| |

| |
| Names | |
|---|---|
|
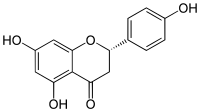
IUPAC name
(2S)-4′,5,7-Trihydroxyflavan-4-one
| |
|
Systematic IUPAC name
(2S)-5,7-Dihydroxy-2-(4-hydroxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Naringetol; Salipurol; Salipurpol
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.006.865 |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H12O5 | |
| Molar mass | 272.256 g·mol−1 |
| Melting point | 251 °C (484 °F; 524 K) [1] |
| 475 mg/L[ citation needed] | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Naringenin is a flavanone from the flavonoid group of polyphenols. It is commonly found in a variety of citrus fruits and is the predominant flavonone in grapefruit. [2] Naringenin has demonstrated numerous biological activities, including anti-inflammatory properties, antioxidant activity and skin healing. [3] [4] [5] [6] It is used as a cosmetic ingredient and dietary supplement. [7] Naringenin (along with furanocoumarins presented in citrus fruits) is thought to be responsible for CYP3A4 suppression in the intestinal wall, that may result in serious changes of pharmacokinetics in drugs related with this enzyme. [8] [9] [10]
Structure
Naringenin has the skeleton structure of a flavanone with three hydroxy groups at the 4′, 5, and 7 carbons. It may be found both in the aglycol form, naringenin, or in its glycosidic form, naringin, which has the addition of the disaccharide neohesperidose attached via a glycosidic linkage at carbon 7.
Like the majority of flavanones, naringenin has a single chiral center at carbon 2, although the optical purity is variable. [11] [12] Racemization of (S)-(−)-naringenin has been shown to occur fairly quickly. [13]
Sources and bioavailability
Naringenin and its glycoside has been found in a variety of herbs and fruits, including grapefruit, [14] bergamot, [15] sour orange, [16] sour cherries, [5] tomatoes, [17] [18] cocoa, [19] Greek oregano, [20] water mint, [21] as well as in beans. [22] Ratios of naringenin to naringin vary among sources, [17] as do enantiomeric ratios. [12]
The naringenin-7-glucoside form seems less bioavailable than the aglycol form. [23]
Grapefruit juice can provide much higher plasma concentrations of naringenin than orange juice. [24] Also found in grapefruit is the related compound kaempferol, which has a hydroxyl group next to the ketone group.
Naringenin can be absorbed from cooked tomato paste. There are 3.8 mg of naringenin in 150 grams of tomato paste. [25]
Biosynthesis and metabolism
Naringenin can be produced from naringin by the hydrolytic action of the liver enzyme naringinase. [26]
Naringenin is derived from malonyl-CoA and 4-coumaroyl-CoA. The latter is derived from phenylalanine. The resulting tetraketide is acted on by chalcone synthase to give the chalcone that then undergoes ring-closure to naringenin. [27]
The enzyme naringenin 8-dimethylallyltransferase uses dimethylallyl diphosphate and (−)-(2S)-naringenin to produce diphosphate and 8-prenylnaringenin. Cunninghamella elegans, a fungal model organism of the mammalian metabolism, can be used to study the naringenin sulfation. [28]
Potential biological effects
This section needs more
reliable medical references for
verification or relies too heavily on
primary sources. (June 2017) |  |
Studies show naringenin has numerous biological activities, including anti-inflammatory, antioxidant, antibacterial, antiviral and anticancer. It is deemed safe for both topical and ingestible use in healthy adults. [7]
Anti-inflammatory
Naringenin’s anti-inflammatory benefits have been well established with multiple in vitro and in vivo studies, revealing it effectively suppresses proinflammatory factors, cytokine and chemokine expressions in inflammation. [3] When taken orally, it has also been shown to reduce inflammatory pain. [29]
Antioxidant
Naringenin has been shown to have significant antioxidant properties. [30] [31] It has been shown to reduce oxidative damage to DNA in vitro and in animal studies. [32] [33] When consumed it has been shown to increase antioxidant markers superoxide dismutase and glutathione. [6]
Endothelium protection
Some human studies and many animal studies have shown the ability of both naringenin and naringin to protect and improve the health of the vascular endothelium. [26] Naringenin reportedly stimulates Nrf2 to protect blood vessels. [34]
Antibacterial, antifungal, and antiviral
Naringenin has an antimicrobial effect on Staphylococcus epidermidis, as well as Staphylococcus aureus, Bacillus subtilis, Micrococcus luteus, and Escherichia coli. [35] Further research has added evidence for antimicrobial effects against Lactococcus lactis, [36] lactobacillus acidophilus, Actinomyces naeslundii, Prevotella oralis, Prevotella melaninogenica, Porphyromonas gingivalis, [37] as well as yeasts such as Candida albicans, Candida tropicalis, and Candida krusei. [38] There is also evidence of antibacterial effects on Helicobacter pylori, though naringenin has not been shown to have any inhibition on urease activity of the microbe. [39]
Naringenin has also been shown to reduce hepatitis C virus production by infected hepatocytes (liver cells) in cell culture. This seems to be secondary to naringenin's ability to inhibit the secretion of very-low-density lipoprotein by the cells. [40] The antiviral effects of naringenin are currently under clinical investigation. [41] Reports of antiviral effects on polioviruses, HSV-1 and HSV-2 have also been made, though replication of the viruses has not been inhibited. [42] [43] In in vitro experiments naringenin also showed a strong antiviral activity against SARS-CoV-2. [44]
Skin Healing
When used in topical formulations, naringenin has been shown to be an anti-inflammatory with skin barrier restoration and antioxidant activities.
UVB radiation is very high energy frequency and wreak havoc on the top layers of the skin by damaging the skin cells and causing DNA mutations that can lead to melanoma and other skin cancers. [45] Naringenin has been proven to reduce UVB-induced skin damage, as well as showing efficacy against oxidative stress and improvement in wound healing. [4] [46] [6]
Naringenin has also been studied on inflammatory skin conditions such as atopic dermatitis and psoriasis. [3] [47] [48] For atopic dermatitis, naringenin was found to sharply suppress inflammatory levels and alleviate symptoms and may suppress the development of atopic dermatitis like skin lesions. [3] [47] In psoriasis, naringenin has been studied to reduce inflammation in psoriatic plaques. [48]
Anticancer
Cytotoxicity has been reduced reportedly by naringenin in cancer cells from breast, stomach, liver, cervix, pancreas, and colon tissues, along with leukaemia cells. [49] [50] The mechanisms behind inhibition of human breast carcinoma growth have been examined, and two theories have been proposed. [51] The first theory is that naringenin inhibits aromatase, thus reducing growth of the tumor. [52] The second mechanism proposes that interactions with estrogen receptors is the cause behind the modulation of growth. [53] New derivatives of naringenin were found to be active against multidrug-resistant cancer. [54]
Fatty Liver Disease
Naringenin may have some benefits for non-alcoholic fatty liver disease. It was proven to reduce hepatic lipid accumulation and inflammation in the livers of mice with non-alcoholic fatty liver disease. [55]
Alzheimer's disease
Naringenin is being researched as a potential treatment for Alzheimer's disease. Naringenin has been demonstrated to improve memory and reduce amyloid and tau proteins in a study using a mouse model of Alzheimer's disease. [56] [57] The effect is believed to be due to a protein present in neurons known as CRMP2 that naringenin binds to. [58]
Safety
Naringenin has been deemed safe to apply topically and can also be ingested safely by healthy adults in doses of 150 to 900 mg, with 300 mg of naringenin twice a day likely to elicit physiological effect. [7]
References
- ^ Naringenin at the Human Metabolome Database
- ^ Felgines C, Texier O, Morand C, Manach C, Scalbert A, Régerat F, Rémésy C (December 2000). "Bioavailability of the flavanone naringenin and its glycosides in rats" (PDF). American Journal of Physiology. Gastrointestinal and Liver Physiology. 279 (6): G1148–G1154. doi: 10.1152/ajpgi.2000.279.6.G1148. PMID 11093936. S2CID 27540043.
