| |

| |
| Names | |
|---|---|
|
IUPAC name
L-Tyrosylglycylglycyl-L-phenylalanyl-L-methionyl-L-threonyl-L-seryl-L-glutaminyl-L-lysyl-L-seryl-L-glutaminyl-L-threonyl-L-prolyl-L-leucyl-L-valyl-L-threonyl-L-leucyl-L-phenylalanyl-L-lysyl-L-asparaginyl-L-alanyl-L-isoleucyl-L-isoleucyl-L-lysyl-L-asparaginyl-L-alanyl-L-tyrosyl-L-lysyl-L-lysylglycyl-L-glutamine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.056.646 |
PubChem
CID
|
|
| UNII | |
| |
| |
| Properties | |
| C158H251N39O46S | |
| Molar mass | 3465.03 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Endorphin (beta-endorphin) is an endogenous opioid neuropeptide and peptide hormone that is produced in certain neurons within the central nervous system and peripheral nervous system. [1] It is one of three endorphins that are produced in humans, the others of which include α-endorphin and γ-endorphin. [2]
There are multiple forms of β-endorphins with the full sequence of Tyr- Gly-Gly- Phe- Met- Thr- Ser- Glu- Lys-Ser- Gln-Thr- Pro- Leu- Val-Thr-Leu-Phe-Lys- Asn- Ala- Ile-Ile-Lys-Asn-Ala-Tyr-Lys-Lys-Gly-Glu (31 amino acids) denoted as β-endorphin(1-31) and variants truncated to the first 26 and 27 amino acids as β-endorphin(1-26) and β-endorphin(1-27). [1] [3] [4] The first 16 amino acids are identical to α-endorphin. β-Endorphin is considered to be a part of the endogenous opioid and endorphin classes of neuropeptides; [1] all of the established endogenous opioid peptides contain the same N-terminal amino acid sequence, Tyr-Gly-Gly-Phe, followed by either -Met or -Leu. [1]
Function of β-endorphin has been known to be associated with hunger, thrill, pain, maternal care, sexual behavior, and reward cognition. In the broadest sense, β-endorphin is primarily utilized in the body to reduce stress and maintain homeostasis. In behavioral research, studies have shown that β-endorphin is released via volume transmission into the ventricular system in response to a variety of stimuli, and novel stimuli in particular. [5]
Formation and structure
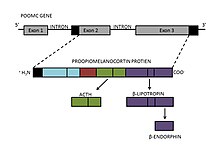
β-Endorphin is found in neurons of the hypothalamus, as well as the pituitary gland. It is derived from β-lipotropin, which is produced in the pituitary gland from a larger peptide precursor, proopiomelanocortin (POMC). [6] POMC is cleaved into two neuropeptides, adrenocorticotropic hormone (ACTH) and β-lipotropin. [7] The formation of β-endorphin is then the result of cleavage of the C-terminal region of β-lipotropin, producing a 31 amino acid-long neuropeptide with an alpha-helical secondary structure. However, POMC also gives rise to other peptide hormones, including α- and γ- melanocyte-stimulating hormone (MSH), resulting from intracellular processing by internal enzymes known as prohormone convertases.
