 | |
| Clinical data | |
|---|---|
| Other names | WY-48624; D-3112 |
|
Routes of administration | Oral |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
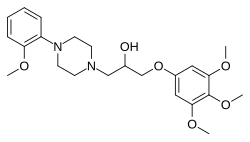
| Formula | C23H32N2O6 |
| Molar mass | 432.517 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Enciprazine ( INN, BAN; enciprazine hydrochloride ( USAN); developmental code names WY-48624, D-3112) is an anxiolytic and antipsychotic of the phenylpiperazine class which was never marketed. [1] [2] [3] [4] It shows high affinity for the α1-adrenergic receptor and 5-HT1A receptor, among other sites. [3] [5] [6] The drug was initially anticipated to produce ortho-methoxyphenylpiperazine (oMeOPP), a serotonin receptor agonist with high affinity for the 5-HT1A receptor, as a significant active metabolite, but subsequent research found this not to be the case. [5]
Synthesis

3,4,5-trimethoxyphenol (aka Antiarol) [642-71-7] (1) is alkylated with epichlorohydrin (2) to give [(3,4,5-Trimethoxyphenoxy)methyl]oxirane [74760-14-8] (3). Opening of the epoxide with o-anisyl-piperazine [35386-24-4] (4) completes the synthesis of enciprazine (5).
See also
- Acaprazine
- Batoprazine
- Eltoprazine
- Enpiprazole
- Fluprazine
- Lidanserin
- Ensaculin
- Mafoprazine
- BMY-14802
- Azaperone
- Fluanisone
References
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 485–. ISBN 978-1-4757-2085-3.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 109–. ISBN 978-94-011-4439-1.
- ^ a b Matheson GK, Knowles A, Gage D, Michel C, Guthrie D, Bauer C, Blackbourne J, Weinzapfel D (1997). "Modification of hypothalamic-pituitary-adrenocortical activity by serotonergic agents in the rat". Pharmacology. 55 (2): 59–65. doi: 10.1159/000139513. PMID 9323305.
- ^ "Enciprazine". AdisInsight. Springer Nature Switzerland AG. Retrieved 2017-06-01.
- ^ a b Scatina JA, Lockhead SR, Cayen MN, Sisenwine SF (1991). "Metabolic disposition of enciprazine, a non-benzodiazepine anxiolytic drug, in rat, dog and man". Xenobiotica. 21 (12): 1591–604. doi: 10.3109/00498259109044408. PMID 1686125.
- ^ Linden M, Helmchen H, Müller-Oerlinghausen B (1988). "Early phase-II semi double-blind study of the new alkaline propanolamine derivative enciprazine (short communication)". Arzneimittelforschung. 38 (6): 814–6. PMID 3178922.
- ^ Engel J, Jakovlev V, Kleemann A (1981). "Enciprazine". Drugs of the Future. 6 (5): 278. doi: 10.1358/dof.1981.006.05.79478.
- ^ Engel J, Fleischhauer I, Jakovlev V, Kleemann A, Kutscher B, Nickel B, Rauer H, Werner U, Szelenyi I, Johanson CE (November 1990). "Chemistry and pharmacology of the non-benzodiazepine anxiolytic enciprazine and related compounds". Journal of Medicinal Chemistry. 33 (11): 2976–81. doi: 10.1021/jm00173a012. PMID 1977910.
- ^ Banoth L, Narayan TK, Banerjee UC (September 2012). "New chemical and chemo-enzymatic routes for the synthesis of (RS)-and (S)-enciprazine". Tetrahedron: Asymmetry. 23 (17): 1272–1278. doi: 10.1016/j.tetasy.2012.08.002.
- ^ Narsaiah AV, Nagaiah B (August 2010). "A simple and efficient asymmetric synthesis of anxiolytic drug enciprazine". Synthesis. 2010 (16): 2705–2707. doi: 10.1055/s-0030-1258173.
External links
 | |
| Clinical data | |
|---|---|
| Other names | WY-48624; D-3112 |
|
Routes of administration | Oral |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C23H32N2O6 |
| Molar mass | 432.517 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Enciprazine ( INN, BAN; enciprazine hydrochloride ( USAN); developmental code names WY-48624, D-3112) is an anxiolytic and antipsychotic of the phenylpiperazine class which was never marketed. [1] [2] [3] [4] It shows high affinity for the α1-adrenergic receptor and 5-HT1A receptor, among other sites. [3] [5] [6] The drug was initially anticipated to produce ortho-methoxyphenylpiperazine (oMeOPP), a serotonin receptor agonist with high affinity for the 5-HT1A receptor, as a significant active metabolite, but subsequent research found this not to be the case. [5]
Synthesis

3,4,5-trimethoxyphenol (aka Antiarol) [642-71-7] (1) is alkylated with epichlorohydrin (2) to give [(3,4,5-Trimethoxyphenoxy)methyl]oxirane [74760-14-8] (3). Opening of the epoxide with o-anisyl-piperazine [35386-24-4] (4) completes the synthesis of enciprazine (5).
See also
- Acaprazine
- Batoprazine
- Eltoprazine
- Enpiprazole
- Fluprazine
- Lidanserin
- Ensaculin
- Mafoprazine
- BMY-14802
- Azaperone
- Fluanisone
References
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 485–. ISBN 978-1-4757-2085-3.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 109–. ISBN 978-94-011-4439-1.
- ^ a b Matheson GK, Knowles A, Gage D, Michel C, Guthrie D, Bauer C, Blackbourne J, Weinzapfel D (1997). "Modification of hypothalamic-pituitary-adrenocortical activity by serotonergic agents in the rat". Pharmacology. 55 (2): 59–65. doi: 10.1159/000139513. PMID 9323305.
- ^ "Enciprazine". AdisInsight. Springer Nature Switzerland AG. Retrieved 2017-06-01.
- ^ a b Scatina JA, Lockhead SR, Cayen MN, Sisenwine SF (1991). "Metabolic disposition of enciprazine, a non-benzodiazepine anxiolytic drug, in rat, dog and man". Xenobiotica. 21 (12): 1591–604. doi: 10.3109/00498259109044408. PMID 1686125.
- ^ Linden M, Helmchen H, Müller-Oerlinghausen B (1988). "Early phase-II semi double-blind study of the new alkaline propanolamine derivative enciprazine (short communication)". Arzneimittelforschung. 38 (6): 814–6. PMID 3178922.
- ^ Engel J, Jakovlev V, Kleemann A (1981). "Enciprazine". Drugs of the Future. 6 (5): 278. doi: 10.1358/dof.1981.006.05.79478.
- ^ Engel J, Fleischhauer I, Jakovlev V, Kleemann A, Kutscher B, Nickel B, Rauer H, Werner U, Szelenyi I, Johanson CE (November 1990). "Chemistry and pharmacology of the non-benzodiazepine anxiolytic enciprazine and related compounds". Journal of Medicinal Chemistry. 33 (11): 2976–81. doi: 10.1021/jm00173a012. PMID 1977910.
- ^ Banoth L, Narayan TK, Banerjee UC (September 2012). "New chemical and chemo-enzymatic routes for the synthesis of (RS)-and (S)-enciprazine". Tetrahedron: Asymmetry. 23 (17): 1272–1278. doi: 10.1016/j.tetasy.2012.08.002.
- ^ Narsaiah AV, Nagaiah B (August 2010). "A simple and efficient asymmetric synthesis of anxiolytic drug enciprazine". Synthesis. 2010 (16): 2705–2707. doi: 10.1055/s-0030-1258173.