- ^ a b c d Karuppagounder V, Arumugam S, Thandavarayan RA, Sreedhar R, Giridharan VV, Pitchaimani V, et al. (May 2016). "Naringenin ameliorates skin inflammation and accelerates phenotypic reprogramming from M1 to M2 macrophage polarization in atopic dermatitis NC/Nga mouse model". Experimental Dermatology. 25 (5): 404–407. doi: 10.1111/exd.12962. PMID 26836240.
- ^ a b Martinez RM, Pinho-Ribeiro FA, Steffen VS, Silva TC, Caviglione CV, Bottura C, et al. (2016-01-07). "Topical Formulation Containing Naringenin: Efficacy against Ultraviolet B Irradiation-Induced Skin Inflammation and Oxidative Stress in Mice". PLOS ONE. 11 (1): e0146296. Bibcode: 2016PLoSO..1146296M. doi: 10.1371/journal.pone.0146296. PMC 4704734. PMID 26741806.
- ^ a b Wang H, Nair MG, Strasburg GM, Booren AM, Gray JI (March 1999). "Antioxidant polyphenols from tart cherries (Prunus cerasus)". Journal of Agricultural and Food Chemistry. 47 (3): 840–844. doi: 10.1021/jf980936f. PMID 10552377.
- ^ a b c Al-Roujayee AS (April 2017). "Naringenin improves the healing process of thermally-induced skin damage in rats". The Journal of International Medical Research. 45 (2): 570–582. doi: 10.1177/0300060517692483. PMC 5536686. PMID 28415935.
- ^ a b c Rebello CJ, Beyl RA, Lertora JJ, Greenway FL, Ravussin E, Ribnicky DM, et al. (January 2020). "Safety and pharmacokinetics of naringenin: A randomized, controlled, single-ascending-dose clinical trial". Diabetes, Obesity & Metabolism. 22 (1): 91–98. doi: 10.1111/dom.13868. PMC 6956701. PMID 31468636.
- ^ Lohezic-Le Devehat, F.; Marigny, K.; Doucet, M.; Javaudin, L. (2002). "[Grapefruit juice and drugs: a hazardous combination?]". Therapie. 57 (5): 432–445. ISSN 0040-5957. PMID 12611197.
- ^ Singh, B. N. (September 1999). "Effects of food on clinical pharmacokinetics". Clinical Pharmacokinetics. 37 (3): 213–255. doi: 10.2165/00003088-199937030-00003. ISSN 0312-5963. PMID 10511919.
- ^ Fuhr, U. (April 1998). "Drug interactions with grapefruit juice. Extent, probable mechanism and clinical relevance". Drug Safety. 18 (4): 251–272. doi: 10.2165/00002018-199818040-00002. ISSN 0114-5916. PMID 9565737.
- ^ Yáñez JA, Andrews PK, Davies NM (April 2007). "Methods of analysis and separation of chiral flavonoids". Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences. 848 (2): 159–181. doi: 10.1016/j.jchromb.2006.10.052. PMID 17113835.
- ^ a b Yáñez JA, Remsberg CM, Miranda ND, Vega-Villa KR, Andrews PK, Davies NM (March 2008). "Pharmacokinetics of selected chiral flavonoids: hesperetin, naringenin and eriodictyol in rats and their content in fruit juices". Biopharmaceutics & Drug Disposition. 29 (2): 63–82. doi: 10.1002/bdd.588. PMID 18058792. S2CID 24051610.
- ^ Krause M, Galensa R (July 1991). "Analysis of enantiomeric flavanones in plant extracts by high-performance liquid chromatography on a cellulose triacetate based chiral stationary phase". Chromatographia. 32 (1–2): 69–72. doi: 10.1007/BF02262470. ISSN 0009-5893. S2CID 95215634.
- ^ Ho PC, Saville DJ, Coville PF, Wanwimolruk S (April 2000). "Content of CYP3A4 inhibitors, naringin, naringenin and bergapten in grapefruit and grapefruit juice products". Pharmaceutica Acta Helvetiae. 74 (4): 379–385. doi: 10.1016/S0031-6865(99)00062-X. PMID 10812937.
- ^ Gattuso G, Barreca D, Gargiulli C, Leuzzi U, Caristi C (August 2007). "Flavonoid composition of Citrus juices". Molecules. 12 (8): 1641–1673. doi: 10.3390/12081641. PMC 6149096. PMID 17960080.
- ^ Gel-Moreto N, Streich R, Galensa R (August 2003). "Chiral separation of diastereomeric flavanone-7-O-glycosides in citrus by capillary electrophoresis". Electrophoresis. 24 (15): 2716–2722. doi: 10.1002/elps.200305486. PMID 12900888. S2CID 40261445.
- ^ a b Minoggio M, Bramati L, Simonetti P, Gardana C, Iemoli L, Santangelo E, et al. (2003-01-01). "Polyphenol pattern and antioxidant activity of different tomato lines and cultivars". Annals of Nutrition & Metabolism. 47 (2): 64–69. doi: 10.1159/000069277. PMID 12652057. S2CID 26333030.
- ^ Vallverdú Queralt A, Odriozola Serrano I, Oms Oliu G, Lamuela Raventós RM, Elez Martínez P, Martín Belloso O (September 2012). "Changes in the polyphenol profile of tomato juices processed by pulsed electric fields". Journal of Agricultural and Food Chemistry. 60 (38): 9667–9672. doi: 10.1021/jf302791k. PMID 22957841.
- ^ Sánchez Rabaneda F, Jáuregui O, Casals I, Andrés Lacueva C, Izquierdo Pulido M, Lamuela Raventós RM (January 2003). "Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao)". Journal of Mass Spectrometry. 38 (1): 35–42. Bibcode: 2003JMSp...38...35S. doi: 10.1002/jms.395. PMID 12526004.
- ^ Exarchou V, Godejohann M, van Beek TA, Gerothanassis IP, Vervoort J (November 2003). "LC-UV-solid-phase extraction-NMR-MS combined with a cryogenic flow probe and its application to the identification of compounds present in Greek oregano". Analytical Chemistry. 75 (22): 6288–6294. doi: 10.1021/ac0347819. PMID 14616013.
- ^ Olsen HT, Stafford GI, van Staden J, Christensen SB, Jäger AK (May 2008). "Isolation of the MAO-inhibitor naringenin from Mentha aquatica L". Journal of Ethnopharmacology. 117 (3): 500–502. doi: 10.1016/j.jep.2008.02.015. PMID 18372132.
- ^ Hungria M, Johnston AW, Phillips DA (1992-05-01). "Effects of flavonoids released naturally from bean (Phaseolus vulgaris) on nodD-regulated gene transcription in Rhizobium leguminosarum bv. phaseoli". Molecular Plant-Microbe Interactions. 5 (3): 199–203. doi: 10.1094/mpmi-5-199. PMID 1421508.
- ^ Choudhury R, Chowrimootoo G, Srai K, Debnam E, Rice-Evans CA (November 1999). "Interactions of the flavonoid naringenin in the gastrointestinal tract and the influence of glycosylation". Biochemical and Biophysical Research Communications. 265 (2): 410–415. doi: 10.1006/bbrc.1999.1695. PMID 10558881.
- ^ Erlund I, Meririnne E, Alfthan G, Aro A (February 2001). "Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice". The Journal of Nutrition. 131 (2): 235–241. doi: 10.1093/jn/131.2.235. PMID 11160539.
- ^ Bugianesi R, Catasta G, Spigno P, D'Uva A, Maiani G (November 2002). "Naringenin from cooked tomato paste is bioavailable in men". The Journal of Nutrition. 132 (11): 3349–3352. doi: 10.1093/jn/132.11.3349. PMID 12421849.
- ^ a b Adetunji JA, Fasae KD, Sekhoacha MP (2023). "The protective roles of citrus flavonoids, naringenin, and naringin on endothelial cell dysfunction in diseases". Heliyon. 9 (6): e17166. Bibcode: 2023Heliy...917166A. doi: 10.1016/j.heliyon.2023.e17166. PMC 10361329. PMID 37484296.
- ^ Wang C, Zhi S, Liu C, Xu F, Zhao A, Wang X, et al. (March 2017). "Characterization of Stilbene Synthase Genes in Mulberry (Morus atropurpurea) and Metabolic Engineering for the Production of Resveratrol in Escherichia coli". Journal of Agricultural and Food Chemistry. 65 (8): 1659–1668. doi: 10.1021/acs.jafc.6b05212. PMID 28168876.
