 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C11H12F3NO |
| Molar mass | 231.218 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Flumexadol ( INN) (developmental code name CERM-1841 or 1841-CERM) is a drug described and researched as a non- opioid analgesic which was never marketed. [1] [2] [3] [4] It has been found to act as an agonist of the serotonin 5-HT1A (pKi = 7.1) and 5-HT2C (pKi = 7.5) receptors and, to a much lesser extent, of the 5-HT2A (pKi = 6.0) receptor. [5] [6] According to Nilsson (2006) in a paper on 5-HT2C receptor agonists as potential anorectics, "The (+)-enantiomer of this compound showed [...] affinity for the 5-HT2C receptor (Ki) 25 nM) [...] and was 40-fold selective over the 5-HT2A receptor in receptor binding studies. Curiously, the racemic version [...], also known as 1841 CERM, was originally reported to possess analgesic properties while no association with 5-HT2C receptor activity was mentioned." [4] It is implied that flumexadol might be employable as an anorectic in addition to analgesic. [4] Though flumexadol itself has never been approved for medical use, oxaflozane (brand name Conflictan) is a prodrug of the compound that was formerly used clinically in France as an antidepressant and anxiolytic agent. [5] [7] [8]
Synthesis
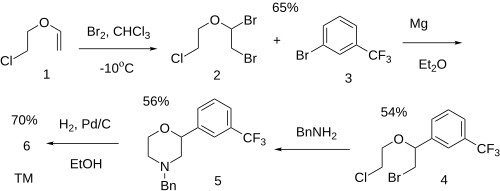
Ex 1: Halogenation of 2-chloro ethyl vinyl ether [110-75-8] (1) with molecular bromine gives 1,2-dibromo-1-(2-chloroethoxy)ethane [14689-94-2] (2). Grignard reaction with 3-bromobenzotrifluoride [401-78-5] (3) gives 1-[2-Bromo-1-(2-chloroethoxy)ethyl]-3-(trifluoromethyl)benzene, CID:12343529 (4).
Ex 4: Treatment with benzylamine gives 4-benzyl-2-[3-(trifluoromethyl) phenyl]morpholine, CID:213531 (5).
Ex 6: Catalytric hydrogenation strips the benzyl protecting group completing the synthesis of flumexadol (6).
See also
References
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 561–. ISBN 978-1-4757-2085-3.
- ^ Hache J, Diamantis W, Sofia D, Streichenberger G (1978). "The pharmacology of 1841 CERM, a new analgesic". Arzneimittel-Forschung. 28 (4): 642–645. PMID 312104.
- ^ Kucharczyk N, Yang JT, Valia KH, Stiefel FJ, Sofia RD (November 1979). "Metabolites of 2-(3-trifluoromethylphenyl)tetrahydro-1,4-oxazine (CERM) 1841) in rats and dogs". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 9 (11): 703–711. doi: 10.3109/00498257909042338. PMID 532219.
- ^ a b c Nilsson BM (July 2006). "5-Hydroxytryptamine 2C (5-HT2C) receptor agonists as potential antiobesity agents". Journal of Medicinal Chemistry. 49 (14): 4023–4034. doi: 10.1021/jm058240i. PMID 16821762.
- ^ a b Bégué JP, Bonnet-Delpon D (2 June 2008). Bioorganic and Medicinal Chemistry of Fluorine. John Wiley & Sons. pp. 303–. ISBN 978-0-470-28187-1.
- ^ Leysen DC (February 1999). "Selective 5-HT2C agonists as potential antidepressants". IDrugs. 2 (2): 109–120. PMID 16160946.
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 909–. ISBN 978-1-4757-2085-3.
- ^ Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. p. 766. ISBN 3-88763-075-0.
- ^ Busch N, Moleyre J, Simond J, Bondivenne R, Labrid C (1976). "Tetrahydro-1, 4-Oxazines. I. New Synthesis Method and a Study of Their Interaction With Tryptaminergic D Receptors". European Journal of Medicinal Chemistry. 11: 201–207.