A significant factor that differentiates β-endorphin from other endogenous opioids is its high affinity for and lasting effect on μ-opioid receptors. [6] The structure of β-endorphin in part accounts for this through its resistance to proteolytic enzymes, as its secondary structure makes it less vulnerable to degradation. [6]

Function and effects
β-Endorphin function is said to be divided into two main categories: local function and global function. Global function of β-endorphin is related to decreasing bodily stress and maintaining homeostasis resulting in pain management, reward effects, and behavioral stability. β-Endorphin in global pathways diffuse to different parts of the body through cerebral spinal fluid in the spinal cord, allowing for β-endorphin release to affect the peripheral nervous system. Localized function of β-endorphin results in release of β-endorphin in different brain regions such as the amygdala or the hypothalamus. [5] The two main methods by which β-endorphin is utilized in the body are peripheral hormonal action [8] and neuroregulation. β-endorphin and other enkephalins are often released with ACTH to modulate hormone system functioning. Neuroregulation by β-endorphin occurs through interference with the function of another neuropeptide, either by direct inhibition of neuropeptide release or induction of a signaling cascade that reduces a neuropeptide's effects. [7]
Opioid agonist
β-Endorphin is an agonist of the opioid receptors; it preferentially binds to the μ-opioid receptor. [1] Evidence suggests that it serves as a primary endogenous ligand for the μ-opioid receptor, [1] [9] the same receptor to which the chemicals extracted from opium, such as morphine, derive their analgesic properties. β-Endorphin has the highest binding affinity of any endogenous opioid for the μ-opioid receptor. [1] [6] [9] Opioid receptors are a class of G-protein coupled receptors, such that when β-endorphin or another opioid binds, a signaling cascade is induced in the cell. [10] Acetylation of the N-terminus of β-endorphin, however, inactivates the neuropeptide, preventing it from binding to its receptor. [6] The opioid receptors are distributed throughout the central nervous system and within the peripheral tissue of neural and non-neural origin. They are also located in high concentrations in the Periaqueductal gray, Locus coeruleus, and the Rostral ventromedial medulla. [11]
Voltage-dependent calcium channels (VDCCs) are important membrane proteins that mediate the depolarization of neurons, and play a major role in promoting the release of neurotransmitters. When endorphin molecules bind to opioid receptors, G proteins activate and dissociate into their constituent Gα and Gβγ sub-units. The Gβγ sub-unit binds to the intracellular loop between the two trans-membrane helices of the VDCC. When the sub-unit binds to the voltage-dependent calcium channel, it produces a voltage-dependent block, which inhibits the channel, preventing the flow of calcium ions into the neuron. Embedded in the cell membrane is also the G protein-coupled inwardly-rectifying potassium channel. When a Gβγ or Gα(GTP) molecule binds to the C-terminus of the potassium channel, it becomes active, and potassium ions are pumped out of the neuron. [12] [13] The activation of the potassium channel and subsequent deactivation of the calcium channel causes membrane hyperpolarization. This is when there is a change in the membrane's potential, so that it becomes more negative. The reduction in calcium ions causes a reduction of neurotransmitter release because calcium is essential for this event to occur. [14] This means that neurotransmitters such as glutamate and substance P cannot be released from the presynaptic terminal of the neurons. These neurotransmitters are vital in the transmission of pain, and as β-Endorphin reduces the release of these substances, there is a strong analgesic effect.
Pain management
β-Endorphin has been primarily studied for its influence on nociception (i.e., pain perception). β-endorphin modulates pain perception both in the central nervous system and the peripheral nervous system. When pain is perceived, pain receptors ( nociceptors) send signals to the dorsal horn of the spinal cord and then up to the hypothalamus through the release of a neuropeptide called substance P. [7] [5] [15] [16] In the peripheral nervous system, this signal causes the recruitment of T-lymphocytes, white blood cells of the immune system, to the area where pain was perceived. [16] T-lymphocytes release β-endorphin in this localized region, allowing it to bind to opioid receptors, causing direct inhibition of substance P. [16] [17] In the central nervous system, β-endorphin binds to opioid receptors in the dorsal root and inhibits the release of substance P in the spinal cord, reducing the number of excitatory pain signals sent to the brain. [16] [15] The hypothalamus responds to the pain signal by releasing β-endorphin through the periaqueductal grey network, which mainly acts to inhibit the release of GABA, a neurotransmitter which prevents the release of dopamine. [7] [15] Thus, the inhibition of GABA release by β-endorphin allows for a greater release of dopamine, in part contributing to the analgesic effect of β-endorphin. [7] [15] The combination of these pathways reduces pain sensation, allowing for the body to stop a pain impulse once it has been sent.
β-Endorphin has approximately 18 to 33 times the analgesic potency of morphine, [18] though its hormonal effect is species dependent. [8]
Exercise
β-Endorphin release in response to exercise has been known and studied since at least the 1980s. [19] Studies have demonstrated that serum concentrations of endogenous opioids, in particular β-endorphin and β-lipotropin, increase in response to both acute exercise and training. [19] The release of β-endorphin during exercise is associated with a phenomenon colloquially known in popular culture as a runner's high. [20]
Sunlight
There is evidence that β-endorphin is released in response to ultraviolet radiation, either through sun exposure or artificial tanning. [21] This is thought to contribute to addiction behavior among excessive sunbathers and users of artificial tanning despite health risks.