- ^ Ibrahim AR (January 2000). "Sulfation of naringenin by Cunninghamella elegans". Phytochemistry. 53 (2): 209–212. Bibcode: 2000PChem..53..209I. doi: 10.1016/S0031-9422(99)00487-2. PMID 10680173.
- ^ Pinho Ribeiro FA, Zarpelon AC, Fattori V, Manchope MF, Mizokami SS, Casagrande R, Verri WA (June 2016). "Naringenin reduces inflammatory pain in mice". Neuropharmacology. 105: 508–519. doi: 10.1016/j.neuropharm.2016.02.019. PMID 26907804. S2CID 35925963.
- ^ Gorinstein S, Leontowicz H, Leontowicz M, Krzemiński R, Gralak M, Delgado-Licon E, et al. (April 2005). "Changes in plasma lipid and antioxidant activity in rats as a result of naringin and red grapefruit supplementation". Journal of Agricultural and Food Chemistry. 53 (8): 3223–3228. doi: 10.1021/jf058014h. PMID 15826081.
- ^ Yu J, Wang L, Walzem RL, Miller EG, Pike LM, Patil BS (March 2005). "Antioxidant activity of citrus limonoids, flavonoids, and coumarins". Journal of Agricultural and Food Chemistry. 53 (6): 2009–2014. doi: 10.1021/jf0484632. PMID 15769128.
- ^ Kumar S, Tiku AB (March 2016). "Biochemical and Molecular Mechanisms of Radioprotective Effects of Naringenin, a Phytochemical from Citrus Fruits". Journal of Agricultural and Food Chemistry. 64 (8): 1676–1685. doi: 10.1021/acs.jafc.5b05067. PMID 26881453.
- ^ Jagetia GC, Reddy TK, Venkatesha VA, Kedlaya R (September 2004). "Influence of naringin on ferric iron induced oxidative damage in vitro". Clinica Chimica Acta; International Journal of Clinical Chemistry. 347 (1–2): 189–197. doi: 10.1016/j.cccn.2004.04.022. PMID 15313158.
- ^ Zhang Q, Liu J, Wu C (2021). "Activation of Nrf2/HO-1 signaling: An important molecular mechanism of herbal medicine in the treatment of atherosclerosis via the protection of vascular endothelial cells from oxidative stress". Journal of Advanced Research. 34: 43–63. doi: 10.1016/j.jare.2021.06.023. PMC 8655139. PMID 35024180.
- ^ Rauha JP, Remes S, Heinonen M, Hopia A, Kähkönen M, Kujala T, et al. (May 2000). "Antimicrobial effects of Finnish plant extracts containing flavonoids and other phenolic compounds". International Journal of Food Microbiology. 56 (1): 3–12. doi: 10.1016/S0168-1605(00)00218-X. PMID 10857921.
- ^ Mandalari G, Bennett RN, Bisignano G, Trombetta D, Saija A, Faulds CB, et al. (December 2007). "Antimicrobial activity of flavonoids extracted from bergamot (Citrus bergamia Risso) peel, a byproduct of the essential oil industry". Journal of Applied Microbiology. 103 (6): 2056–2064. doi: 10.1111/j.1365-2672.2007.03456.x. PMID 18045389. S2CID 2043029.
- ^ Koru Ö, Toksoy F, Açıkel CH, Tunca YM, Baysallar M, Üsküdar Güçlü A, et al. (2007-06-01). "In vitro antimicrobial activity of propolis samples from different geographical origins against certain oral pathogens". Anaerobe. 13 (3–4): 140–145. doi: 10.1016/j.anaerobe.2007.02.001. PMID 17475517.
- ^ Uzel A, Sorkun K, Önçağ Ö, Coǧulu D, Gençay Ö, Salih B (2005-04-25). "Chemical compositions and antimicrobial activities of four different Anatolian propolis samples". Microbiological Research. 160 (2): 189–195. doi: 10.1016/j.micres.2005.01.002. hdl: 11655/19951. PMID 15881836.
- ^ Bae EA, Han MJ, Kim DH (June 1999). "In vitro anti-Helicobacter pylori activity of some flavonoids and their metabolites". Planta Medica. 65 (5): 442–443. doi: 10.1055/s-2006-960805. PMID 10454900. S2CID 260284591.
- ^ Nahmias Y, Goldwasser J, Casali M, van Poll D, Wakita T, Chung RT, Yarmush ML (May 2008). "Apolipoprotein B-dependent hepatitis C virus secretion is inhibited by the grapefruit flavonoid naringenin". Hepatology. 47 (5): 1437–1445. doi: 10.1002/hep.22197. PMC 4500072. PMID 18393287.
- ^ "A Pilot Study of the Grapefruit Flavonoid Naringenin for HCV Infection". clinicaltrials.gov. Archived from the original on 2010-10-01.
- ^ Mucsi I, Prágai BM (July 1985). "Inhibition of virus multiplication and alteration of cyclic AMP level in cell cultures by flavonoids". Experientia. 41 (7): 930–931. doi: 10.1007/BF01970018. PMID 2989000. S2CID 6174141.
- ^ Lyu SY, Rhim JY, Park WB (November 2005). "Antiherpetic activities of flavonoids against herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) in vitro". Archives of Pharmacal Research. 28 (11): 1293–1301. doi: 10.1007/BF02978215. PMID 16350858. S2CID 34495208.
- ^ Clementi N, Scagnolari C, D'Amore A, Palombi F, Criscuolo E, Frasca F, et al. (January 2021). "Naringenin is a powerful inhibitor of SARS-CoV-2 infection in vitro". Pharmacological Research. 163: 105255. doi: 10.1016/j.phrs.2020.105255. PMC 7574776. PMID 33096221.
- ^ Alexander H. "What's the difference between UVA and UVB rays?". MD Anderson Cancer Center. Retrieved 2024-01-02.
- ^ Salehi M, Ehterami A, Farzamfar S, Vaez A, Ebrahimi-Barough S (February 2021). "Accelerating healing of excisional wound with alginate hydrogel containing naringenin in rat model". Drug Delivery and Translational Research. 11 (1): 142–153. doi: 10.1007/s13346-020-00731-6. PMID 32086788. S2CID 211234647.
- ^ a b Kim TH, Kim GD, Ahn HJ, Cho JJ, Park YS, Park CS (October 2013). "The inhibitory effect of naringenin on atopic dermatitis induced by DNFB in NC/Nga mice". Life Sciences. 93 (15): 516–524. doi: 10.1016/j.lfs.2013.07.027. PMID 23933131.
- ^ a b Alalaiwe A, Lin CF, Hsiao CY, Chen EL, Lin CY, Lien WC, Fang JY (May 2020). "Development of flavanone and its derivatives as topical agents against psoriasis: The prediction of therapeutic efficiency through skin permeation evaluation and cell-based assay". International Journal of Pharmaceutics. 581: 119256. doi: 10.1016/j.ijpharm.2020.119256. PMID 32220586. S2CID 214694347.
- ^ Kanno S, Tomizawa A, Hiura T, Osanai Y, Shouji A, Ujibe M, et al. (March 2005). "Inhibitory effects of naringenin on tumor growth in human cancer cell lines and sarcoma S-180-implanted mice". Biological & Pharmaceutical Bulletin. 28 (3): 527–530. doi: 10.1248/bpb.28.527. PMID 15744083.
- ^ Hermawan A, Ikawati M, Jenie RI, Khumaira A, Putri H, Nurhayati IP, et al. (January 2021). "Identification of potential therapeutic target of naringenin in breast cancer stem cells inhibition by bioinformatics and in vitro studies". Saudi Pharmaceutical Journal. 29 (1): 12–26. doi: 10.1016/j.jsps.2020.12.002. PMC 7873751. PMID 33603536.
- ^ So FV, Guthrie N, Chambers AF, Moussa M, Carroll KK (1996-01-01). "Inhibition of human breast cancer cell proliferation and delay of mammary tumorigenesis by flavonoids and citrus juices". Nutrition and Cancer. 26 (2): 167–181. doi: 10.1080/01635589609514473. PMID 8875554.
