 | |
| Clinical data | |
|---|---|
| Trade names | Hytrin, Zayasel, others |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a693046 |
| License data | |
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 90–94% |
| Elimination half-life | 12 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.118.191 |
| Chemical and physical data | |
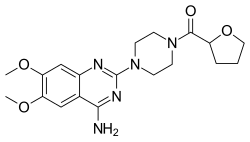
| Formula | C19H25N5O4 |
| Molar mass | 387.440 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Terazosin, sold under the brand name Hytrin among others, is a medication used to treat symptoms of an enlarged prostate and high blood pressure. [1] For high blood pressure, it is a less preferred option. [1] It is taken by mouth. [1]
Common side effects include dizziness, headache, tiredness, swelling, nausea, and low blood pressure with standing. [1] Severe side effects may include priapism and low blood pressure. [1] Prostate cancer should be ruled out before starting treatment. [1] It is an alpha-1 blocker and works by relaxing blood vessels and the opening of the bladder. [1]
Terazosin was patented in 1975 and came into medical use in 1985. [2] It is available as a generic medication. [3] In 2021, it was the 234th most commonly prescribed medication in the United States, with more than 1 million prescriptions. [4] [5]
Synthesis
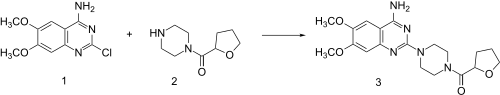
Reaction of piperazine with 2-furoyl chloride followed by catalytic hydrogenation of the furan ring leads to 2. This, when heated in the presence of 2-chloro-6,7-dimethoxyquinazolin-4-amine (1) undergoes direct alkylation to terazosin (3).
Research
A 2022 study suggests that terazosin may have the potential to confer neuroprotection upon motor neurons in motor neuron disease, as a result of its ability to activate PGK1. [7]
References
- ^ a b c d e f g "Terazosin Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 17 March 2019.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 455. ISBN 9783527607495.
- ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 768. ISBN 9780857113382.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Terazosin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ US 4026894, Winn M, Kyncl J, Dunnigan DA, Jones PH, issued 31 May 1977, assigned to Abbott
- ^ Chaytow H, Carroll E, Gordon D, Huang YT, van der Hoorn D, Smith HL, et al. (September 2022). "Targeting phosphoglycerate kinase 1 with terazosin improves motor neuron phenotypes in multiple models of amyotrophic lateral sclerosis". eBioMedicine. 83: 104202. doi: 10.1016/j.ebiom.2022.104202. PMC 9482929. PMID 35963713.
 | |
| Clinical data | |
|---|---|
| Trade names | Hytrin, Zayasel, others |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a693046 |
| License data | |
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 90–94% |
| Elimination half-life | 12 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.118.191 |
| Chemical and physical data | |
| Formula | C19H25N5O4 |
| Molar mass | 387.440 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Terazosin, sold under the brand name Hytrin among others, is a medication used to treat symptoms of an enlarged prostate and high blood pressure. [1] For high blood pressure, it is a less preferred option. [1] It is taken by mouth. [1]
Common side effects include dizziness, headache, tiredness, swelling, nausea, and low blood pressure with standing. [1] Severe side effects may include priapism and low blood pressure. [1] Prostate cancer should be ruled out before starting treatment. [1] It is an alpha-1 blocker and works by relaxing blood vessels and the opening of the bladder. [1]
Terazosin was patented in 1975 and came into medical use in 1985. [2] It is available as a generic medication. [3] In 2021, it was the 234th most commonly prescribed medication in the United States, with more than 1 million prescriptions. [4] [5]
Synthesis

Reaction of piperazine with 2-furoyl chloride followed by catalytic hydrogenation of the furan ring leads to 2. This, when heated in the presence of 2-chloro-6,7-dimethoxyquinazolin-4-amine (1) undergoes direct alkylation to terazosin (3).
Research
A 2022 study suggests that terazosin may have the potential to confer neuroprotection upon motor neurons in motor neuron disease, as a result of its ability to activate PGK1. [7]
References
- ^ a b c d e f g "Terazosin Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 17 March 2019.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 455. ISBN 9783527607495.
- ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 768. ISBN 9780857113382.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Terazosin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ US 4026894, Winn M, Kyncl J, Dunnigan DA, Jones PH, issued 31 May 1977, assigned to Abbott
- ^ Chaytow H, Carroll E, Gordon D, Huang YT, van der Hoorn D, Smith HL, et al. (September 2022). "Targeting phosphoglycerate kinase 1 with terazosin improves motor neuron phenotypes in multiple models of amyotrophic lateral sclerosis". eBioMedicine. 83: 104202. doi: 10.1016/j.ebiom.2022.104202. PMC 9482929. PMID 35963713.