Mechanism of action
β-Endorphin acts as an agonist that binds to various types of G protein–coupled receptors(GPCRs), most notably to the mu, delta, and kappa opioid receptors. The receptors are responsible for supra-spinal analgesia.[ medical citation needed]
History
β-Endorphin was discovered in camel pituitary extracts by C.H. Li and David Chung. [22] The primary structure of β-endorphin was unknowingly determined 10 years earlier, when Li and colleagues analyzed the sequence of another neuropeptide produced in the pituitary gland, γ-lipotropin. They noticed that the C-terminus region of this neuropeptide was similar to that of some enkephalins, suggesting that it may have a similar function to these neuropeptides. The C-terminal sequence of γ-lipotropin turned out to be the primary sequence of the β-endorphin. [6]
References
- ^
a
b
c
d
e
f
g Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 7: Neuropeptides". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 184, 190, 192.
ISBN
978-0-07-148127-4.
Opioid Peptides
β-Endorphin (also a pituitary hormone) ...
Opioid peptides are encoded by three distinct genes. These precursors include POMC, from which the opioid peptide β-endorphin and several nonopioid peptides are derived, as discussed earlier; proenkephalin, from which met-enkephalin and leu-enkephalin are derived; and prodynorphin, which is the precursor of dynorphin and related peptides. Although they come from different precursors, opioid peptides share significant amino acid sequence identity. Specifically, all of the well-validated endogenous opioids contain the same four N-terminal amino acids (Tyr-Gly-Gly-Phe), followed by either Met or Leu ... Among endogenous opioid peptides, β-endorphin binds preferentially to μ receptors. ... Shared opioid peptide sequences. Although they vary in length from as few as five amino acids (enkephalins) to as many as 31 (β-endorphin), the endogenous opioid peptides shown here contain a shared N-terminal sequence followed by either Met or Leu. - ^ Li Y, Lefever MR, Muthu D, Bidlack JM, Bilsky EJ, Polt R (February 2012). "Opioid glycopeptide analgesics derived from endogenous enkephalins and endorphins". Future Medicinal Chemistry. 4 (2). Table 1: Endogenous opioid peptides. doi: 10.4155/fmc.11.195. PMC 3306179. PMID 22300099.
- ^ Pilozzi A, Carro C, Huang X (December 2020). "Roles of β-Endorphin in Stress, Behavior, Neuroinflammation, and Brain Energy Metabolism". International Journal of Molecular Sciences. 22 (1): 338. doi: 10.3390/ijms22010338. PMC 7796446. PMID 33396962.
- ^ DBGET
- ^ a b c Veening JG, Barendregt HP (January 2015). "The effects of beta-endorphin: state change modification". Fluids and Barriers of the CNS. 12: 3. doi: 10.1186/2045-8118-12-3. PMC 4429837. PMID 25879522.
- ^ a b c d e f Smyth DG (May 2016). "60 YEARS OF POMC: Lipotropin and beta-endorphin: a perspective". Journal of Molecular Endocrinology. 56 (4): T13-25. doi: 10.1530/JME-16-0033. PMID 26903509.
- ^ a b c d e Dalayeun JF, Norès JM, Bergal S (1993). "Physiology of beta-endorphins. A close-up view and a review of the literature". Biomedicine & Pharmacotherapy. 47 (8): 311–20. doi: 10.1016/0753-3322(93)90080-5. PMID 7520295.
- ^ a b Foley KM, Kourides IA, Inturrisi CE, Kaiko RF, Zaroulis CG, Posner JB, Houde RW, Li CH (October 1979). "beta-Endorphin: analgesic and hormonal effects in humans". Proceedings of the National Academy of Sciences of the United States of America. 76 (10): 5377–81. Bibcode: 1979PNAS...76.5377F. doi: 10.1073/pnas.76.10.5377. PMC 413146. PMID 291954.
- ^
a
b Borsodi A, Caló G, Chavkin C, Christie MJ, Civelli O, Cox BM, Devi LA, Evans C, Henderson G, Höllt V, Kieffer B, Kitchen I, Kreek MJ, Liu-Chen LY, Meunier JC, Portoghese PS, Shippenberg TS, Simon EJ, Toll L, Traynor JR, Ueda H, Wong YH (15 March 2017).