- ^ van Meeuwen JA, Korthagen N, de Jong PC, Piersma AH, van den Berg M (June 2007). "(Anti)estrogenic effects of phytochemicals on human primary mammary fibroblasts, MCF-7 cells and their co-culture". Toxicology and Applied Pharmacology. 221 (3): 372–383. doi: 10.1016/j.taap.2007.03.016. PMID 17482226.
- ^ Harmon AW, Patel YM (May 2004). "Naringenin inhibits glucose uptake in MCF-7 breast cancer cells: a mechanism for impaired cellular proliferation". Breast Cancer Research and Treatment. 85 (2): 103–110. doi: 10.1023/B:BREA.0000025397.56192.e2. PMID 15111768. S2CID 24436665.
- ^ Ferreira RJ, Baptista R, Moreno A, Madeira PG, Khonkarn R, Baubichon-Cortay H, et al. (April 2018). "Optimizing the flavanone core toward new selective nitrogen-containing modulators of ABC transporters". Future Medicinal Chemistry. 10 (7): 725–741. doi: 10.4155/fmc-2017-0228. PMID 29570361.
- ^ Fitzpatrick E (September 2018). "Non-alcoholic fatty liver disease". Oxford Medicine Online. doi: 10.1093/med/9780198759928.003.0061.
- ^ Ghofrani S, Joghataei MT, Mohseni S, Baluchnejadmojarad T, Bagheri M, Khamse S, Roghani M (October 2015). "Naringenin improves learning and memory in an Alzheimer's disease rat model: Insights into the underlying mechanisms". European Journal of Pharmacology. 764: 195–201. doi: 10.1016/j.ejphar.2015.07.001. PMID 26148826.
- ^ Yang Z, Kuboyama T, Tohda C (April 2019). "Naringenin promotes microglial M2 polarization and Aβ degradation enzyme expression". Phytotherapy Research. 33 (4): 1114–1121. doi: 10.1002/ptr.6305. PMID 30768735. S2CID 73449033.
- ^ Yang Z, Kuboyama T, Tohda C (19 June 2017). "A Systematic Strategy for Discovering a Therapeutic Drug for Alzheimer's Disease and Its Target Molecule". Frontiers in Pharmacology. 8: 340. doi: 10.3389/fphar.2017.00340. PMC 5474478. PMID 28674493.
Further reading
- inhibitory effect on the human cytochrome P450 isoform CYP1A2 resulting in delayed clearance of substances and protective effect against P4501A2-activated protoxicants.Fuhr U, Klittich K, Staib AH (April 1993). "Inhibitory effect of grapefruit juice and its bitter principal, naringenin, on CYP1A2 dependent metabolism of caffeine in man". British Journal of Clinical Pharmacology. 35 (4): 431–436. doi: 10.1111/j.1365-2125.1993.tb04162.x. PMC 1381556. PMID 8485024.Ueng YF, Chang YL, Oda Y, Park SS, Liao JF, Lin MF, Chen CF (1999). "In vitro and in vivo effects of naringin on cytochrome P450-dependent monooxygenase in mouse liver". Life Sciences. 65 (24): 2591–2602. doi: 10.1016/s0024-3205(99)00528-7. PMID 10619367.
- Wistuba D, Trapp O, Gel-Moreto N, Galensa R, Schurig V (May 2006). "Stereoisomeric separation of flavanones and flavanone-7-O-glycosides by capillary electrophoresis and determination of interconversion barriers". Analytical Chemistry. 78 (10): 3424–3433. doi: 10.1021/ac0600499. PMID 16689546.
- Krause M, Galensa R (1991). "High-performance liquid chromatography of diastereomeric flavanone glycosides in Citrus on a β-cyclodextrin-bonded stationary phase (Cyclobond I)". Journal of Chromatography A. 588 (1–2): 41–45. doi: 10.1016/0021-9673(91)85005-z.
- Gaggeri R, Rossi D, Collina S, Mannucci B, Baierl M, Juza M (August 2011). "Quick development of an analytical enantioselective high performance liquid chromatography separation and preparative scale-up for the flavonoid Naringenin". Journal of Chromatography A. 1218 (32): 5414–5422. doi: 10.1016/j.chroma.2011.02.038. PMID 21397238.
- Wan L, Sun X, Wang X, Li Y, Yu Q, Guo C (April 2011). "A stereospecific HPLC method and its application in determination of pharmacokinetics profile of two enantiomers of naringenin in rats". Journal of Chromatographic Science. 49 (4): 316–320. doi: 10.1093/chrsci/49.4.316. PMID 21439124.
- Naringenin also produces BDNF-dependent antidepressant-like effects in mice.Yi LT, Liu BB, Li J, Luo L, Liu Q, Geng D, et al. (January 2014). "BDNF signaling is necessary for the antidepressant-like effect of naringenin". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 48: 135–141. doi: 10.1016/j.pnpbp.2013.10.002. PMID 24121063. S2CID 24620048.
- Gao K, Henning SM, Niu Y, Youssefian AA, Seeram NP, Xu A, Heber D (February 2006). "The citrus flavonoid naringenin stimulates DNA repair in prostate cancer cells". The Journal of Nutritional Biochemistry. 17 (2): 89–95. doi: 10.1016/j.jnutbio.2005.05.009. PMID 16111881.
- Katavic PL, Lamb K, Navarro H, Prisinzano TE (August 2007). "Flavonoids as opioid receptor ligands: identification and preliminary structure-activity relationships". Journal of Natural Products. 70 (8): 1278–1282. doi: 10.1021/np070194x. PMC 2265593. PMID 17685652.
- Naringenin has been reported to induce apoptosis in preadipocytes.Hsu CL, Huang SL, Yen GC (June 2006). "Inhibitory effect of phenolic acids on the proliferation of 3T3-L1 preadipocytes in relation to their antioxidant activity". Journal of Agricultural and Food Chemistry. 54 (12): 4191–4197. doi: 10.1021/jf0609882. PMID 16756346.
- Naringenin seems to protect LDLR-deficient mice from the obesity effects of a high-fat diet.Mulvihill EE, Allister EM, Sutherland BG, Telford DE, Sawyez CG, Edwards JY, et al. (October 2009). "Naringenin prevents dyslipidemia, apolipoprotein B overproduction, and hyperinsulinemia in LDL receptor-null mice with diet-induced insulin resistance". Diabetes. 58 (10): 2198–2210. doi: 10.2337/db09-0634. PMC 2750228. PMID 19592617.
- Naringenin lowers the plasma and hepatic cholesterol concentrations by suppressing HMG-CoA reductase and ACAT in rats fed a high-cholesterol diet.Lee SH, Park YB, Bae KH, Bok SH, Kwon YK, Lee ES, Choi MS (1999). "Cholesterol-lowering activity of naringenin via inhibition of 3-hydroxy-3-methylglutaryl coenzyme A reductase and acyl coenzyme A:cholesterol acyltransferase in rats". Annals of Nutrition & Metabolism. 43 (3): 173–180. doi: 10.1159/000012783. PMID 10545673. S2CID 5685548.
- Naringenin has been demonstrated to improve memory and reduce amyloid and tau proteins in a study using a mouse model of Alzheimer's disease.Ghofrani S, Joghataei MT, Mohseni S, Baluchnejadmojarad T, Bagheri M, Khamse S, Roghani M (October 2015). "Naringenin improves learning and memory in an Alzheimer's disease rat model: Insights into the underlying mechanisms". European Journal of Pharmacology. 764: 195–201. doi: 10.1016/j.ejphar.2015.07.001. PMID 26148826.Yang Z, Kuboyama T, Tohda C (April 2019). "Naringenin promotes microglial M2 polarization and Aβ degradation enzyme expression". Phytotherapy Research. 33 (4): 1114–1121. doi: 10.1002/ptr.6305. PMID 30768735. S2CID 73449033.Yang Z, Kuboyama T, Tohda C (19 June 2017). "A Systematic Strategy for Discovering a Therapeutic Drug for Alzheimer's Disease and Its Target Molecule". Frontiers in Pharmacology. 8: 340. doi: 10.3389/fphar.2017.00340. PMC 5474478. PMID 28674493.

