 | |
| Clinical data | |
|---|---|
| Other names | CP-101606 |
| ATC code |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.222.813 |
| Chemical and physical data | |
| Formula | C20H25NO3 |
| Molar mass | 327.424 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
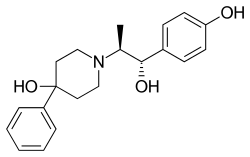
Traxoprodil (developmental code name CP-101606) is a drug developed by Pfizer which acts as an NMDA antagonist, selective for the NR2B subunit. [1] [2] It has neuroprotective, [3] analgesic, [4] and anti-Parkinsonian effects in animal studies. [5] [6] Traxoprodil has been researched in humans as a potential treatment to lessen the damage to the brain after stroke, [7] [8] [9] [10] but results from clinical trials showed only modest benefit. [11] The drug was found to cause EKG abnormalities (QT prolongation) and its clinical development was stopped. [12] More recent animal studies have suggested traxoprodil may exhibit rapid-acting antidepressant effects similar to those of ketamine, [13] although there is some evidence for similar psychoactive side effects and abuse potential at higher doses, [14] which might limit clinical acceptance of traxoprodil for this application.
Traxoprodil showed ketamine-like rapidly-acting antidepressant effects in a small clinical trial of 30 patients with depression who were non-responders to 6 weeks of paroxetine treatment. [15] The response rate was 60%, relative to 20% for placebo, and 33% of the participants met remission criteria by day five following a single administration. [15] After one week, 78% of responders still showed an antidepressant response, and after 15 days, 42% did so. [15] In the study, half of the participants had to have their dose lowered due to a high incidence of dissociative side effects at the higher doses. [15] Development was stopped due to incidence of QTc prolongation. [15] Other NR2B subunit-selective antagonists of the NMDA receptor are still under development for depression, such as rislenemdaz (CERC-301, MK-0657). [15]
See also
References
- ^ Chenard BL, Bordner J, Butler TW, Chambers LK, Collins MA, De Costa DL, Ducat MF, Dumont ML, Fox CB, Mena EE (August 1995). "(1S,2S)-1-(4-hydroxyphenyl)-2-(4-hydroxy-4-phenylpiperidino)-1-propanol: a potent new neuroprotectant which blocks N-methyl-D-aspartate responses". Journal of Medicinal Chemistry. 38 (16): 3138–45. doi: 10.1021/jm00016a017. PMID 7636876.
- ^ Brimecombe JC, Boeckman FA, Aizenman E (September 1997). "Functional consequences of NR2 subunit composition in single recombinant N-methyl-D-aspartate receptors". Proceedings of the National Academy of Sciences of the United States of America. 94 (20): 11019–24. Bibcode: 1997PNAS...9411019B. doi: 10.1073/pnas.94.20.11019. PMC 23569. PMID 9380752.
- ^ Di X, Bullock R, Watson J, Fatouros P, Chenard B, White F, Corwin F (November 1997). "Effect of CP101,606, a novel NR2B subunit antagonist of the N-methyl-D-aspartate receptor, on the volume of ischemic brain damage off cytotoxic brain edema after middle cerebral artery occlusion in the feline brain". Stroke: A Journal of Cerebral Circulation. 28 (11): 2244–51. doi: 10.1161/01.str.28.11.2244. PMID 9368572.
- ^ Taniguchi K, Shinjo K, Mizutani M, Shimada K, Ishikawa T, Menniti FS, Nagahisa A (November 1997). "Antinociceptive activity of CP-101,606, an NMDA receptor NR2B subunit antagonist". British Journal of Pharmacology. 122 (5): 809–12. doi: 10.1038/sj.bjp.0701445. PMC 1565002. PMID 9384494.
