 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
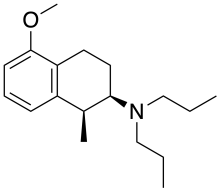
| Formula | C18H29NO |
| Molar mass | 275.436 g·mol−1 |
| 3D model ( JSmol) | |
| |
| (verify) | |
UH-232 ((+)-UH232) is a drug which acts as a subtype selective mixed agonist-antagonist for dopamine receptors, acting as a weak partial agonist at the D3 subtype, [1] and an antagonist at D2Sh autoreceptors on dopaminergic nerve terminals. [2] [3] [4] It causes dopamine release in the brain and has a stimulant effect, [5] [6] [7] as well as blocking the behavioural effects of cocaine. [8] It may also serve as a 5-HT2A receptor agonist, based on animal studies. [9] It was investigated in clinical trials for the treatment of schizophrenia, but unexpectedly caused symptoms to become worse. [9]
(+)-AJ76
The N-monopropyl derivative (+)-AJ76 is an active metabolite of UH-232 and has practically identical effects.

See also
References
- ^ Griffon N, Pilon C, Schwartz JC, Sokoloff P (August 1995). "The preferential dopamine D3 receptor ligand, (+)-UH232, is a partial agonist". European Journal of Pharmacology. 282 (1–3): R3-4. doi: 10.1016/0014-2999(95)00460-3. PMID 7498261.
- ^ Svensson K, Johansson AM, Magnusson T, Carlsson A (November 1986). "(+)-AJ 76 and (+)-UH 232: central stimulants acting as preferential dopamine autoreceptor antagonists". Naunyn-Schmiedeberg's Archives of Pharmacology. 334 (3): 234–45. doi: 10.1007/BF00508777. PMID 2880302. S2CID 6686333.
- ^ Waters N, Lagerkvist S, Löfberg L, Piercey M, Carlsson A (September 1993). "The dopamine D3 receptor and autoreceptor preferring antagonists (+)-AJ76 and (+)-UH232; a microdialysis study". European Journal of Pharmacology. 242 (2): 151–63. doi: 10.1016/0014-2999(93)90075-S. PMID 8253112.
- ^ Aretha CW, Sinha A, Galloway MP (August 1995). "Dopamine D3-preferring ligands act at synthesis modulating autoreceptors". The Journal of Pharmacology and Experimental Therapeutics. 274 (2): 609–13. PMID 7636720.
- ^ Waters N, Hansson L, Löfberg L, Carlsson A (January 1994). "Intracerebral infusion of (+)-AJ76 and (+)-UH232: effects on dopamine release and metabolism in vivo". European Journal of Pharmacology. 251 (2–3): 181–90. doi: 10.1016/0014-2999(94)90399-9. PMID 8149975.
- ^ Sotnikova TD, Gainetdinov RR, Grekhova TV, Rayevsky KS (March 2001). "Effects of intrastriatal infusion of D2 and D3 dopamine receptor preferring antagonists on dopamine release in rat dorsal striatum (in vivo microdialysis study)". Pharmacological Research. 43 (3): 283–90. doi: 10.1006/phrs.2000.0773. PMID 11401421.
- ^ Millan MJ, Seguin L, Gobert A, Cussac D, Brocco M (July 2004). "The role of dopamine D3 compared with D2 receptors in the control of locomotor activity: a combined behavioural and neurochemical analysis with novel, selective antagonists in rats". Psychopharmacology. 174 (3): 341–57. doi: 10.1007/s00213-003-1770-x. PMID 14985929. S2CID 1592299.
- ^ Piercey MF, Lum JT, Hoffmann WE, Carlsson A, Ljung E, Svensson K (August 1992). "Antagonism of cocaine's pharmacological effects by the stimulant dopaminergic antagonists, (+)-AJ76 and (+)-UH232". Brain Research. 588 (2): 217–22. doi: 10.1016/0006-8993(92)91578-3. PMID 1393576. S2CID 44478271.