| |

| |
| Names | |
|---|---|
|
IUPAC name
(2S)-4′,5,7-Trihydroxyflavan-4-one
| |
|
Systematic IUPAC name
(2S)-5,7-Dihydroxy-2-(4-hydroxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Naringetol; Salipurol; Salipurpol
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.006.865 |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H12O5 | |
| Molar mass | 272.256 g·mol−1 |
| Melting point | 251 °C (484 °F; 524 K) [1] |
| 475 mg/L[ citation needed] | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Naringenin is a flavanone from the flavonoid group of polyphenols. It is commonly found in a variety of citrus fruits and is the predominant flavonone in grapefruit. [2] Naringenin has demonstrated numerous biological activities, including anti-inflammatory properties, antioxidant activity and skin healing. [3] [4] [5] [6] It is used as a cosmetic ingredient and dietary supplement. [7] Naringenin (along with furanocoumarins presented in citrus fruits) is thought to be responsible for CYP3A4 suppression in the intestinal wall, that may result in serious changes of pharmacokinetics in drugs related with this enzyme. [8] [9] [10]
Structure
Naringenin has the skeleton structure of a flavanone with three hydroxy groups at the 4′, 5, and 7 carbons. It may be found both in the aglycol form, naringenin, or in its glycosidic form, naringin, which has the addition of the disaccharide neohesperidose attached via a glycosidic linkage at carbon 7.
Like the majority of flavanones, naringenin has a single chiral center at carbon 2, although the optical purity is variable. [11] [12] Racemization of (S)-(−)-naringenin has been shown to occur fairly quickly. [13]
Sources and bioavailability
Naringenin and its glycoside has been found in a variety of herbs and fruits, including grapefruit, [14] bergamot, [15] sour orange, [16] sour cherries, [5] tomatoes, [17] [18] cocoa, [19] Greek oregano, [20] water mint, [21] as well as in beans. [22] Ratios of naringenin to naringin vary among sources, [17] as do enantiomeric ratios. [12]
The naringenin-7-glucoside form seems less bioavailable than the aglycol form. [23]
Grapefruit juice can provide much higher plasma concentrations of naringenin than orange juice. [24] Also found in grapefruit is the related compound kaempferol, which has a hydroxyl group next to the ketone group.
Naringenin can be absorbed from cooked tomato paste. There are 3.8 mg of naringenin in 150 grams of tomato paste. [25]
Biosynthesis and metabolism
Naringenin can be produced from naringin by the hydrolytic action of the liver enzyme naringinase. [26]
Naringenin is derived from malonyl-CoA and 4-coumaroyl-CoA. The latter is derived from phenylalanine. The resulting tetraketide is acted on by chalcone synthase to give the chalcone that then undergoes ring-closure to naringenin. [27]
The enzyme naringenin 8-dimethylallyltransferase uses dimethylallyl diphosphate and (−)-(2S)-naringenin to produce diphosphate and 8-prenylnaringenin. Cunninghamella elegans, a fungal model organism of the mammalian metabolism, can be used to study the naringenin sulfation. [28]
Potential biological effects
This section needs more
reliable medical references for
verification or relies too heavily on
primary sources. (June 2017) |  |
Studies show naringenin has numerous biological activities, including anti-inflammatory, antioxidant, antibacterial, antiviral and anticancer. It is deemed safe for both topical and ingestible use in healthy adults. [7]
Anti-inflammatory
Naringenin’s anti-inflammatory benefits have been well established with multiple in vitro and in vivo studies, revealing it effectively suppresses proinflammatory factors, cytokine and chemokine expressions in inflammation. [3] When taken orally, it has also been shown to reduce inflammatory pain. [29]
Antioxidant
Naringenin has been shown to have significant antioxidant properties. [30] [31] It has been shown to reduce oxidative damage to DNA in vitro and in animal studies. [32] [33] When consumed it has been shown to increase antioxidant markers superoxide dismutase and glutathione. [6]
Endothelium protection
Some human studies and many animal studies have shown the ability of both naringenin and naringin to protect and improve the health of the vascular endothelium. [26] Naringenin reportedly stimulates Nrf2 to protect blood vessels. [34]
Antibacterial, antifungal, and antiviral
Naringenin has an antimicrobial effect on Staphylococcus epidermidis, as well as Staphylococcus aureus, Bacillus subtilis, Micrococcus luteus, and Escherichia coli. [35] Further research has added evidence for antimicrobial effects against Lactococcus lactis, [36] lactobacillus acidophilus, Actinomyces naeslundii, Prevotella oralis, Prevotella melaninogenica, Porphyromonas gingivalis, [37] as well as yeasts such as Candida albicans, Candida tropicalis, and Candida krusei. [38] There is also evidence of antibacterial effects on Helicobacter pylori, though naringenin has not been shown to have any inhibition on urease activity of the microbe. [39]
Naringenin has also been shown to reduce hepatitis C virus production by infected hepatocytes (liver cells) in cell culture. This seems to be secondary to naringenin's ability to inhibit the secretion of very-low-density lipoprotein by the cells. [40] The antiviral effects of naringenin are currently under clinical investigation. [41] Reports of antiviral effects on polioviruses, HSV-1 and HSV-2 have also been made, though replication of the viruses has not been inhibited. [42] [43] In in vitro experiments naringenin also showed a strong antiviral activity against SARS-CoV-2. [44]
Skin Healing
When used in topical formulations, naringenin has been shown to be an anti-inflammatory with skin barrier restoration and antioxidant activities.
UVB radiation is very high energy frequency and wreak havoc on the top layers of the skin by damaging the skin cells and causing DNA mutations that can lead to melanoma and other skin cancers. [45] Naringenin has been proven to reduce UVB-induced skin damage, as well as showing efficacy against oxidative stress and improvement in wound healing. [4] [46] [6]
Naringenin has also been studied on inflammatory skin conditions such as atopic dermatitis and psoriasis. [3] [47] [48] For atopic dermatitis, naringenin was found to sharply suppress inflammatory levels and alleviate symptoms and may suppress the development of atopic dermatitis like skin lesions. [3] [47] In psoriasis, naringenin has been studied to reduce inflammation in psoriatic plaques. [48]
Anticancer
Cytotoxicity has been reduced reportedly by naringenin in cancer cells from breast, stomach, liver, cervix, pancreas, and colon tissues, along with leukaemia cells. [49] [50] The mechanisms behind inhibition of human breast carcinoma growth have been examined, and two theories have been proposed. [51] The first theory is that naringenin inhibits aromatase, thus reducing growth of the tumor. [52] The second mechanism proposes that interactions with estrogen receptors is the cause behind the modulation of growth. [53] New derivatives of naringenin were found to be active against multidrug-resistant cancer. [54]
Fatty Liver Disease
Naringenin may have some benefits for non-alcoholic fatty liver disease. It was proven to reduce hepatic lipid accumulation and inflammation in the livers of mice with non-alcoholic fatty liver disease. [55]
Alzheimer's disease
Naringenin is being researched as a potential treatment for Alzheimer's disease. Naringenin has been demonstrated to improve memory and reduce amyloid and tau proteins in a study using a mouse model of Alzheimer's disease. [56] [57] The effect is believed to be due to a protein present in neurons known as CRMP2 that naringenin binds to. [58]
Safety
Naringenin has been deemed safe to apply topically and can also be ingested safely by healthy adults in doses of 150 to 900 mg, with 300 mg of naringenin twice a day likely to elicit physiological effect. [7]
References
- ^ Naringenin at the Human Metabolome Database
- ^ Felgines C, Texier O, Morand C, Manach C, Scalbert A, Régerat F, Rémésy C (December 2000). "Bioavailability of the flavanone naringenin and its glycosides in rats" (PDF). American Journal of Physiology. Gastrointestinal and Liver Physiology. 279 (6): G1148–G1154. doi: 10.1152/ajpgi.2000.279.6.G1148. PMID 11093936. S2CID 27540043.
