 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C15H14N2 |
| Molar mass | 222.291 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
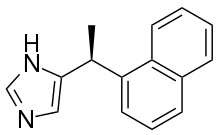
4-NEMD is a potent sedative drug which acts as a selective alpha-2 adrenergic agonist. It is closely related to dexmedetomidine but is several times more potent. [1] Like other alpha-2 agonists, it produces sedative and muscle relaxant effects but without producing respiratory depression. It is not currently used in medicine but has been researched as the basis for a potential new generation of alpha-2 agonist drugs, [2] [3] which may have selectivity for the different subtypes of the alpha-2 receptor. [4] It has two isomers, with the (S) isomer being the more potent, as with medetomidine. [5] 4-NEMD was also investigated by the United States military as an anaesthetic agent, most likely for use in surgery but possibly also for use as a non-lethal incapacitating agent, although this has not been officially confirmed. [6]
References
- ^ Hong SS, Romstedt KJ, Feller DR, Hsu FL, Cupps TL, Lyon RA, Miller DD (July 1994). "A structure-activity relationship study of benzylic modifications of 4-[1-(1-naphthyl)ethyl]-1H-imidazoles on alpha 1- and alpha 2-adrenergic receptors". Journal of Medicinal Chemistry. 37 (15): 2328–33. doi: 10.1021/jm00041a011. PMID 7914537.
- ^ Zhang X, Yao XT, Dalton JT, Shams G, Lei L, Patil PN, et al. (July 1996). "Medetomidine analogs as alpha 2-adrenergic ligands. 2. Design, synthesis, and biological activity of conformationally restricted naphthalene derivatives of medetomidine". Journal of Medicinal Chemistry. 39 (15): 3001–13. doi: 10.1021/jm9506074. PMID 8709134.
- ^ Zhang X, De Los Angeles JE, He MY, Dalton JT, Shams G, Lei L, et al. (September 1997). "Medetomidine analogs as alpha 2-adrenergic ligands. 3. Synthesis and biological evaluation of a new series of medetomidine analogs and their potential binding interactions with alpha 2-adrenoceptors involving a "methyl pocket"". Journal of Medicinal Chemistry. 40 (19): 3014–24. doi: 10.1021/jm960642q. PMID 9301663.
- ^ Lalchandani SG, Zhang X, Hong SS, Liggett SB, Li W, Moore BM, et al. (January 2004). "Medetomidine analogs as selective agonists for the human alpha2-adrenoceptors". Biochemical Pharmacology. 67 (1): 87–96. doi: 10.1016/j.bcp.2003.08.043. PMID 14667931.
- ^ Hong SS, Romstedt KJ, Feller DR, Hsu FL, George C, Cupps TL, et al. (1992). "Resolution and adrenergic activities of the optical isomers of 4-[1-(1-naphthyl)ethyl]-1H-imidazole". Chirality. 4 (7): 432–8. doi: 10.1002/chir.530040706. PMID 1361151.
- ^ US 5151526, Hsu FL, Ashman WP, "4-(1-(1-naphthalenyl)ethyl)-1H- imidazole, method of making and use as an anesthetic agent."
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C15H14N2 |
| Molar mass | 222.291 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
4-NEMD is a potent sedative drug which acts as a selective alpha-2 adrenergic agonist. It is closely related to dexmedetomidine but is several times more potent. [1] Like other alpha-2 agonists, it produces sedative and muscle relaxant effects but without producing respiratory depression. It is not currently used in medicine but has been researched as the basis for a potential new generation of alpha-2 agonist drugs, [2] [3] which may have selectivity for the different subtypes of the alpha-2 receptor. [4] It has two isomers, with the (S) isomer being the more potent, as with medetomidine. [5] 4-NEMD was also investigated by the United States military as an anaesthetic agent, most likely for use in surgery but possibly also for use as a non-lethal incapacitating agent, although this has not been officially confirmed. [6]
References
- ^ Hong SS, Romstedt KJ, Feller DR, Hsu FL, Cupps TL, Lyon RA, Miller DD (July 1994). "A structure-activity relationship study of benzylic modifications of 4-[1-(1-naphthyl)ethyl]-1H-imidazoles on alpha 1- and alpha 2-adrenergic receptors". Journal of Medicinal Chemistry. 37 (15): 2328–33. doi: 10.1021/jm00041a011. PMID 7914537.
- ^ Zhang X, Yao XT, Dalton JT, Shams G, Lei L, Patil PN, et al. (July 1996). "Medetomidine analogs as alpha 2-adrenergic ligands. 2. Design, synthesis, and biological activity of conformationally restricted naphthalene derivatives of medetomidine". Journal of Medicinal Chemistry. 39 (15): 3001–13. doi: 10.1021/jm9506074. PMID 8709134.
- ^ Zhang X, De Los Angeles JE, He MY, Dalton JT, Shams G, Lei L, et al. (September 1997). "Medetomidine analogs as alpha 2-adrenergic ligands. 3. Synthesis and biological evaluation of a new series of medetomidine analogs and their potential binding interactions with alpha 2-adrenoceptors involving a "methyl pocket"". Journal of Medicinal Chemistry. 40 (19): 3014–24. doi: 10.1021/jm960642q. PMID 9301663.
- ^ Lalchandani SG, Zhang X, Hong SS, Liggett SB, Li W, Moore BM, et al. (January 2004). "Medetomidine analogs as selective agonists for the human alpha2-adrenoceptors". Biochemical Pharmacology. 67 (1): 87–96. doi: 10.1016/j.bcp.2003.08.043. PMID 14667931.
- ^ Hong SS, Romstedt KJ, Feller DR, Hsu FL, George C, Cupps TL, et al. (1992). "Resolution and adrenergic activities of the optical isomers of 4-[1-(1-naphthyl)ethyl]-1H-imidazole". Chirality. 4 (7): 432–8. doi: 10.1002/chir.530040706. PMID 1361151.
- ^ US 5151526, Hsu FL, Ashman WP, "4-(1-(1-naphthalenyl)ethyl)-1H- imidazole, method of making and use as an anesthetic agent."