 | |
| Clinical data | |
|---|---|
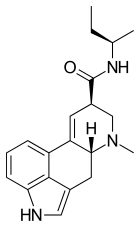
| Other names | (6aR,9R)- N- (R)- 2-butyl- 7-methyl- 4,6,6a,7,8,9- hexahydroindolo- [4,3-fg] quinoline- 9-carboxamide |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C20H25N3O |
| Molar mass | 323.440 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Lysergic acid 2-butyl amide (2-Butyllysergamide, LSB) is an analogue of LSD originally developed by Richard Pioch at Eli Lilly in the 1950s, [2] but mostly publicised through research conducted by the team led by David E. Nichols at Purdue University. It is a structural isomer of LSD, with the two ethyl groups on the amide nitrogen having been replaced by a single sec-butyl group, joined at the 2-position. [3] It is one of the few lysergamide derivatives to exceed the potency of LSD in animal drug discrimination assays, with the (R) isomer having an ED50 of 33nmol/kg for producing drug-appropriate responding, vs 48nmol/kg for LSD itself. The corresponding (R)-2-pentyl analogue has higher binding affinity for the 5-HT1A and 5-HT2A receptors, but is less potent in producing drug-appropriate responding, suggesting that the butyl compound has a higher efficacy at the receptor target. [4] The drug discrimination assay for LSD in rats involves both 5-HT1A and 5-HT2A mediated components, and while lysergic acid 2-butyl amide is more potent than LSD as a 5-HT1A agonist, it is slightly less potent as a 5-HT2A agonist, and so would probably be slightly less potent than LSD as a hallucinogen in humans. The main use for this drug has been in studies of the binding site at the 5-HT2A receptor through which LSD exerts most of its pharmacological effects, [5] with the stereoselective activity of these unsymmetric monoalkyl lysergamides foreshadowing the subsequent development of compounds such as lysergic acid 2,4-dimethylazetidide (LSZ).
See also
References
- ^ "Arrêté du 20 mai 2021 modifiant l'arrêté du 22 février 1990 fixant la liste des substances classées comme stupéfiants". www.legifrance.gouv.fr (in French). 20 May 2021.
- ^ US patent 2997470, Richard P. Pioch, "LYSERGIC ACID AMIDES", published 1956-03-05, issued 1961-08-22
- ^ Oberlender R, Pfaff RC, Johnson MP, Huang XM, Nichols DE (January 1992). "Stereoselective LSD-like activity in d-lysergic acid amides of (R)- and (S)-2-aminobutane". Journal of Medicinal Chemistry. 35 (2): 203–11. doi: 10.1021/jm00080a001. PMID 1732537.
- ^ Monte AP, Marona-Lewicka D, Kanthasamy A, Sanders-Bush E, Nichols DE (March 1995). "Stereoselective LSD-like activity in a series of d-lysergic acid amides of (R)- and (S)-2-aminoalkanes". Journal of Medicinal Chemistry. 38 (6): 958–66. doi: 10.1021/jm00006a015. PMID 7699712.
- ^ David E. Nichols. LSD and Its Lysergamide Cousins. The Heffter Review of Psychedelic Research. 2001;2:80-87.
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3– 7 |
| ||||||||||||||||||||||||||||||||||||||
|
Lysergic acid derivatives |
|
|---|---|
|
Psychedelic lysergamides |
|
| Clavines | |
| Other ergolines | |
| Natural sources |
Morning glory: Argyreia nervosa (Hawaiian Baby Woodrose), Ipomoea spp.(Morning Glory, Tlitliltzin, Badoh Negro), Rivea corymbosa (Coaxihuitl, Ololiúqui) |
- CS1 French-language sources (fr)
- Articles with short description
- Short description matches Wikidata
- Drugs with non-standard legal status
- Articles with changed ChemSpider identifier
- Articles with changed InChI identifier
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
 | |
| Clinical data | |
|---|---|
| Other names | (6aR,9R)- N- (R)- 2-butyl- 7-methyl- 4,6,6a,7,8,9- hexahydroindolo- [4,3-fg] quinoline- 9-carboxamide |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C20H25N3O |
| Molar mass | 323.440 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Lysergic acid 2-butyl amide (2-Butyllysergamide, LSB) is an analogue of LSD originally developed by Richard Pioch at Eli Lilly in the 1950s, [2] but mostly publicised through research conducted by the team led by David E. Nichols at Purdue University. It is a structural isomer of LSD, with the two ethyl groups on the amide nitrogen having been replaced by a single sec-butyl group, joined at the 2-position. [3] It is one of the few lysergamide derivatives to exceed the potency of LSD in animal drug discrimination assays, with the (R) isomer having an ED50 of 33nmol/kg for producing drug-appropriate responding, vs 48nmol/kg for LSD itself. The corresponding (R)-2-pentyl analogue has higher binding affinity for the 5-HT1A and 5-HT2A receptors, but is less potent in producing drug-appropriate responding, suggesting that the butyl compound has a higher efficacy at the receptor target. [4] The drug discrimination assay for LSD in rats involves both 5-HT1A and 5-HT2A mediated components, and while lysergic acid 2-butyl amide is more potent than LSD as a 5-HT1A agonist, it is slightly less potent as a 5-HT2A agonist, and so would probably be slightly less potent than LSD as a hallucinogen in humans. The main use for this drug has been in studies of the binding site at the 5-HT2A receptor through which LSD exerts most of its pharmacological effects, [5] with the stereoselective activity of these unsymmetric monoalkyl lysergamides foreshadowing the subsequent development of compounds such as lysergic acid 2,4-dimethylazetidide (LSZ).
See also
References
- ^ "Arrêté du 20 mai 2021 modifiant l'arrêté du 22 février 1990 fixant la liste des substances classées comme stupéfiants". www.legifrance.gouv.fr (in French). 20 May 2021.
- ^ US patent 2997470, Richard P. Pioch, "LYSERGIC ACID AMIDES", published 1956-03-05, issued 1961-08-22
- ^ Oberlender R, Pfaff RC, Johnson MP, Huang XM, Nichols DE (January 1992). "Stereoselective LSD-like activity in d-lysergic acid amides of (R)- and (S)-2-aminobutane". Journal of Medicinal Chemistry. 35 (2): 203–11. doi: 10.1021/jm00080a001. PMID 1732537.
- ^ Monte AP, Marona-Lewicka D, Kanthasamy A, Sanders-Bush E, Nichols DE (March 1995). "Stereoselective LSD-like activity in a series of d-lysergic acid amides of (R)- and (S)-2-aminoalkanes". Journal of Medicinal Chemistry. 38 (6): 958–66. doi: 10.1021/jm00006a015. PMID 7699712.
- ^ David E. Nichols. LSD and Its Lysergamide Cousins. The Heffter Review of Psychedelic Research. 2001;2:80-87.
|
Psychedelics ( 5-HT2A agonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Dissociatives ( NMDAR antagonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Deliriants ( mAChR antagonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3– 7 |
| ||||||||||||||||||||||||||||||||||||||
|
Lysergic acid derivatives |
|
|---|---|
|
Psychedelic lysergamides |
|
| Clavines | |
| Other ergolines | |
| Natural sources |
Morning glory: Argyreia nervosa (Hawaiian Baby Woodrose), Ipomoea spp.(Morning Glory, Tlitliltzin, Badoh Negro), Rivea corymbosa (Coaxihuitl, Ololiúqui) |
- CS1 French-language sources (fr)
- Articles with short description
- Short description matches Wikidata
- Drugs with non-standard legal status
- Articles with changed ChemSpider identifier
- Articles with changed InChI identifier
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields