016/s0014-2999(97)01393-9. PMID 9537821.
- ^ a b c d e f g h i Cusack B, Nelson A, Richelson E (1994). "Binding of antidepressants to human brain receptors: focus on newer generation compounds". Psychopharmacology. 114 (4): 559–65. doi: 10.1007/bf02244985. PMID 7855217. S2CID 21236268.
- ^ Stanton T, Bolden-Watson C, Cusack B, Richelson E (1993). "Antagonism of the five cloned human muscarinic cholinergic receptors expressed in CHO-K1 cells by antidepressants and antihistaminics". Biochem. Pharmacol. 45 (11): 2352–4. doi: 10.1016/0006-2952(93)90211-e. PMID 8100134.
- ^ Garattini S (1985). "Active drug metabolites. An overview of their relevance in clinical pharmacokinetics". Clinical Pharmacokinetics. 10 (3): 216–227. doi: 10.2165/00003088-198510030-00002. PMID 2861928. S2CID 21305772.
- ^ Raffa RB, Shank RP, Vaught JL (1992). "Etoperidone, trazodone and MCPP: in vitro and in vivo identification of serotonin 5-HT1A (antagonistic) activity". Psychopharmacology. 108 (3): 320–326. doi: 10.1007/BF02245118. PMID 1387963. S2CID 24965789.
- ^ Dörwald FZ, ed. (2012). "46. Arylalkylamines". Lead optimization for medicinal chemists : pharmacokinetic properties of functional groups and organic compounds. Weinheim: Wiley-VCH. ISBN 9783527645640.
- ^ Eison MS, Taylor DB, Riblet LA (1987). "Atypical Psychotropic Agents". In Williams M, Malick JB (eds.). Drug Discovery and Development. Springer Science & Business Media. p. 390. ISBN 9781461248286.
- ^ Silvestrini B (1986). "Trazodone and the mental pain hypothesis of depression". Neuropsychobiology. 15 (Suppl 1): 2–9. doi: 10.1159/000118270. PMID 3014372.
- ^ a b c d Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 527–. ISBN 978-1-4757-2085-3.
- ^ "Proposed INN List 36" (PDF). Supplement to the WHO Chronicle. 30 (9). 1976.
- ^ "Recommended INN List 17" (PDF). Supplement to the WHO Chronicle. 31 (10). 1977.
- ^ a b McPherson EM (2007). Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Burlington: Elsevier. p. 1533. ISBN 978-0-8155-1856-3.
- ^ Kirchner V, Kelly CA, Harvey RJ (2001). "Thioridazine for dementia". The Cochrane Database of Systematic Reviews (3): CD000464. doi: 10.1002/14651858.CD000464. PMC 7034526. PMID 11686961.
External links
-
 Media related to
Etoperidone at Wikimedia Commons
Media related to
Etoperidone at Wikimedia Commons