"Opioid receptors: μ receptor". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 26 May 2017.
Principal endogenous agonists (Human)
β-endorphin (POMC, P01189), [Met]enkephalin (PENK, P01210), [Leu]enkephalin (PENK, P01210) ...
Comments: β-Endorphin is the highest potency endogenous ligand - ^ Livingston KE, Traynor JR (2018). "Allostery at opioid receptors: modulation with small molecule ligands". British Journal of Pharmacology. 175 (14): 2846–2856. doi: 10.1111/bph.13823. PMC 6016636. PMID 28419415.
- ^ Al-Hasani R, Bruchas MR (December 2011). "Molecular mechanisms of opioid receptor-dependent signaling and behavior". Anesthesiology. 115 (6): 1363–81. doi: 10.1097/ALN.0b013e318238bba6. PMC 3698859. PMID 22020140.
- ^ Yamada M, Inanobe A, Kurachi Y (December 1998). "G protein regulation of potassium ion channels". Pharmacological Reviews. 50 (4): 723–60. PMID 9860808.
- ^ Reuveny E, Slesinger PA, Inglese J, Morales JM, Iñiguez-Liuhi JA, Lefkowitz RJ, Bourne HR, Jan YN, Jan LY (July 1994). "Activation of the Cloned Muscarinic Potassium Channel by G Protein βγ Subunits". Nature. 370 (6485): 143–146. Bibcode: 1994Natur.370..143R. doi: 10.1038/370143a0. PMID 8022483. S2CID 4345632.
- ^ Kosten TR, George TP (July 2002). "The neurobiology of opioid dependence: implications for treatment". Science & Practice Perspectives. 1 (1): 13–20. doi: 10.1151/spp021113. PMC 2851054. PMID 18567959.
- ^ a b c d Sprouse-Blum AS, Smith G, Sugai D, Parsa FD (March 2010). "Understanding endorphins and their importance in pain management". Hawaii Medical Journal. 69 (3): 70–1. PMC 3104618. PMID 20397507.
- ^ a b c d Luan YH, Wang D, Yu Q, Chai XQ (February 2017). "Action of β-endorphin and nonsteroidal anti-inflammatory drugs, and the possible effects of nonsteroidal anti-inflammatory drugs on β-endorphin". Journal of Clinical Anesthesia. 37: 123–128. doi: 10.1016/j.jclinane.2016.12.016. PMID 28235500.
- ^ Plein LM, Rittner HL (2018). "Opioids and the immune system – friend or foe". British Journal of Pharmacology. 175 (14): 2717–2725. doi: 10.1111/bph.13750. PMC 6016673. PMID 28213891.
- ^ Loh HH, Tseng LF, Wei E, Li CH (August 1976). "beta-endorphin is a potent analgesic agent". Proceedings of the National Academy of Sciences of the United States of America. 73 (8): 2895–8. Bibcode: 1976PNAS...73.2895L. doi: 10.1073/pnas.73.8.2895. PMC 430793. PMID 8780.
- ^ a b Harber VJ, Sutton JR (March–April 1984). "Endorphins and exercise". Sports Medicine. 1 (2): 154–71. doi: 10.2165/00007256-198401020-00004. PMID 6091217. S2CID 6435497.
- ^ Goldberg J (19 February 2014). "Exercise and Depression". WebMD. Retrieved 14 July 2014.
- ^ "Addicted to the Sun". hms.harvard.edu. 19 June 2014. Retrieved 29 August 2023.
- ^ Li CH, Chung D (April 1976). "Isolation and structure of an untriakontapeptide with opiate activity from camel pituitary glands". Proceedings of the National Academy of Sciences of the United States of America. 73 (4): 1145–8. Bibcode: 1976PNAS...73.1145L. doi: 10.1073/pnas.73.4.1145. PMC 430217. PMID 1063395.
