This article has multiple issues. Please help
improve it or discuss these issues on the
talk page. (
Learn how and when to remove these template messages)
|
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
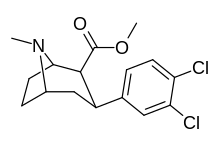
| Formula | C15H17Cl2NO2 |
| Molar mass | 314.21 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Dichloropane ((−)-2β-Carbomethoxy-3β-(3,4-dichlorophenyl)tropane, RTI-111, O-401) is a stimulant of the phenyltropane class that acts as a serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI) with IC50 values of 3.13, 18, and 0.79 nM, respectively. [1] In animal studies, dichloropane had a slower onset and longer duration of action compared to cocaine. [2] [3]
Methylecgonidine is the direct precursor to this compound. [4]
Trans -CO2Me group
The thermodynamic isomer with a trans -CO2Me group is still active. This isomer was used by Neurosearch to make three different phenyltropanes which were tested in clinical trials.
- Tesofensine
- Brasofensine
- NS-2359 (GSK-372,475)
See also
References
- ^ Carroll FI, Blough BE, Nie Z, Kuhar MJ, Howell LL, Navarro HA (April 2005). "Synthesis and monoamine transporter binding properties of 3beta-(3',4'-disubstituted phenyl)tropane-2beta-carboxylic acid methyl esters". Journal of Medicinal Chemistry. 48 (8): 2767–71. doi: 10.1021/jm040185a. PMID 15828814.
- ^ Ranaldi R, Anderson KG, Carroll FI, Woolverton WL (December 2000). "Reinforcing and discriminative stimulus effects of RTI 111, a 3-phenyltropane analog, in rhesus monkeys: interaction with methamphetamine". Psychopharmacology. 153 (1): 103–10. doi: 10.1007/s002130000602. PMID 11255920. S2CID 29716872.
- ^ Cook CD, Carroll IF, Beardsley PM (December 2001). "Cocaine-like discriminative stimulus effects of novel cocaine and 3-phenyltropane analogs in the rat". Psychopharmacology. 159 (1): 58–63. doi: 10.1007/s002130100891. PMID 11797070. S2CID 25696981.
- ^ Carroll FI, Mascarella SW, Kuzemko MA, Gao Y, Abraham P, Lewin AH, et al. (September 1994). "Synthesis, ligand binding, and QSAR (CoMFA and classical) study of 3 beta-(3'-substituted phenyl)-, 3 beta-(4'-substituted phenyl)-, and 3 beta-(3',4'-disubstituted phenyl)tropane-2 beta-carboxylic acid methyl esters". Journal of Medicinal Chemistry. 37 (18): 2865–73. doi: 10.1021/jm00044a007. PMID 8071935.
This article has multiple issues. Please help
improve it or discuss these issues on the
talk page. (
Learn how and when to remove these template messages)
|
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C15H17Cl2NO2 |
| Molar mass | 314.21 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Dichloropane ((−)-2β-Carbomethoxy-3β-(3,4-dichlorophenyl)tropane, RTI-111, O-401) is a stimulant of the phenyltropane class that acts as a serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI) with IC50 values of 3.13, 18, and 0.79 nM, respectively. [1] In animal studies, dichloropane had a slower onset and longer duration of action compared to cocaine. [2] [3]
Methylecgonidine is the direct precursor to this compound. [4]
Trans -CO2Me group
The thermodynamic isomer with a trans -CO2Me group is still active. This isomer was used by Neurosearch to make three different phenyltropanes which were tested in clinical trials.
- Tesofensine
- Brasofensine
- NS-2359 (GSK-372,475)
See also
References
- ^ Carroll FI, Blough BE, Nie Z, Kuhar MJ, Howell LL, Navarro HA (April 2005). "Synthesis and monoamine transporter binding properties of 3beta-(3',4'-disubstituted phenyl)tropane-2beta-carboxylic acid methyl esters". Journal of Medicinal Chemistry. 48 (8): 2767–71. doi: 10.1021/jm040185a. PMID 15828814.
- ^ Ranaldi R, Anderson KG, Carroll FI, Woolverton WL (December 2000). "Reinforcing and discriminative stimulus effects of RTI 111, a 3-phenyltropane analog, in rhesus monkeys: interaction with methamphetamine". Psychopharmacology. 153 (1): 103–10. doi: 10.1007/s002130000602. PMID 11255920. S2CID 29716872.
- ^ Cook CD, Carroll IF, Beardsley PM (December 2001). "Cocaine-like discriminative stimulus effects of novel cocaine and 3-phenyltropane analogs in the rat". Psychopharmacology. 159 (1): 58–63. doi: 10.1007/s002130100891. PMID 11797070. S2CID 25696981.
- ^ Carroll FI, Mascarella SW, Kuzemko MA, Gao Y, Abraham P, Lewin AH, et al. (September 1994). "Synthesis, ligand binding, and QSAR (CoMFA and classical) study of 3 beta-(3'-substituted phenyl)-, 3 beta-(4'-substituted phenyl)-, and 3 beta-(3',4'-disubstituted phenyl)tropane-2 beta-carboxylic acid methyl esters". Journal of Medicinal Chemistry. 37 (18): 2865–73. doi: 10.1021/jm00044a007. PMID 8071935.