- ^ Steece-Collier K, Chambers LK, Jaw-Tsai SS, Menniti FS, Greenamyre JT (May 2000). "Antiparkinsonian actions of CP-101,606, an antagonist of NR2B subunit-containing N-methyl-d-aspartate receptors". Experimental Neurology. 163 (1): 239–43. doi: 10.1006/exnr.2000.7374. PMID 10785463. S2CID 23889057.
- ^ Nash JE, Ravenscroft P, McGuire S, Crossman AR, Menniti FS, Brotchie JM (August 2004). "The NR2B-selective NMDA receptor antagonist CP-101,606 exacerbates L-DOPA-induced dyskinesia and provides mild potentiation of anti-parkinsonian effects of L-DOPA in the MPTP-lesioned marmoset model of Parkinson's disease". Experimental Neurology. 188 (2): 471–9. doi: 10.1016/j.expneurol.2004.05.004. PMID 15246846. S2CID 41219412.
- ^ Merchant RE, Bullock MR, Carmack CA, Shah AK, Wilner KD, Ko G, Williams SA (1999). "A double-blind, placebo-controlled study of the safety, tolerability and pharmacokinetics of CP-101,606 in patients with a mild or moderate traumatic brain injury". Annals of the New York Academy of Sciences. 890 (1): 42–50. Bibcode: 1999NYASA.890...42M. doi: 10.1111/j.1749-6632.1999.tb07979.x. PMID 10668412. S2CID 25863512.
- ^ Chazot PL (November 2000). "CP-101606 Pfizer Inc". Current Opinion in Investigational Drugs. 1 (3): 370–4. PMID 11249721.
- ^ Kundrotiene J, Cebers G, Wägner A, Liljequist S (January 2004). "The NMDA NR2B subunit-selective receptor antagonist, CP-101,606, enhances the functional recovery the NMDA NR2B subunit-selective receptor and reduces brain damage after cortical compression-induced brain ischemia". Journal of Neurotrauma. 21 (1): 83–93. doi: 10.1089/089771504772695977. PMID 14987468.
- ^ Wang CX, Shuaib A (April 2005). "NMDA/NR2B selective antagonists in the treatment of ischemic brain injury". Current Drug Targets. CNS and Neurological Disorders. 4 (2): 143–51. doi: 10.2174/1568007053544183. PMID 15857299.
- ^ Yurkewicz L, Weaver J, Bullock MR, Marshall LF (December 2005). "The effect of the selective NMDA receptor antagonist traxoprodil in the treatment of traumatic brain injury". Journal of Neurotrauma. 22 (12): 1428–43. doi: 10.1089/neu.2005.22.1428. PMID 16379581.
- ^ Löscher W, Rogawski MA (2002). "Chapter 3: Epilepsy". In Lodge D, Danysz W, Parsons CG (eds.). Ionotropic Glutamate Receptors as Therapeutic Targets. FP Graham Publishing Co., Johnson City, TN. pp. 91–132.
- ^ Preskorn SH, Baker B, Kolluri S, Menniti FS, Krams M, Landen JW (December 2008). "An innovative design to establish proof of concept of the antidepressant effects of the NR2B subunit selective N-methyl-D-aspartate antagonist, CP-101,606, in patients with treatment-refractory major depressive disorder". Journal of Clinical Psychopharmacology. 28 (6): 631–7. doi: 10.1097/JCP.0b013e31818a6cea. PMID 19011431. S2CID 29461252.
- ^ Nicholson KL, Mansbach RS, Menniti FS, Balster RL (December 2007). "The phencyclidine-like discriminative stimulus effects and reinforcing properties of the NR2B-selective N-methyl-D-aspartate antagonist CP-101 606 in rats and rhesus monkeys". Behavioural Pharmacology. 18 (8): 731–43. doi: 10.1097/FBP.0b013e3282f14ed6. PMID 17989511. S2CID 32577038.