- ^ a b c d Karuppagounder V, Arumugam S, Thandavarayan RA, Sreedhar R, Giridharan VV, Pitchaimani V, et al. (May 2016). "Naringenin ameliorates skin inflammation and accelerates phenotypic reprogramming from M1 to M2 macrophage polarization in atopic dermatitis NC/Nga mouse model". Experimental Dermatology. 25 (5): 404–407. doi: 10.1111/exd.12962. PMID 26836240.
- ^ a b Martinez RM, Pinho-Ribeiro FA, Steffen VS, Silva TC, Caviglione CV, Bottura C, et al. (2016-01-07). "Topical Formulation Containing Naringenin: Efficacy against Ultraviolet B Irradiation-Induced Skin Inflammation and Oxidative Stress in Mice". PLOS ONE. 11 (1): e0146296. Bibcode: 2016PLoSO..1146296M. doi: 10.1371/journal.pone.0146296. PMC 4704734. PMID 26741806.
- ^ a b Wang H, Nair MG, Strasburg GM, Booren AM, Gray JI (March 1999). "Antioxidant polyphenols from tart cherries (Prunus cerasus)". Journal of Agricultural and Food Chemistry. 47 (3): 840–844. doi: 10.1021/jf980936f. PMID 10552377.
- ^ a b c Al-Roujayee AS (April 2017). "Naringenin improves the healing process of thermally-induced skin damage in rats". The Journal of International Medical Research. 45 (2): 570–582. doi: 10.1177/0300060517692483. PMC 5536686. PMID 28415935.
- ^ a b c Rebello CJ, Beyl RA, Lertora JJ, Greenway FL, Ravussin E, Ribnicky DM, et al. (January 2020). "Safety and pharmacokinetics of naringenin: A randomized, controlled, single-ascending-dose clinical trial". Diabetes, Obesity & Metabolism. 22 (1): 91–98. doi: 10.1111/dom.13868. PMC 6956701. PMID 31468636.
- ^ Lohezic-Le Devehat, F.; Marigny, K.; Doucet, M.; Javaudin, L. (2002). "[Grapefruit juice and drugs: a hazardous combination?]". Therapie. 57 (5): 432–445. ISSN 0040-5957. PMID 12611197.
- ^ Singh, B. N. (September 1999). "Effects of food on clinical pharmacokinetics". Clinical Pharmacokinetics. 37 (3): 213–255. doi: 10.2165/00003088-199937030-00003. ISSN 0312-5963. PMID 10511919.
- ^ Fuhr, U. (April 1998). "Drug interactions with grapefruit juice. Extent, probable mechanism and clinical relevance". Drug Safety. 18 (4): 251–272. doi: 10.2165/00002018-199818040-00002. ISSN 0114-5916. PMID 9565737.
- ^ Yáñez JA, Andrews PK, Davies NM (April 2007). "Methods of analysis and separation of chiral flavonoids". Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences. 848 (2): 159–181. doi: 10.1016/j.jchromb.2006.10.052. PMID 17113835.
- ^ a b Yáñez JA, Remsberg CM, Miranda ND, Vega-Villa KR, Andrews PK, Davies NM (March 2008). "Pharmacokinetics of selected chiral flavonoids: hesperetin, naringenin and eriodictyol in rats and their content in fruit juices". Biopharmaceutics & Drug Disposition. 29 (2): 63–82. doi: 10.1002/bdd.588. PMID 18058792. S2CID 24051610.
- ^ Krause M, Galensa R (July 1991). "Analysis of enantiomeric flavanones in plant extracts by high-performance liquid chromatography on a cellulose triacetate based chiral stationary phase". Chromatographia. 32 (1–2): 69–72. doi: 10.1007/BF02262470. ISSN 0009-5893. S2CID 95215634.
- ^ Ho PC, Saville DJ, Coville PF, Wanwimolruk S (April 2000). "Content of CYP3A4 inhibitors, naringin, naringenin and bergapten in grapefruit and grapefruit juice products". Pharmaceutica Acta Helvetiae. 74 (4): 379–385. doi: 10.1016/S0031-6865(99)00062-X. PMID 10812937.
- ^ Gattuso G, Barreca D, Gargiulli C, Leuzzi U, Caristi C (August 2007). "Flavonoid composition of Citrus juices". Molecules. 12 (8): 1641–1673. doi: 10.3390/12081641. PMC 6149096. PMID 17960080.
- ^ Gel-Moreto N, Streich R, Galensa R (August 2003). "Chiral separation of diastereomeric flavanone-7-O-glycosides in citrus by capillary electrophoresis". Electrophoresis. 24 (15): 2716–2722. doi: 10.1002/elps.200305486. PMID 12900888. S2CID 40261445.
- ^ a b Minoggio M, Bramati L, Simonetti P, Gardana C, Iemoli L, Santangelo E, et al. (2003-01-01). "Polyphenol pattern and antioxidant activity of different tomato lines and cultivars". Annals of Nutrition & Metabolism. 47 (2): 64–69. doi: 10.1159/000069277. PMID 12652057. S2CID 26333030.
- ^ Vallverdú Queralt A, Odriozola Serrano I, Oms Oliu G, Lamuela Raventós RM, Elez Martínez P, Martín Belloso O (September 2012). "Changes in the polyphenol profile of tomato juices processed by pulsed electric fields". Journal of Agricultural and Food Chemistry. 60 (38): 9667–9672. doi: 10.1021/jf302791k. PMID 22957841.
- ^ Sánchez Rabaneda F, Jáuregui O, Casals I, Andrés Lacueva C, Izquierdo Pulido M, Lamuela Raventós RM (January 2003). "Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao)". Journal of Mass Spectrometry. 38 (1): 35–42. Bibcode: 2003JMSp...38...35S. doi: 10.1002/jms.395. PMID 12526004.
- ^ Exarchou V, Godejohann M, van Beek TA, Gerothanassis IP, Vervoort J (November 2003). "LC-UV-solid-phase extraction-NMR-MS combined with a cryogenic flow probe and its application to the identification of compounds present in Greek oregano". Analytical Chemistry. 75 (22): 6288–6294. doi: 10.1021/ac0347819. PMID 14616013.
- ^ Olsen HT, Stafford GI, van Staden J, Christensen SB, Jäger AK (May 2008). "Isolation of the MAO-inhibitor naringenin from Mentha aquatica L". Journal of Ethnopharmacology. 117 (3): 500–502. doi: 10.1016/j.jep.2008.02.015. PMID 18372132.
- ^ Hungria M, Johnston AW, Phillips DA (1992-05-01). "Effects of flavonoids released naturally from bean (Phaseolus vulgaris) on nodD-regulated gene transcription in Rhizobium leguminosarum bv. phaseoli". Molecular Plant-Microbe Interactions. 5 (3): 199–203. doi: 10.1094/mpmi-5-199. PMID 1421508.
- ^ Choudhury R, Chowrimootoo G, Srai K, Debnam E, Rice-Evans CA (November 1999). "Interactions of the flavonoid naringenin in the gastrointestinal tract and the influence of glycosylation". Biochemical and Biophysical Research Communications. 265 (2): 410–415. doi: 10.1006/bbrc.1999.1695. PMID 10558881.
- ^ Erlund I, Meririnne E, Alfthan G, Aro A (February 2001). "Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice". The Journal of Nutrition. 131 (2): 235–241. doi: 10.1093/jn/131.2.235. PMID 11160539.
- ^ Bugianesi R, Catasta G, Spigno P, D'Uva A, Maiani G (November 2002). "Naringenin from cooked tomato paste is bioavailable in men". The Journal of Nutrition. 132 (11): 3349–3352. doi: 10.1093/jn/132.11.3349. PMID 12421849.
- ^ a b Adetunji JA, Fasae KD, Sekhoacha MP (2023). "The protective roles of citrus flavonoids, naringenin, and naringin on endothelial cell dysfunction in diseases". Heliyon. 9 (6): e17166. Bibcode: 2023Heliy...917166A. doi: 10.1016/j.heliyon.2023.e17166. PMC 10361329. PMID 37484296.
- ^ Wang C, Zhi S, Liu C, Xu F, Zhao A, Wang X, et al. (March 2017). "Characterization of Stilbene Synthase Genes in Mulberry (Morus atropurpurea) and Metabolic Engineering for the Production of Resveratrol in Escherichia coli". Journal of Agricultural and Food Chemistry. 65 (8): 1659–1668. doi: 10.1021/acs.jafc.6b05212. PMID 28168876.