- ^ a b Lahti AC, Weiler M, Carlsson A, Tamminga CA (1998). "Effects of the D3 and autoreceptor-preferring dopamine antagonist (+)-UH232 in schizophrenia". Journal of Neural Transmission. 105 (6–7): 719–34. doi: 10.1007/s007020050091. PMID 9826114. S2CID 31976950.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C18H29NO |
| Molar mass | 275.436 g·mol−1 |
| 3D model ( JSmol) | |
| |
| (verify) | |
UH-232 ((+)-UH232) is a drug which acts as a subtype selective mixed agonist-antagonist for dopamine receptors, acting as a weak partial agonist at the D3 subtype, [1] and an antagonist at D2Sh autoreceptors on dopaminergic nerve terminals. [2] [3] [4] It causes dopamine release in the brain and has a stimulant effect, [5] [6] [7] as well as blocking the behavioural effects of cocaine. [8] It may also serve as a 5-HT2A receptor agonist, based on animal studies. [9] It was investigated in clinical trials for the treatment of schizophrenia, but unexpectedly caused symptoms to become worse. [9]
(+)-AJ76
The N-monopropyl derivative (+)-AJ76 is an active metabolite of UH-232 and has practically identical effects.

See also
References
- ^ Griffon N, Pilon C, Schwartz JC, Sokoloff P (August 1995). "The preferential dopamine D3 receptor ligand, (+)-UH232, is a partial agonist". European Journal of Pharmacology. 282 (1–3): R3-4. doi: 10.1016/0014-2999(95)00460-3. PMID 7498261.
- ^ Svensson K, Johansson AM, Magnusson T, Carlsson A (November 1986). "(+)-AJ 76 and (+)-UH 232: central stimulants acting as preferential dopamine autoreceptor antagonists". Naunyn-Schmiedeberg's Archives of Pharmacology. 334 (3): 234–45. doi: 10.1007/BF00508777. PMID 2880302. S2CID 6686333.
- ^ Waters N, Lagerkvist S, Löfberg L, Piercey M, Carlsson A (September 1993). "The dopamine D3 receptor and autoreceptor preferring antagonists (+)-AJ76 and (+)-UH232; a microdialysis study". European Journal of Pharmacology. 242 (2): 151–63. doi: 10.1016/0014-2999(93)90075-S. PMID 8253112.
- ^ Aretha CW, Sinha A, Galloway MP (August 1995). "Dopamine D3-preferring ligands act at synthesis modulating autoreceptors". The Journal of Pharmacology and Experimental Therapeutics. 274 (2): 609–13. PMID 7636720.
- ^ Waters N, Hansson L, Löfberg L, Carlsson A (January 1994). "Intracerebral infusion of (+)-AJ76 and (+)-UH232: effects on dopamine release and metabolism in vivo". European Journal of Pharmacology. 251 (2–3): 181–90. doi: 10.1016/0014-2999(94)90399-9. PMID 8149975.
- ^ Sotnikova TD, Gainetdinov RR, Grekhova TV, Rayevsky KS (March 2001). "Effects of intrastriatal infusion of D2 and D3 dopamine receptor preferring antagonists on dopamine release in rat dorsal striatum (in vivo microdialysis study)". Pharmacological Research. 43 (3): 283–90. doi: 10.1006/phrs.2000.0773. PMID 11401421.
- ^ Millan MJ, Seguin L, Gobert A, Cussac D, Brocco M (July 2004). "The role of dopamine D3 compared with D2 receptors in the control of locomotor activity: a combined behavioural and neurochemical analysis with novel, selective antagonists in rats". Psychopharmacology. 174 (3): 341–57. doi: 10.1007/s00213-003-1770-x. PMID 14985929. S2CID 1592299.
- ^ Piercey MF, Lum JT, Hoffmann WE, Carlsson A, Ljung E, Svensson K (August 1992). "Antagonism of cocaine's pharmacological effects by the stimulant dopaminergic antagonists, (+)-AJ76 and (+)-UH232". Brain Research. 588 (2): 217–22. doi: 10.1016/0006-8993(92)91578-3. PMID 1393576. S2CID 44478271.
- ^ a b Lahti AC, Weiler M, Carlsson A, Tamminga CA (1998). "Effects of the D3 and autoreceptor-preferring dopamine antagonist (+)-UH232 in schizophrenia". Journal of Neural Transmission. 105 (6–7): 719–34. doi: 10.1007/s007020050091. PMID 9826114. S2CID 31976950.