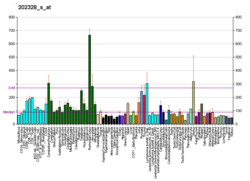
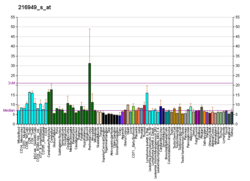
Polycystin 1 (PC1) is a protein that in humans is encoded by the PKD1 gene. [5] [6] Mutations of PKD1 are associated with most cases of autosomal dominant polycystic kidney disease, a severe hereditary disorder of the kidneys characterised by the development of renal cysts and severe kidney dysfunction. [7]

PC1 is a membrane-bound protein 4303 amino acids in length expressed largely upon the primary cilium, as well as apical membranes, adherens junctions, and desmosomes. [8] It has 11 transmembrane domains, a large extracellular N-terminal domain, and a short (about 200 amino acid) cytoplasmic C-terminal domain. [8] [9] This intracellular domain contains a coiled-coil domain through which PC1 interacts with polycystin 2 (PC2), a membrane-bound Ca2+-permeable ion channel.
PC1 has been proposed to act as a G protein–coupled receptor. [8] [10] The C-terminal domain may be cleaved in a number of different ways. In one instance, a ~35 kDa portion of the tail has been found to accumulate in the cell nucleus in response to decreased fluid flow in the mouse kidney. [11] In another instance, a 15 kDa fragment may be yielded, interacting with transcriptional activator and co-activator STAT6 and p100, or components of the canonical Wnt signaling pathway in an inhibitory manner. [12] [13]
The structure of the human PKD1-PKD2 complex has been solved by cryo-electron microscopy, which showed a 1:3 ratio of PKD1 and PKD2 in the structure. PKD1 consists of a voltage-gated ion channel fold that interacts with PKD2. [14]
PC1 mediates mechanosensation of fluid flow by the primary cilium in the renal epithelium and of mechanical deformation of articular cartilage. [15]
Splice variants encoding different isoforms have been noted for PKD1. The gene is closely linked to six pseudogenes in a known duplicated region on chromosome 16p. [16]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000008710 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000032855 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Hughes J, Ward CJ, Peral B, Aspinwall R, Clark K, San Millán JL, Gamble V, Harris PC (June 1995). "The polycystic kidney disease 1 (PKD1) gene encodes a novel protein with multiple cell recognition domains". Nature Genetics. 10 (2): 151–60. doi: 10.1038/ng0695-151. PMID 7663510. S2CID 20636101.
- ^ "Polycystic kidney disease: the complete structure of the PKD1 gene and its protein. The International Polycystic Kidney Disease Consortium". Cell. 81 (2): 289–98. April 1995. doi: 10.1016/0092-8674(95)90339-9. PMID 7736581. S2CID 11114706.
- ^ Torres VE, Harris PC, Pirson Y (April 2007). "Autosomal dominant polycystic kidney disease". Lancet. 369 (9569): 1287–301. doi: 10.1016/S0140-6736(07)60601-1. PMID 17434405. S2CID 1700992.
- ^ a b c Zhou J (2009). "Polycystins and primary cilia: primers for cell cycle progression". Annual Review of Physiology. 71: 83–113. doi: 10.1146/annurev.physiol.70.113006.100621. PMID 19572811.
- ^ Dalagiorgou G, Basdra EK, Papavassiliou AG (October 2010). "Polycystin-1: function as a mechanosensor". The International Journal of Biochemistry & Cell Biology. 42 (10): 1610–3. doi: 10.1016/j.biocel.2010.06.017. PMID 20601082.
- ^ Trudel M, Yao Q, Qian F (January 2016). "The Role of G-Protein-Coupled Receptor Proteolysis Site Cleavage of Polycystin-1 in Renal Physiology and Polycystic Kidney Disease". Cells. 5 (1): 3. doi: 10.3390/cells5010003. PMC 4810088. PMID 26805887.