- ^ a b c d e f Machado-Vieira R, Henter ID, Zarate CA (2017). "New targets for rapid antidepressant action". Prog. Neurobiol. 152: 21–37. doi: 10.1016/j.pneurobio.2015.12.001. PMC 4919246. PMID 26724279.
 | |
| Clinical data | |
|---|---|
| Other names | CP-101606 |
| ATC code |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.222.813 |
| Chemical and physical data | |
| Formula | C20H25NO3 |
| Molar mass | 327.424 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Traxoprodil (developmental code name CP-101606) is a drug developed by Pfizer which acts as an NMDA antagonist, selective for the NR2B subunit. [1] [2] It has neuroprotective, [3] analgesic, [4] and anti-Parkinsonian effects in animal studies. [5] [6] Traxoprodil has been researched in humans as a potential treatment to lessen the damage to the brain after stroke, [7] [8] [9] [10] but results from clinical trials showed only modest benefit. [11] The drug was found to cause EKG abnormalities (QT prolongation) and its clinical development was stopped. [12] More recent animal studies have suggested traxoprodil may exhibit rapid-acting antidepressant effects similar to those of ketamine, [13] although there is some evidence for similar psychoactive side effects and abuse potential at higher doses, [14] which might limit clinical acceptance of traxoprodil for this application.
Traxoprodil showed ketamine-like rapidly-acting antidepressant effects in a small clinical trial of 30 patients with depression who were non-responders to 6 weeks of paroxetine treatment. [15] The response rate was 60%, relative to 20% for placebo, and 33% of the participants met remission criteria by day five following a single administration. [15] After one week, 78% of responders still showed an antidepressant response, and after 15 days, 42% did so. [15] In the study, half of the participants had to have their dose lowered due to a high incidence of dissociative side effects at the higher doses. [15] Development was stopped due to incidence of QTc prolongation. [15] Other NR2B subunit-selective antagonists of the NMDA receptor are still under development for depression, such as rislenemdaz (CERC-301, MK-0657). [15]
See also
References
- ^ Chenard BL, Bordner J, Butler TW, Chambers LK, Collins MA, De Costa DL, Ducat MF, Dumont ML, Fox CB, Mena EE (August 1995). "(1S,2S)-1-(4-hydroxyphenyl)-2-(4-hydroxy-4-phenylpiperidino)-1-propanol: a potent new neuroprotectant which blocks N-methyl-D-aspartate responses". Journal of Medicinal Chemistry. 38 (16): 3138–45. doi: 10.1021/jm00016a017. PMID 7636876.
- ^ Brimecombe JC, Boeckman FA, Aizenman E (September 1997). "Functional consequences of NR2 subunit composition in single recombinant N-methyl-D-aspartate receptors". Proceedings of the National Academy of Sciences of the United States of America. 94 (20): 11019–24. Bibcode: 1997PNAS...9411019B. doi: 10.1073/pnas.94.20.11019. PMC 23569. PMID 9380752.
- ^ Di X, Bullock R, Watson J, Fatouros P, Chenard B, White F, Corwin F (November 1997). "Effect of CP101,606, a novel NR2B subunit antagonist of the N-methyl-D-aspartate receptor, on the volume of ischemic brain damage off cytotoxic brain edema after middle cerebral artery occlusion in the feline brain". Stroke: A Journal of Cerebral Circulation. 28 (11): 2244–51. doi: 10.1161/01.str.28.11.2244. PMID 9368572.
- ^ Taniguchi K, Shinjo K, Mizutani M, Shimada K, Ishikawa T, Menniti FS, Nagahisa A (November 1997). "Antinociceptive activity of CP-101,606, an NMDA receptor NR2B subunit antagonist". British Journal of Pharmacology. 122 (5): 809–12. doi: 10.1038/sj.bjp.0701445. PMC 1565002. PMID 9384494.
- ^ Steece-Collier K, Chambers LK, Jaw-Tsai SS, Menniti FS, Greenamyre JT (May 2000). "Antiparkinsonian actions of CP-101,606, an antagonist of NR2B subunit-containing N-methyl-d-aspartate receptors". Experimental Neurology. 163 (1): 239–43. doi: 10.1006/exnr.2000.7374. PMID 10785463. S2CID 23889057.
