| |
| Names | |
|---|---|
|
IUPAC name
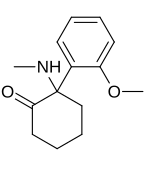
2-(2-Methoxyphenyl)-2-(methylamino)cyclohexanone
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C14H19NO2 | |
| Molar mass | 233.311 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Methoxyketamine or 2-MeO-2-deschloroketamine is a designer drug of the arylcyclohexylamine class first reported in 1963. [1] It is an analog of ketamine in which the chlorine atom has been replaced with a methoxy group. Its synthesis by rearrangement of an amino ketone has been reported. [2] As an arylcyclohexylamine, methoxyketamine most likely functions as an NMDA receptor antagonist. It produces sedative, hallucinogenic, and (at high doses) anesthetic effects, but with a lower potency than ketamine itself.
See also
References
- ^ BE 634208, Stevens, Calvin L., "Amino ketones", published 1963
- ^ Stevens, Calvin L.; Thuillier, Andre; Taylor, K. Grant; Daniher, Francis A.; Dickerson, James P.; Hanson, Harry T.; Nielsen, Norman A.; Tikotkar, N. A.; Weier, Richard M. (1966). "Amino Ketone Rearrangements. VII.1 Synthesis of 2-Methylamino-2-Substituted Phenylcyclohexanones". The Journal of Organic Chemistry. 31 (8): 2601. doi: 10.1021/jo01346a034.
| |||||||||||||||||||||||||||||||||||||
|
| This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- Chemical articles with multiple compound IDs
- Multiple chemicals in an infobox that need indexing
- Chemical articles with multiple CAS registry numbers
- Articles without EBI source
- Articles without KEGG source
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- All stub articles

| |
| Names | |
|---|---|
|
IUPAC name
2-(2-Methoxyphenyl)-2-(methylamino)cyclohexanone
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C14H19NO2 | |
| Molar mass | 233.311 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Methoxyketamine or 2-MeO-2-deschloroketamine is a designer drug of the arylcyclohexylamine class first reported in 1963. [1] It is an analog of ketamine in which the chlorine atom has been replaced with a methoxy group. Its synthesis by rearrangement of an amino ketone has been reported. [2] As an arylcyclohexylamine, methoxyketamine most likely functions as an NMDA receptor antagonist. It produces sedative, hallucinogenic, and (at high doses) anesthetic effects, but with a lower potency than ketamine itself.
See also
References
- ^ BE 634208, Stevens, Calvin L., "Amino ketones", published 1963
- ^ Stevens, Calvin L.; Thuillier, Andre; Taylor, K. Grant; Daniher, Francis A.; Dickerson, James P.; Hanson, Harry T.; Nielsen, Norman A.; Tikotkar, N. A.; Weier, Richard M. (1966). "Amino Ketone Rearrangements. VII.1 Synthesis of 2-Methylamino-2-Substituted Phenylcyclohexanones". The Journal of Organic Chemistry. 31 (8): 2601. doi: 10.1021/jo01346a034.
|
Psychedelics ( 5-HT2A agonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Dissociatives ( NMDAR antagonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Deliriants ( mAChR antagonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
|
| This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- Chemical articles with multiple compound IDs
- Multiple chemicals in an infobox that need indexing
- Chemical articles with multiple CAS registry numbers
- Articles without EBI source
- Articles without KEGG source
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- All stub articles