 | |
| Clinical data | |
|---|---|
| Other names | Ciramadol, WY-15705 |
|
Routes of administration | Oral |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
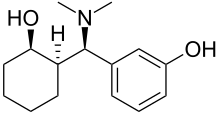
| Formula | C15H23NO2 |
| Molar mass | 249.354 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Ciramadol (WY-15,705) is an opioid analgesic that was developed in the late 1970s [1] and is related to phencyclidine, tramadol, tapentadol and venlafaxine. [2] It is a mixed agonist- antagonist for the μ-opioid receptor with relatively low abuse potential [3] and a ceiling on respiratory depression [4] which makes it a relatively safe drug. It has a slightly higher potency and effectiveness as an analgesic than codeine, [5] but is weaker than morphine. [6] Other side effects include sedation and nausea but these are generally less severe than with other similar drugs. [7]

The Claisen-Schmidt condensation between 3-(methoxymethoxy)benzaldehyde [13709-05-2] (1) and cyclohexanone (2) afforded CID:54364197 (3). Michael addition of dimethylamine leads the aminoketone, i.e. 2-[dimethylamino-[3-(methoxymethoxy)phenyl]methyl]cyclohexan-1-one, CID21518320. Reduction of the ketone proceeds stereospecifically to afford the cis aminoalcohol [51356-58-2] (4). Mild hydrolysis of the product gives the free phenol ciramadol (5).
- ^ US 3928626, "Benzylamine Analgesics"
- ^ Cochrane AD, Bell R, Sullivan JR, Shaw J (November 1979). "Ciramadol. A new analgesic". The Medical Journal of Australia. 2 (9): 501–2. doi: 10.5694/j.1326-5377.1979.tb125803.x. PMID 522808.
- ^ Preston KL, Bigelow GE, Liebson IA (March 1987). "Comparative evaluation of morphine, pentazocine and ciramadol in postaddicts". The Journal of Pharmacology and Experimental Therapeutics. 240 (3): 900–10. PMID 3559982.
- ^ Romagnoli A, Keats AS (1986). "Low ceiling respiratory depression by ciramadol". International Journal of Clinical Pharmacology Research. 6 (6): 451–5. PMID 3100462.
- ^ Downing JW, Brock-Utne JG, Holloway AM (December 1983). "Ciramadol--a new synthetic analgesic. A double-blind comparison with oral codeine for postoperative pain relief". South African Medical Journal = Suid-Afrikaanse Tydskrif vir Geneeskunde. 64 (25): 978–82. PMID 6359486.
- ^ Powell WF (November 1985). "A double-blind comparison of multiple intramuscular doses of ciramadol, morphine, and placebo for the treatment of postoperative pain". Anesthesia and Analgesia. 64 (11): 1101–7. doi: 10.1213/00000539-198511000-00012. PMID 3901821.
- ^ Stambaugh JE, McAdams J (February 1987). "Comparison of the analgesic efficacy and safety oral ciramadol, codeine, and placebo in patients with chronic cancer pain". Journal of Clinical Pharmacology. 27 (2): 162–6. doi: 10.1002/j.1552-4604.1987.tb02178.x. PMID 3680568. S2CID 21229861.
- ^ Yardley, J. P.; Fletcher, H.; Russell, P. B. (1978). "A potent benzylamine analgesic:(−)cis-2(α-dimethylamino-m-hydroxybenzyl)cyclohexanol". Experientia 34(9): 1124–1125. doi:10.1007/BF01922905.
- ^ Paton, D.M.; Castaner, J.; Ciramadol. Drugs Fut 1980, 5, 6, 283.
- ^ Peter B Russell, John P Yardley, U.S. patent 3,928,626, U.S. patent 4,017,637 (1975 and 1977 to American Home Products Corporation).
- ^ John Patrick Yardley, Peter Byrom Russell, DE 2317183 (1986 to American Home Prod).