External links
- CID 16132316 from PubChem – β-endorphin
- CID 3081525 from PubChem – β-endorphin (1-9)
- CID 133304 from PubChem – β-endorphin (2-9)
- β-endorphin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)

| |

| |
| Names | |
|---|---|
|
IUPAC name
L-Tyrosylglycylglycyl-L-phenylalanyl-L-methionyl-L-threonyl-L-seryl-L-glutaminyl-L-lysyl-L-seryl-L-glutaminyl-L-threonyl-L-prolyl-L-leucyl-L-valyl-L-threonyl-L-leucyl-L-phenylalanyl-L-lysyl-L-asparaginyl-L-alanyl-L-isoleucyl-L-isoleucyl-L-lysyl-L-asparaginyl-L-alanyl-L-tyrosyl-L-lysyl-L-lysylglycyl-L-glutamine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.056.646 |
PubChem
CID
|
|
| UNII | |
| |
| |
| Properties | |
| C158H251N39O46S | |
| Molar mass | 3465.03 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Endorphin (beta-endorphin) is an endogenous opioid neuropeptide and peptide hormone that is produced in certain neurons within the central nervous system and peripheral nervous system. [1] It is one of three endorphins that are produced in humans, the others of which include α-endorphin and γ-endorphin. [2]
There are multiple forms of β-endorphins with the full sequence of Tyr- Gly-Gly- Phe- Met- Thr- Ser- Glu- Lys-Ser- Gln-Thr- Pro- Leu- Val-Thr-Leu-Phe-Lys- Asn- Ala- Ile-Ile-Lys-Asn-Ala-Tyr-Lys-Lys-Gly-Glu (31 amino acids) denoted as β-endorphin(1-31) and variants truncated to the first 26 and 27 amino acids as β-endorphin(1-26) and β-endorphin(1-27). [1] [3] [4] The first 16 amino acids are identical to α-endorphin. β-Endorphin is considered to be a part of the endogenous opioid and endorphin classes of neuropeptides; [1] all of the established endogenous opioid peptides contain the same N-terminal amino acid sequence, Tyr-Gly-Gly-Phe, followed by either -Met or -Leu. [1]
Function of β-endorphin has been known to be associated with hunger, thrill, pain, maternal care, sexual behavior, and reward cognition. In the broadest sense, β-endorphin is primarily utilized in the body to reduce stress and maintain homeostasis. In behavioral research, studies have shown that β-endorphin is released via volume transmission into the ventricular system in response to a variety of stimuli, and novel stimuli in particular. [5]
Formation and structure
β-Endorphin is found in neurons of the hypothalamus, as well as the pituitary gland. It is derived from β-lipotropin, which is produced in the pituitary gland from a larger peptide precursor, proopiomelanocortin (POMC). [6] POMC is cleaved into two neuropeptides, adrenocorticotropic hormone (ACTH) and β-lipotropin. [7] The formation of β-endorphin is then the result of cleavage of the C-terminal region of β-lipotropin, producing a 31 amino acid-long neuropeptide with an alpha-helical secondary structure. However, POMC also gives rise to other peptide hormones, including α- and γ- melanocyte-stimulating hormone (MSH), resulting from intracellular processing by internal enzymes known as prohormone convertases.
A significant factor that differentiates β-endorphin from other endogenous opioids is its high affinity for and lasting effect on μ-opioid receptors. [6] The structure of β-endorphin in part accounts for this through its resistance to proteolytic enzymes, as its secondary structure makes it less vulnerable to degradation. [6]


Function and effects
β-Endorphin function is said to be divided into two main categories: local function and global function. Global function of β-endorphin is related to decreasing bodily stress and maintaining homeostasis resulting in pain management, reward effects, and behavioral stability. β-Endorphin in global pathways diffuse to different parts of the body through cerebral spinal fluid in the spinal cord, allowing for β-endorphin release to affect the peripheral nervous system. Localized function of β-endorphin results in release of β-endorphin in different brain regions such as the amygdala or the hypothalamus. [5] The two main methods by which β-endorphin is utilized in the body are peripheral hormonal action [8] and neuroregulation. β-endorphin and other enkephalins are often released with ACTH to modulate hormone system functioning. Neuroregulation by β-endorphin occurs through interference with the function of another neuropeptide, either by direct inhibition of neuropeptide release or induction of a signaling cascade that reduces a neuropeptide's effects. [7]
Opioid agonist