- ^ Nash JE, Ravenscroft P, McGuire S, Crossman AR, Menniti FS, Brotchie JM (August 2004). "The NR2B-selective NMDA receptor antagonist CP-101,606 exacerbates L-DOPA-induced dyskinesia and provides mild potentiation of anti-parkinsonian effects of L-DOPA in the MPTP-lesioned marmoset model of Parkinson's disease". Experimental Neurology. 188 (2): 471–9. doi: 10.1016/j.expneurol.2004.05.004. PMID 15246846. S2CID 41219412.
- ^ Merchant RE, Bullock MR, Carmack CA, Shah AK, Wilner KD, Ko G, Williams SA (1999). "A double-blind, placebo-controlled study of the safety, tolerability and pharmacokinetics of CP-101,606 in patients with a mild or moderate traumatic brain injury". Annals of the New York Academy of Sciences. 890 (1): 42–50. Bibcode: 1999NYASA.890...42M. doi: 10.1111/j.1749-6632.1999.tb07979.x. PMID 10668412. S2CID 25863512.
- ^ Chazot PL (November 2000). "CP-101606 Pfizer Inc". Current Opinion in Investigational Drugs. 1 (3): 370–4. PMID 11249721.
- ^ Kundrotiene J, Cebers G, Wägner A, Liljequist S (January 2004). "The NMDA NR2B subunit-selective receptor antagonist, CP-101,606, enhances the functional recovery the NMDA NR2B subunit-selective receptor and reduces brain damage after cortical compression-induced brain ischemia". Journal of Neurotrauma. 21 (1): 83–93. doi: 10.1089/089771504772695977. PMID 14987468.
- ^ Wang CX, Shuaib A (April 2005). "NMDA/NR2B selective antagonists in the treatment of ischemic brain injury". Current Drug Targets. CNS and Neurological Disorders. 4 (2): 143–51. doi: 10.2174/1568007053544183. PMID 15857299.
- ^ Yurkewicz L, Weaver J, Bullock MR, Marshall LF (December 2005). "The effect of the selective NMDA receptor antagonist traxoprodil in the treatment of traumatic brain injury". Journal of Neurotrauma. 22 (12): 1428–43. doi: 10.1089/neu.2005.22.1428. PMID 16379581.
- ^ Löscher W, Rogawski MA (2002). "Chapter 3: Epilepsy". In Lodge D, Danysz W, Parsons CG (eds.). Ionotropic Glutamate Receptors as Therapeutic Targets. FP Graham Publishing Co., Johnson City, TN. pp. 91–132.
- ^ Preskorn SH, Baker B, Kolluri S, Menniti FS, Krams M, Landen JW (December 2008). "An innovative design to establish proof of concept of the antidepressant effects of the NR2B subunit selective N-methyl-D-aspartate antagonist, CP-101,606, in patients with treatment-refractory major depressive disorder". Journal of Clinical Psychopharmacology. 28 (6): 631–7. doi: 10.1097/JCP.0b013e31818a6cea. PMID 19011431. S2CID 29461252.
- ^ Nicholson KL, Mansbach RS, Menniti FS, Balster RL (December 2007). "The phencyclidine-like discriminative stimulus effects and reinforcing properties of the NR2B-selective N-methyl-D-aspartate antagonist CP-101 606 in rats and rhesus monkeys". Behavioural Pharmacology. 18 (8): 731–43. doi: 10.1097/FBP.0b013e3282f14ed6. PMID 17989511. S2CID 32577038.
- ^ a b c d e f Machado-Vieira R, Henter ID, Zarate CA (2017). "New targets for rapid antidepressant action". Prog. Neurobiol. 152: 21–37. doi: 10.1016/j.pneurobio.2015.12.001. PMC 4919246. PMID 26724279.