- ^ John P. Yardley & Peter B. Russell, U.S. patent 4,155,935 (1979 to Wyeth LLC).
 | |
| Clinical data | |
|---|---|
| Other names | Ciramadol, WY-15705 |
|
Routes of administration | Oral |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C15H23NO2 |
| Molar mass | 249.354 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Ciramadol (WY-15,705) is an opioid analgesic that was developed in the late 1970s [1] and is related to phencyclidine, tramadol, tapentadol and venlafaxine. [2] It is a mixed agonist- antagonist for the μ-opioid receptor with relatively low abuse potential [3] and a ceiling on respiratory depression [4] which makes it a relatively safe drug. It has a slightly higher potency and effectiveness as an analgesic than codeine, [5] but is weaker than morphine. [6] Other side effects include sedation and nausea but these are generally less severe than with other similar drugs. [7]

The Claisen-Schmidt condensation between 3-(methoxymethoxy)benzaldehyde [13709-05-2] (1) and cyclohexanone (2) afforded CID:54364197 (3). Michael addition of dimethylamine leads the aminoketone, i.e. 2-[dimethylamino-[3-(methoxymethoxy)phenyl]methyl]cyclohexan-1-one, CID21518320. Reduction of the ketone proceeds stereospecifically to afford the cis aminoalcohol [51356-58-2] (4). Mild hydrolysis of the product gives the free phenol ciramadol (5).
- ^ US 3928626, "Benzylamine Analgesics"
- ^ Cochrane AD, Bell R, Sullivan JR, Shaw J (November 1979). "Ciramadol. A new analgesic". The Medical Journal of Australia. 2 (9): 501–2. doi: 10.5694/j.1326-5377.1979.tb125803.x. PMID 522808.
- ^ Preston KL, Bigelow GE, Liebson IA (March 1987). "Comparative evaluation of morphine, pentazocine and ciramadol in postaddicts". The Journal of Pharmacology and Experimental Therapeutics. 240 (3): 900–10. PMID 3559982.
- ^ Romagnoli A, Keats AS (1986). "Low ceiling respiratory depression by ciramadol". International Journal of Clinical Pharmacology Research. 6 (6): 451–5. PMID 3100462.
- ^ Downing JW, Brock-Utne JG, Holloway AM (December 1983). "Ciramadol--a new synthetic analgesic. A double-blind comparison with oral codeine for postoperative pain relief". South African Medical Journal = Suid-Afrikaanse Tydskrif vir Geneeskunde. 64 (25): 978–82. PMID 6359486.
- ^ Powell WF (November 1985). "A double-blind comparison of multiple intramuscular doses of ciramadol, morphine, and placebo for the treatment of postoperative pain". Anesthesia and Analgesia. 64 (11): 1101–7. doi: 10.1213/00000539-198511000-00012. PMID 3901821.
- ^ Stambaugh JE, McAdams J (February 1987). "Comparison of the analgesic efficacy and safety oral ciramadol, codeine, and placebo in patients with chronic cancer pain". Journal of Clinical Pharmacology. 27 (2): 162–6. doi: 10.1002/j.1552-4604.1987.tb02178.x. PMID 3680568. S2CID 21229861.
- ^ Yardley, J. P.; Fletcher, H.; Russell, P. B. (1978). "A potent benzylamine analgesic:(−)cis-2(α-dimethylamino-m-hydroxybenzyl)cyclohexanol". Experientia 34(9): 1124–1125. doi:10.1007/BF01922905.
- ^ Paton, D.M.; Castaner, J.; Ciramadol. Drugs Fut 1980, 5, 6, 283.
- ^ Peter B Russell, John P Yardley, U.S. patent 3,928,626, U.S. patent 4,017,637 (1975 and 1977 to American Home Products Corporation).
- ^ John Patrick Yardley, Peter Byrom Russell, DE 2317183 (1986 to American Home Prod).
- ^ John P. Yardley & Peter B. Russell, U.S. patent 4,155,935 (1979 to Wyeth LLC).