 | |
| Clinical data | |
|---|---|
| Trade names | Elanone-V |
|
Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.042.166 |
| Chemical and physical data | |
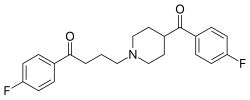
| Formula | C22H23F2NO2 |
| Molar mass | 371.428 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Lenperone (Elanone-V) is a typical antipsychotic of the butyrophenone chemical class. [1] It was first reported as an anti-emetic in 1974, [2] and its use in treatment of acute schizophrenia was reported in 1975. [1] Related early antipsychotic agents include declenperone and milenperone.
Lenperone was never approved by the FDA for use in humans in the United States, [3] but prior to 1989 it was approved for use in veterinary medicine for sedation. [4] [5] [6]
Synthesis
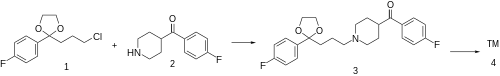
The alkylation between 2-(3-chloropropyl)-2-(4-fluorophenyl)-1,3-dioxolane [3308-94-9] (1) and 4-(4-fluorobenzoyl)piperidine [56346-57-7] (2) gives 2-(p-fluorophenyl)-2-{3-[4-(p-fluorobenzoyl)piperidino]propyl}-1,3-dioxolane, CID:20318874 (3). Deprotection of the ketal function completes the synthesis of lenperone (4).
See also
Chemically related drugs containing the same 4-(p-fluorobenzoyl)piperidine group:
References
- ^ a b Harris M (1975). "Treatment of acute schizophrenia with a new butyrophenone-lenperone". Journal of Clinical Pharmacology. 15 (2–3): 187–190. doi: 10.1002/j.1552-4604.1975.tb02355.x. PMID 1091666. S2CID 28602974.
- ^ a b FR 2227868, Ward JW, Leonard CA, "Antiemetic compositions containing piperidine derivatives", published 1974-11-29
-
^ Miller AC, Khan AM, Castro Bigalli AA, Sewell KA, King AR, Ghadermarzi S, et al. (2019).
"Neuroleptanalgesia for acute abdominal pain: a systematic review". Journal of Pain Research. 12: 787–801.
doi:
10.2147/JPR.S187798.
PMC
6396833.
PMID
30881092.
Table 1. Marketed butyrophenones with approval status and indication
- ^ Booth NJ (1982). "Psychotropic Agents". In Booth NH, McDonald LE (ed.). Veterinary Pharmacology and Therapeutics (5th ed.). Ames, Iowa: Iowa State University Press. pp. 321–345.
- ^ Johnson SE, Zelner A, Sherding RG (April 1989). "Effect of lenperone hydrochloride on gastroesophageal sphincter pressure in healthy dogs". Canadian Journal of Veterinary Research. 53 (2): 248–250. PMC 1255555. PMID 2565757.
-
^ FDA Veterinarian. U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration, Center for Veterinary Medicine. 1988.
The firm requested withdrawal of approval because the products are no longer being marketed. Effective date: July 13, 1989
- ^ Duncan RL, Helsley GC, Welstead WJ, DaVanzo JP, Funderburk WH, Lunsford CD (January 1970). "Aroylpiperidines and pyrrolidines. A new class of potent central nervous system depressants". Journal of Medicinal Chemistry. 13 (1): 1–6. doi: 10.1021/jm00295a001. PMID 5460893.
- ^ CN 107011133, Yanbiao K, Jie L, issued 2017, assigned to University of Science and Technology of China USTC.
- ^ Liu J, Hu KF, Qu JP, Kang YB (October 2017). "Organopromoted Selectivity-Switchable Synthesis of Polyketones". Organic Letters. 19 (20): 5593–5596. doi: 10.1021/acs.orglett.7b02731. PMID 28981291.
 | |
| Clinical data | |
|---|---|
| Trade names | Elanone-V |
|
Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.042.166 |
| Chemical and physical data | |
| Formula | C22H23F2NO2 |
| Molar mass | 371.428 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Lenperone (Elanone-V) is a typical antipsychotic of the butyrophenone chemical class. [1] It was first reported as an anti-emetic in 1974, [2] and its use in treatment of acute schizophrenia was reported in 1975. [1] Related early antipsychotic agents include declenperone and milenperone.
Lenperone was never approved by the FDA for use in humans in the United States, [3] but prior to 1989 it was approved for use in veterinary medicine for sedation. [4] [5] [6]
Synthesis

The alkylation between 2-(3-chloropropyl)-2-(4-fluorophenyl)-1,3-dioxolane [3308-94-9] (1) and 4-(4-fluorobenzoyl)piperidine [56346-57-7] (2) gives 2-(p-fluorophenyl)-2-{3-[4-(p-fluorobenzoyl)piperidino]propyl}-1,3-dioxolane, CID:20318874 (3). Deprotection of the ketal function completes the synthesis of lenperone (4).
See also
Chemically related drugs containing the same 4-(p-fluorobenzoyl)piperidine group:
References
- ^ a b Harris M (1975). "Treatment of acute schizophrenia with a new butyrophenone-lenperone". Journal of Clinical Pharmacology. 15 (2–3): 187–190. doi: 10.1002/j.1552-4604.1975.tb02355.x. PMID 1091666. S2CID 28602974.
- ^ a b FR 2227868, Ward JW, Leonard CA, "Antiemetic compositions containing piperidine derivatives", published 1974-11-29
-
^ Miller AC, Khan AM, Castro Bigalli AA, Sewell KA, King AR, Ghadermarzi S, et al. (2019).
"Neuroleptanalgesia for acute abdominal pain: a systematic review". Journal of Pain Research. 12: 787–801.
doi:
10.2147/JPR.S187798.
PMC
6396833.
PMID
30881092.
Table 1. Marketed butyrophenones with approval status and indication
- ^ Booth NJ (1982). "Psychotropic Agents". In Booth NH, McDonald LE (ed.). Veterinary Pharmacology and Therapeutics (5th ed.). Ames, Iowa: Iowa State University Press. pp. 321–345.
- ^ Johnson SE, Zelner A, Sherding RG (April 1989). "Effect of lenperone hydrochloride on gastroesophageal sphincter pressure in healthy dogs". Canadian Journal of Veterinary Research. 53 (2): 248–250. PMC 1255555. PMID 2565757.
-
^ FDA Veterinarian. U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration, Center for Veterinary Medicine. 1988.
The firm requested withdrawal of approval because the products are no longer being marketed. Effective date: July 13, 1989
- ^ Duncan RL, Helsley GC, Welstead WJ, DaVanzo JP, Funderburk WH, Lunsford CD (January 1970). "Aroylpiperidines and pyrrolidines. A new class of potent central nervous system depressants". Journal of Medicinal Chemistry. 13 (1): 1–6. doi: 10.1021/jm00295a001. PMID 5460893.
- ^ CN 107011133, Yanbiao K, Jie L, issued 2017, assigned to University of Science and Technology of China USTC.
- ^ Liu J, Hu KF, Qu JP, Kang YB (October 2017). "Organopromoted Selectivity-Switchable Synthesis of Polyketones". Organic Letters. 19 (20): 5593–5596. doi: 10.1021/acs.orglett.7b02731. PMID 28981291.