- ^ Chauvet V, Tian X, Husson H, Grimm DH, Wang T, Hiesberger T, Hieseberger T, Igarashi P, Bennett AM, Ibraghimov-Beskrovnaya O, Somlo S, Caplan MJ (November 2004). "Mechanical stimuli induce cleavage and nuclear translocation of the polycystin-1 C terminus". The Journal of Clinical Investigation. 114 (10): 1433–43. doi: 10.1172/JCI21753. PMC 1052027. PMID 15545994.
- ^ Low SH, Vasanth S, Larson CH, Mukherjee S, Sharma N, Kinter MT, Kane ME, Obara T, Weimbs T (January 2006). "Polycystin-1, STAT6, and P100 function in a pathway that transduces ciliary mechanosensation and is activated in polycystic kidney disease". Developmental Cell. 10 (1): 57–69. doi: 10.1016/j.devcel.2005.12.005. PMID 16399078.
- ^ Lal M, Song X, Pluznick JL, Di Giovanni V, Merrick DM, Rosenblum ND, Chauvet V, Gottardi CJ, Pei Y, Caplan MJ (October 2008). "Polycystin-1 C-terminal tail associates with beta-catenin and inhibits canonical Wnt signaling". Human Molecular Genetics. 17 (20): 3105–17. doi: 10.1093/hmg/ddn208. PMC 2722884. PMID 18632682.
- ^ Shi Y, Mei C, Zhou Q, Wang T, Yu S, Lei J, Ge X, Hu F, Su Q (2018-09-07). "Structure of the human PKD1-PKD2 complex". Science. 361 (6406): eaat9819. doi: 10.1126/science.aat9819. ISSN 0036-8075. PMID 30093605.
- ^ Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, Elia AE, Lu W, Brown EM, Quinn SJ, Ingber DE, Zhou J (February 2003). "Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells". Nature Genetics. 33 (2): 129–37. doi: 10.1038/ng1076. PMID 12514735. S2CID 23149223.
- ^ "Entrez Gene: PKD1 polycystic kidney disease 1 (autosomal dominant)".
| PKD1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | PKD1, PBP, Pc-1, TRPP1, polycystin 1, transient receptor potential channel interacting, PC1 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 601313; MGI: 97603; HomoloGene: 250; GeneCards: PKD1; OMA: PKD1 - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Polycystin 1 (PC1) is a protein that in humans is encoded by the PKD1 gene. [5] [6] Mutations of PKD1 are associated with most cases of autosomal dominant polycystic kidney disease, a severe hereditary disorder of the kidneys characterised by the development of renal cysts and severe kidney dysfunction. [7]

PC1 is a membrane-bound protein 4303 amino acids in length expressed largely upon the primary cilium, as well as apical membranes, adherens junctions, and desmosomes. [8] It has 11 transmembrane domains, a large extracellular N-terminal domain, and a short (about 200 amino acid) cytoplasmic C-terminal domain. [8] [9] This intracellular domain contains a coiled-coil domain through which PC1 interacts with polycystin 2 (PC2), a membrane-bound Ca2+-permeable ion channel.
PC1 has been proposed to act as a G protein–coupled receptor. [8] [10] The C-terminal domain may be cleaved in a number of different ways. In one instance, a ~35 kDa portion of the tail has been found to accumulate in the cell nucleus in response to decreased fluid flow in the mouse kidney. [11] In another instance, a 15 kDa fragment may be yielded, interacting with transcriptional activator and co-activator STAT6 and p100, or components of the canonical Wnt signaling pathway in an inhibitory manner. [12] [13]
The structure of the human PKD1-PKD2 complex has been solved by cryo-electron microscopy, which showed a 1:3 ratio of PKD1 and PKD2 in the structure. PKD1 consists of a voltage-gated ion channel fold that interacts with PKD2. [14]
PC1 mediates mechanosensation of fluid flow by the primary cilium in the renal epithelium and of mechanical deformation of articular cartilage. [15]
Splice variants encoding different isoforms have been noted for PKD1. The gene is closely linked to six pseudogenes in a known duplicated region on chromosome 16p. [16]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000008710 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000032855 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Hughes J, Ward CJ, Peral B, Aspinwall R, Clark K, San Millán JL, Gamble V, Harris PC (June 1995). "The polycystic kidney disease 1 (PKD1) gene encodes a novel protein with multiple cell recognition domains". Nature Genetics. 10 (2): 151–60. doi: 10.1038/ng0695-151. PMID 7663510. S2CID 20636101.