- ^ FR 95182, "Process for the synthesis of 2-4-disubstituted tetrahydro 1,4-oxazines", assigned to Centre Europeen de Recherches Mauvernay CERM
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C11H12F3NO |
| Molar mass | 231.218 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Flumexadol ( INN) (developmental code name CERM-1841 or 1841-CERM) is a drug described and researched as a non- opioid analgesic which was never marketed. [1] [2] [3] [4] It has been found to act as an agonist of the serotonin 5-HT1A (pKi = 7.1) and 5-HT2C (pKi = 7.5) receptors and, to a much lesser extent, of the 5-HT2A (pKi = 6.0) receptor. [5] [6] According to Nilsson (2006) in a paper on 5-HT2C receptor agonists as potential anorectics, "The (+)-enantiomer of this compound showed [...] affinity for the 5-HT2C receptor (Ki) 25 nM) [...] and was 40-fold selective over the 5-HT2A receptor in receptor binding studies. Curiously, the racemic version [...], also known as 1841 CERM, was originally reported to possess analgesic properties while no association with 5-HT2C receptor activity was mentioned." [4] It is implied that flumexadol might be employable as an anorectic in addition to analgesic. [4] Though flumexadol itself has never been approved for medical use, oxaflozane (brand name Conflictan) is a prodrug of the compound that was formerly used clinically in France as an antidepressant and anxiolytic agent. [5] [7] [8]
Synthesis

Ex 1: Halogenation of 2-chloro ethyl vinyl ether [110-75-8] (1) with molecular bromine gives 1,2-dibromo-1-(2-chloroethoxy)ethane [14689-94-2] (2). Grignard reaction with 3-bromobenzotrifluoride [401-78-5] (3) gives 1-[2-Bromo-1-(2-chloroethoxy)ethyl]-3-(trifluoromethyl)benzene, CID:12343529 (4).
Ex 4: Treatment with benzylamine gives 4-benzyl-2-[3-(trifluoromethyl) phenyl]morpholine, CID:213531 (5).
Ex 6: Catalytric hydrogenation strips the benzyl protecting group completing the synthesis of flumexadol (6).
See also
References
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 561–. ISBN 978-1-4757-2085-3.
- ^ Hache J, Diamantis W, Sofia D, Streichenberger G (1978). "The pharmacology of 1841 CERM, a new analgesic". Arzneimittel-Forschung. 28 (4): 642–645. PMID 312104.
- ^ Kucharczyk N, Yang JT, Valia KH, Stiefel FJ, Sofia RD (November 1979). "Metabolites of 2-(3-trifluoromethylphenyl)tetrahydro-1,4-oxazine (CERM) 1841) in rats and dogs". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 9 (11): 703–711. doi: 10.3109/00498257909042338. PMID 532219.
- ^ a b c Nilsson BM (July 2006). "5-Hydroxytryptamine 2C (5-HT2C) receptor agonists as potential antiobesity agents". Journal of Medicinal Chemistry. 49 (14): 4023–4034. doi: 10.1021/jm058240i. PMID 16821762.
- ^ a b Bégué JP, Bonnet-Delpon D (2 June 2008). Bioorganic and Medicinal Chemistry of Fluorine. John Wiley & Sons. pp. 303–. ISBN 978-0-470-28187-1.
- ^ Leysen DC (February 1999). "Selective 5-HT2C agonists as potential antidepressants". IDrugs. 2 (2): 109–120. PMID 16160946.
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 909–. ISBN 978-1-4757-2085-3.
- ^ Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. p. 766. ISBN 3-88763-075-0.
- ^ Busch N, Moleyre J, Simond J, Bondivenne R, Labrid C (1976). "Tetrahydro-1, 4-Oxazines. I. New Synthesis Method and a Study of Their Interaction With Tryptaminergic D Receptors". European Journal of Medicinal Chemistry. 11: 201–207.
- ^ FR 95182, "Process for the synthesis of 2-4-disubstituted tetrahydro 1,4-oxazines", assigned to Centre Europeen de Recherches Mauvernay CERM