- ^ Ibrahim AR (January 2000). "Sulfation of naringenin by Cunninghamella elegans". Phytochemistry. 53 (2): 209–212. Bibcode: 2000PChem..53..209I. doi: 10.1016/S0031-9422(99)00487-2. PMID 10680173.
- ^ Pinho Ribeiro FA, Zarpelon AC, Fattori V, Manchope MF, Mizokami SS, Casagrande R, Verri WA (June 2016). "Naringenin reduces inflammatory pain in mice". Neuropharmacology. 105: 508–519. doi: 10.1016/j.neuropharm.2016.02.019. PMID 26907804. S2CID 35925963.
- ^ Gorinstein S, Leontowicz H, Leontowicz M, Krzemiński R, Gralak M, Delgado-Licon E, et al. (April 2005). "Changes in plasma lipid and antioxidant activity in rats as a result of naringin and red grapefruit supplementation". Journal of Agricultural and Food Chemistry. 53 (8): 3223–3228. doi: 10.1021/jf058014h. PMID 15826081.
- ^ Yu J, Wang L, Walzem RL, Miller EG, Pike LM, Patil BS (March 2005). "Antioxidant activity of citrus limonoids, flavonoids, and coumarins". Journal of Agricultural and Food Chemistry. 53 (6): 2009–2014. doi: 10.1021/jf0484632. PMID 15769128.
- ^ Kumar S, Tiku AB (March 2016). "Biochemical and Molecular Mechanisms of Radioprotective Effects of Naringenin, a Phytochemical from Citrus Fruits". Journal of Agricultural and Food Chemistry. 64 (8): 1676–1685. doi: 10.1021/acs.jafc.5b05067. PMID 26881453.
- ^ Jagetia GC, Reddy TK, Venkatesha VA, Kedlaya R (September 2004). "Influence of naringin on ferric iron induced oxidative damage in vitro". Clinica Chimica Acta; International Journal of Clinical Chemistry. 347 (1–2): 189–197. doi: 10.1016/j.cccn.2004.04.022. PMID 15313158.
- ^ Zhang Q, Liu J, Wu C (2021). "Activation of Nrf2/HO-1 signaling: An important molecular mechanism of herbal medicine in the treatment of atherosclerosis via the protection of vascular endothelial cells from oxidative stress". Journal of Advanced Research. 34: 43–63. doi: 10.1016/j.jare.2021.06.023. PMC 8655139. PMID 35024180.
- ^ Rauha JP, Remes S, Heinonen M, Hopia A, Kähkönen M, Kujala T, et al. (May 2000). "Antimicrobial effects of Finnish plant extracts containing flavonoids and other phenolic compounds". International Journal of Food Microbiology. 56 (1): 3–12. doi: 10.1016/S0168-1605(00)00218-X. PMID 10857921.
- ^ Mandalari G, Bennett RN, Bisignano G, Trombetta D, Saija A, Faulds CB, et al. (December 2007). "Antimicrobial activity of flavonoids extracted from bergamot (Citrus bergamia Risso) peel, a byproduct of the essential oil industry". Journal of Applied Microbiology. 103 (6): 2056–2064. doi: 10.1111/j.1365-2672.2007.03456.x. PMID 18045389. S2CID 2043029.
- ^ Koru Ö, Toksoy F, Açıkel CH, Tunca YM, Baysallar M, Üsküdar Güçlü A, et al. (2007-06-01). "In vitro antimicrobial activity of propolis samples from different geographical origins against certain oral pathogens". Anaerobe. 13 (3–4): 140–145. doi: 10.1016/j.anaerobe.2007.02.001. PMID 17475517.
- ^ Uzel A, Sorkun K, Önçağ Ö, Coǧulu D, Gençay Ö, Salih B (2005-04-25). "Chemical compositions and antimicrobial activities of four different Anatolian propolis samples". Microbiological Research. 160 (2): 189–195. doi: 10.1016/j.micres.2005.01.002. hdl: 11655/19951. PMID 15881836.
- ^ Bae EA, Han MJ, Kim DH (June 1999). "In vitro anti-Helicobacter pylori activity of some flavonoids and their metabolites". Planta Medica. 65 (5): 442–443. doi: 10.1055/s-2006-960805. PMID 10454900. S2CID 260284591.
- ^ Nahmias Y, Goldwasser J, Casali M, van Poll D, Wakita T, Chung RT, Yarmush ML (May 2008). "Apolipoprotein B-dependent hepatitis C virus secretion is inhibited by the grapefruit flavonoid naringenin". Hepatology. 47 (5): 1437–1445. doi: 10.1002/hep.22197. PMC 4500072. PMID 18393287.
- ^ "A Pilot Study of the Grapefruit Flavonoid Naringenin for HCV Infection". clinicaltrials.gov. Archived from the original on 2010-10-01.
- ^ Mucsi I, Prágai BM (July 1985). "Inhibition of virus multiplication and alteration of cyclic AMP level in cell cultures by flavonoids". Experientia. 41 (7): 930–931. doi: 10.1007/BF01970018. PMID 2989000. S2CID 6174141.
- ^ Lyu SY, Rhim JY, Park WB (November 2005). "Antiherpetic activities of flavonoids against herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) in vitro". Archives of Pharmacal Research. 28 (11): 1293–1301. doi: 10.1007/BF02978215. PMID 16350858. S2CID 34495208.
- ^ Clementi N, Scagnolari C, D'Amore A, Palombi F, Criscuolo E, Frasca F, et al. (January 2021). "Naringenin is a powerful inhibitor of SARS-CoV-2 infection in vitro". Pharmacological Research. 163: 105255. doi: 10.1016/j.phrs.2020.105255. PMC 7574776. PMID 33096221.
- ^ Alexander H. "What's the difference between UVA and UVB rays?". MD Anderson Cancer Center. Retrieved 2024-01-02.
- ^ Salehi M, Ehterami A, Farzamfar S, Vaez A, Ebrahimi-Barough S (February 2021). "Accelerating healing of excisional wound with alginate hydrogel containing naringenin in rat model". Drug Delivery and Translational Research. 11 (1): 142–153. doi: 10.1007/s13346-020-00731-6. PMID 32086788. S2CID 211234647.
- ^ a b Kim TH, Kim GD, Ahn HJ, Cho JJ, Park YS, Park CS (October 2013). "The inhibitory effect of naringenin on atopic dermatitis induced by DNFB in NC/Nga mice". Life Sciences. 93 (15): 516–524. doi: 10.1016/j.lfs.2013.07.027. PMID 23933131.
- ^ a b Alalaiwe A, Lin CF, Hsiao CY, Chen EL, Lin CY, Lien WC, Fang JY (May 2020). "Development of flavanone and its derivatives as topical agents against psoriasis: The prediction of therapeutic efficiency through skin permeation evaluation and cell-based assay". International Journal of Pharmaceutics. 581: 119256. doi: 10.1016/j.ijpharm.2020.119256. PMID 32220586. S2CID 214694347.
- ^ Kanno S, Tomizawa A, Hiura T, Osanai Y, Shouji A, Ujibe M, et al. (March 2005). "Inhibitory effects of naringenin on tumor growth in human cancer cell lines and sarcoma S-180-implanted mice". Biological & Pharmaceutical Bulletin. 28 (3): 527–530. doi: 10.1248/bpb.28.527. PMID 15744083.
- ^ Hermawan A, Ikawati M, Jenie RI, Khumaira A, Putri H, Nurhayati IP, et al. (January 2021). "Identification of potential therapeutic target of naringenin in breast cancer stem cells inhibition by bioinformatics and in vitro studies". Saudi Pharmaceutical Journal. 29 (1): 12–26. doi: 10.1016/j.jsps.2020.12.002. PMC 7873751. PMID 33603536.
- ^ So FV, Guthrie N, Chambers AF, Moussa M, Carroll KK (1996-01-01). "Inhibition of human breast cancer cell proliferation and delay of mammary tumorigenesis by flavonoids and citrus juices". Nutrition and Cancer. 26 (2): 167–181. doi: 10.1080/01635589609514473. PMID 8875554.