- ^ "Polycystic kidney disease: the complete structure of the PKD1 gene and its protein. The International Polycystic Kidney Disease Consortium". Cell. 81 (2): 289–98. April 1995. doi: 10.1016/0092-8674(95)90339-9. PMID 7736581. S2CID 11114706.
- ^ Torres VE, Harris PC, Pirson Y (April 2007). "Autosomal dominant polycystic kidney disease". Lancet. 369 (9569): 1287–301. doi: 10.1016/S0140-6736(07)60601-1. PMID 17434405. S2CID 1700992.
- ^ a b c Zhou J (2009). "Polycystins and primary cilia: primers for cell cycle progression". Annual Review of Physiology. 71: 83–113. doi: 10.1146/annurev.physiol.70.113006.100621. PMID 19572811.
- ^ Dalagiorgou G, Basdra EK, Papavassiliou AG (October 2010). "Polycystin-1: function as a mechanosensor". The International Journal of Biochemistry & Cell Biology. 42 (10): 1610–3. doi: 10.1016/j.biocel.2010.06.017. PMID 20601082.
- ^ Trudel M, Yao Q, Qian F (January 2016). "The Role of G-Protein-Coupled Receptor Proteolysis Site Cleavage of Polycystin-1 in Renal Physiology and Polycystic Kidney Disease". Cells. 5 (1): 3. doi: 10.3390/cells5010003. PMC 4810088. PMID 26805887.
- ^ Chauvet V, Tian X, Husson H, Grimm DH, Wang T, Hiesberger T, Hieseberger T, Igarashi P, Bennett AM, Ibraghimov-Beskrovnaya O, Somlo S, Caplan MJ (November 2004). "Mechanical stimuli induce cleavage and nuclear translocation of the polycystin-1 C terminus". The Journal of Clinical Investigation. 114 (10): 1433–43. doi: 10.1172/JCI21753. PMC 1052027. PMID 15545994.
- ^ Low SH, Vasanth S, Larson CH, Mukherjee S, Sharma N, Kinter MT, Kane ME, Obara T, Weimbs T (January 2006). "Polycystin-1, STAT6, and P100 function in a pathway that transduces ciliary mechanosensation and is activated in polycystic kidney disease". Developmental Cell. 10 (1): 57–69. doi: 10.1016/j.devcel.2005.12.005. PMID 16399078.
- ^ Lal M, Song X, Pluznick JL, Di Giovanni V, Merrick DM, Rosenblum ND, Chauvet V, Gottardi CJ, Pei Y, Caplan MJ (October 2008). "Polycystin-1 C-terminal tail associates with beta-catenin and inhibits canonical Wnt signaling". Human Molecular Genetics. 17 (20): 3105–17. doi: 10.1093/hmg/ddn208. PMC 2722884. PMID 18632682.
- ^ Shi Y, Mei C, Zhou Q, Wang T, Yu S, Lei J, Ge X, Hu F, Su Q (2018-09-07). "Structure of the human PKD1-PKD2 complex". Science. 361 (6406): eaat9819. doi: 10.1126/science.aat9819. ISSN 0036-8075. PMID 30093605.
- ^ Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, Elia AE, Lu W, Brown EM, Quinn SJ, Ingber DE, Zhou J (February 2003). "Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells". Nature Genetics. 33 (2): 129–37. doi: 10.1038/ng1076. PMID 12514735. S2CID 23149223.
- ^ "Entrez Gene: PKD1 polycystic kidney disease 1 (autosomal dominant)".