| |
| Names | |
|---|---|
|
IUPAC name
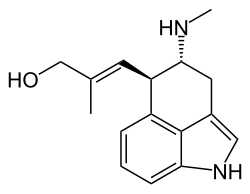
[9(9a)E]-9-Methyl-9,9a-didehydro-7,8-seco-9a-homoergolin-8-ol
| |
|
Systematic IUPAC name
(2E)-2-Methyl-3-[(4R,5R)-4-(methylamino)-1,3,4,5-tetrahydrobenzo[cd]indol-5-yl]prop-2-en-1-ol | |
| Other names
chanoclavin-l
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C16H20N2O | |
| Molar mass | 256.34 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Chanoclavine, also known as chanoclavin-l is a tri-cyclic ergot alkaloid ( ergoline) isolate of certain fungi. It is mainly produced by members of the genus claviceps. [1] Long used in traditional Chinese medicine, it was found in 1987 mouse studies to stimulate dopamine D2 receptors in the brain. [2]
See also
References
- ^ Lorenz, N; Haarmann, T; Pazoutová, S; Jung, M; Tudzynski, P (2009). "The ergot alkaloid gene cluster: Functional analyses and evolutionary aspects". Phytochemistry. 70 (15–16): 1822–32. doi: 10.1016/j.phytochem.2009.05.023. PMID 19695648.
- ^ Watanabe, H; Somei, M; Sekihara, S; Nakagawa, K; Yamada, F (1987). "Dopamine receptor stimulating effects of chanoclavine analogues, tricyclic ergot alkaloids, in the brain". Japanese Journal of Pharmacology. 45 (4): 501–6. doi: 10.1254/jjp.45.501. PMID 3127619.

| |
| Names | |
|---|---|
|
IUPAC name
[9(9a)E]-9-Methyl-9,9a-didehydro-7,8-seco-9a-homoergolin-8-ol
| |
|
Systematic IUPAC name
(2E)-2-Methyl-3-[(4R,5R)-4-(methylamino)-1,3,4,5-tetrahydrobenzo[cd]indol-5-yl]prop-2-en-1-ol | |
| Other names
chanoclavin-l
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C16H20N2O | |
| Molar mass | 256.34 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Chanoclavine, also known as chanoclavin-l is a tri-cyclic ergot alkaloid ( ergoline) isolate of certain fungi. It is mainly produced by members of the genus claviceps. [1] Long used in traditional Chinese medicine, it was found in 1987 mouse studies to stimulate dopamine D2 receptors in the brain. [2]
See also
References
- ^ Lorenz, N; Haarmann, T; Pazoutová, S; Jung, M; Tudzynski, P (2009). "The ergot alkaloid gene cluster: Functional analyses and evolutionary aspects". Phytochemistry. 70 (15–16): 1822–32. doi: 10.1016/j.phytochem.2009.05.023. PMID 19695648.
- ^ Watanabe, H; Somei, M; Sekihara, S; Nakagawa, K; Yamada, F (1987). "Dopamine receptor stimulating effects of chanoclavine analogues, tricyclic ergot alkaloids, in the brain". Japanese Journal of Pharmacology. 45 (4): 501–6. doi: 10.1254/jjp.45.501. PMID 3127619.