- ^ van Meeuwen JA, Korthagen N, de Jong PC, Piersma AH, van den Berg M (June 2007). "(Anti)estrogenic effects of phytochemicals on human primary mammary fibroblasts, MCF-7 cells and their co-culture". Toxicology and Applied Pharmacology. 221 (3): 372–383. doi: 10.1016/j.taap.2007.03.016. PMID 17482226.
- ^ Harmon AW, Patel YM (May 2004). "Naringenin inhibits glucose uptake in MCF-7 breast cancer cells: a mechanism for impaired cellular proliferation". Breast Cancer Research and Treatment. 85 (2): 103–110. doi: 10.1023/B:BREA.0000025397.56192.e2. PMID 15111768. S2CID 24436665.
- ^ Ferreira RJ, Baptista R, Moreno A, Madeira PG, Khonkarn R, Baubichon-Cortay H, et al. (April 2018). "Optimizing the flavanone core toward new selective nitrogen-containing modulators of ABC transporters". Future Medicinal Chemistry. 10 (7): 725–741. doi: 10.4155/fmc-2017-0228. PMID 29570361.
- ^ Fitzpatrick E (September 2018). "Non-alcoholic fatty liver disease". Oxford Medicine Online. doi: 10.1093/med/9780198759928.003.0061.
- ^ Ghofrani S, Joghataei MT, Mohseni S, Baluchnejadmojarad T, Bagheri M, Khamse S, Roghani M (October 2015). "Naringenin improves learning and memory in an Alzheimer's disease rat model: Insights into the underlying mechanisms". European Journal of Pharmacology. 764: 195–201. doi: 10.1016/j.ejphar.2015.07.001. PMID 26148826.
- ^ Yang Z, Kuboyama T, Tohda C (April 2019). "Naringenin promotes microglial M2 polarization and Aβ degradation enzyme expression". Phytotherapy Research. 33 (4): 1114–1121. doi: 10.1002/ptr.6305. PMID 30768735. S2CID 73449033.
- ^ Yang Z, Kuboyama T, Tohda C (19 June 2017). "A Systematic Strategy for Discovering a Therapeutic Drug for Alzheimer's Disease and Its Target Molecule". Frontiers in Pharmacology. 8: 340. doi: 10.3389/fphar.2017.00340. PMC 5474478. PMID 28674493.
Further reading
- inhibitory effect on the human cytochrome P450 isoform CYP1A2 resulting in delayed clearance of substances and protective effect against P4501A2-activated protoxicants.Fuhr U, Klittich K, Staib AH (April 1993). "Inhibitory effect of grapefruit juice and its bitter principal, naringenin, on CYP1A2 dependent metabolism of caffeine in man". British Journal of Clinical Pharmacology. 35 (4): 431–436. doi: 10.1111/j.1365-2125.1993.tb04162.x. PMC 1381556. PMID 8485024.Ueng YF, Chang YL, Oda Y, Park SS, Liao JF, Lin MF, Chen CF (1999). "In vitro and in vivo effects of naringin on cytochrome P450-dependent monooxygenase in mouse liver". Life Sciences. 65 (24): 2591–2602. doi: 10.1016/s0024-3205(99)00528-7. PMID 10619367.
- Wistuba D, Trapp O, Gel-Moreto N, Galensa R, Schurig V (May 2006). "Stereoisomeric separation of flavanones and flavanone-7-O-glycosides by capillary electrophoresis and determination of interconversion barriers". Analytical Chemistry. 78 (10): 3424–3433. doi: 10.1021/ac0600499. PMID 16689546.
- Krause M, Galensa R (1991). "High-performance liquid chromatography of diastereomeric flavanone glycosides in Citrus on a β-cyclodextrin-bonded stationary phase (Cyclobond I)". Journal of Chromatography A. 588 (1–2): 41–45. doi: 10.1016/0021-9673(91)85005-z.
- Gaggeri R, Rossi D, Collina S, Mannucci B, Baierl M, Juza M (August 2011). "Quick development of an analytical enantioselective high performance liquid chromatography separation and preparative scale-up for the flavonoid Naringenin". Journal of Chromatography A. 1218 (32): 5414–5422. doi: 10.1016/j.chroma.2011.02.038. PMID 21397238.
- Wan L, Sun X, Wang X, Li Y, Yu Q, Guo C (April 2011). "A stereospecific HPLC method and its application in determination of pharmacokinetics profile of two enantiomers of naringenin in rats". Journal of Chromatographic Science. 49 (4): 316–320. doi: 10.1093/chrsci/49.4.316. PMID 21439124.
- Naringenin also produces BDNF-dependent antidepressant-like effects in mice.Yi LT, Liu BB, Li J, Luo L, Liu Q, Geng D, et al. (January 2014). "BDNF signaling is necessary for the antidepressant-like effect of naringenin". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 48: 135–141. doi: 10.1016/j.pnpbp.2013.10.002. PMID 24121063. S2CID 24620048.
- Gao K, Henning SM, Niu Y, Youssefian AA, Seeram NP, Xu A, Heber D (February 2006). "The citrus flavonoid naringenin stimulates DNA repair in prostate cancer cells". The Journal of Nutritional Biochemistry. 17 (2): 89–95. doi: 10.1016/j.jnutbio.2005.05.009. PMID 16111881.
- Katavic PL, Lamb K, Navarro H, Prisinzano TE (August 2007). "Flavonoids as opioid receptor ligands: identification and preliminary structure-activity relationships". Journal of Natural Products. 70 (8): 1278–1282. doi: 10.1021/np070194x. PMC 2265593. PMID 17685652.
- Naringenin has been reported to induce apoptosis in preadipocytes.Hsu CL, Huang SL, Yen GC (June 2006). "Inhibitory effect of phenolic acids on the proliferation of 3T3-L1 preadipocytes in relation to their antioxidant activity". Journal of Agricultural and Food Chemistry. 54 (12): 4191–4197. doi: 10.1021/jf0609882. PMID 16756346.
- Naringenin seems to protect LDLR-deficient mice from the obesity effects of a high-fat diet.Mulvihill EE, Allister EM, Sutherland BG, Telford DE, Sawyez CG, Edwards JY, et al. (October 2009). "Naringenin prevents dyslipidemia, apolipoprotein B overproduction, and hyperinsulinemia in LDL receptor-null mice with diet-induced insulin resistance". Diabetes. 58 (10): 2198–2210. doi: 10.2337/db09-0634. PMC 2750228. PMID 19592617.
- Naringenin lowers the plasma and hepatic cholesterol concentrations by suppressing HMG-CoA reductase and ACAT in rats fed a high-cholesterol diet.Lee SH, Park YB, Bae KH, Bok SH, Kwon YK, Lee ES, Choi MS (1999). "Cholesterol-lowering activity of naringenin via inhibition of 3-hydroxy-3-methylglutaryl coenzyme A reductase and acyl coenzyme A:cholesterol acyltransferase in rats". Annals of Nutrition & Metabolism. 43 (3): 173–180. doi: 10.1159/000012783. PMID 10545673. S2CID 5685548.
- Naringenin has been demonstrated to improve memory and reduce amyloid and tau proteins in a study using a mouse model of Alzheimer's disease.Ghofrani S, Joghataei MT, Mohseni S, Baluchnejadmojarad T, Bagheri M, Khamse S, Roghani M (October 2015). "Naringenin improves learning and memory in an Alzheimer's disease rat model: Insights into the underlying mechanisms". European Journal of Pharmacology. 764: 195–201. doi: 10.1016/j.ejphar.2015.07.001. PMID 26148826.Yang Z, Kuboyama T, Tohda C (April 2019). "Naringenin promotes microglial M2 polarization and Aβ degradation enzyme expression". Phytotherapy Research. 33 (4): 1114–1121. doi: 10.1002/ptr.6305. PMID 30768735. S2CID 73449033.Yang Z, Kuboyama T, Tohda C (19 June 2017). "A Systematic Strategy for Discovering a Therapeutic Drug for Alzheimer's Disease and Its Target Molecule". Frontiers in Pharmacology. 8: 340. doi: 10.3389/fphar.2017.00340. PMC 5474478. PMID 28674493.