β-Endorphin is an agonist of the opioid receptors; it preferentially binds to the μ-opioid receptor. [1] Evidence suggests that it serves as a primary endogenous ligand for the μ-opioid receptor, [1] [9] the same receptor to which the chemicals extracted from opium, such as morphine, derive their analgesic properties. β-Endorphin has the highest binding affinity of any endogenous opioid for the μ-opioid receptor. [1] [6] [9] Opioid receptors are a class of G-protein coupled receptors, such that when β-endorphin or another opioid binds, a signaling cascade is induced in the cell. [10] Acetylation of the N-terminus of β-endorphin, however, inactivates the neuropeptide, preventing it from binding to its receptor. [6] The opioid receptors are distributed throughout the central nervous system and within the peripheral tissue of neural and non-neural origin. They are also located in high concentrations in the Periaqueductal gray, Locus coeruleus, and the Rostral ventromedial medulla. [11]
Voltage-dependent calcium channels (VDCCs) are important membrane proteins that mediate the depolarization of neurons, and play a major role in promoting the release of neurotransmitters. When endorphin molecules bind to opioid receptors, G proteins activate and dissociate into their constituent Gα and Gβγ sub-units. The Gβγ sub-unit binds to the intracellular loop between the two trans-membrane helices of the VDCC. When the sub-unit binds to the voltage-dependent calcium channel, it produces a voltage-dependent block, which inhibits the channel, preventing the flow of calcium ions into the neuron. Embedded in the cell membrane is also the G protein-coupled inwardly-rectifying potassium channel. When a Gβγ or Gα(GTP) molecule binds to the C-terminus of the potassium channel, it becomes active, and potassium ions are pumped out of the neuron. [12] [13] The activation of the potassium channel and subsequent deactivation of the calcium channel causes membrane hyperpolarization. This is when there is a change in the membrane's potential, so that it becomes more negative. The reduction in calcium ions causes a reduction of neurotransmitter release because calcium is essential for this event to occur. [14] This means that neurotransmitters such as glutamate and substance P cannot be released from the presynaptic terminal of the neurons. These neurotransmitters are vital in the transmission of pain, and as β-Endorphin reduces the release of these substances, there is a strong analgesic effect.
Pain management
β-Endorphin has been primarily studied for its influence on nociception (i.e., pain perception). β-endorphin modulates pain perception both in the central nervous system and the peripheral nervous system. When pain is perceived, pain receptors ( nociceptors) send signals to the dorsal horn of the spinal cord and then up to the hypothalamus through the release of a neuropeptide called substance P. [7] [5] [15] [16] In the peripheral nervous system, this signal causes the recruitment of T-lymphocytes, white blood cells of the immune system, to the area where pain was perceived. [16] T-lymphocytes release β-endorphin in this localized region, allowing it to bind to opioid receptors, causing direct inhibition of substance P. [16] [17] In the central nervous system, β-endorphin binds to opioid receptors in the dorsal root and inhibits the release of substance P in the spinal cord, reducing the number of excitatory pain signals sent to the brain. [16] [15] The hypothalamus responds to the pain signal by releasing β-endorphin through the periaqueductal grey network, which mainly acts to inhibit the release of GABA, a neurotransmitter which prevents the release of dopamine. [7] [15] Thus, the inhibition of GABA release by β-endorphin allows for a greater release of dopamine, in part contributing to the analgesic effect of β-endorphin. [7] [15] The combination of these pathways reduces pain sensation, allowing for the body to stop a pain impulse once it has been sent.
β-Endorphin has approximately 18 to 33 times the analgesic potency of morphine, [18] though its hormonal effect is species dependent. [8]
Exercise
β-Endorphin release in response to exercise has been known and studied since at least the 1980s. [19] Studies have demonstrated that serum concentrations of endogenous opioids, in particular β-endorphin and β-lipotropin, increase in response to both acute exercise and training. [19] The release of β-endorphin during exercise is associated with a phenomenon colloquially known in popular culture as a runner's high. [20]
Sunlight
There is evidence that β-endorphin is released in response to ultraviolet radiation, either through sun exposure or artificial tanning. [21] This is thought to contribute to addiction behavior among excessive sunbathers and users of artificial tanning despite health risks.
Mechanism of action
β-Endorphin acts as an agonist that binds to various types of G protein–coupled receptors(GPCRs), most notably to the mu, delta, and kappa opioid receptors. The receptors are responsible for supra-spinal analgesia.[ medical citation needed]
History
β-Endorphin was discovered in camel pituitary extracts by C.H. Li and David Chung. [22] The primary structure of β-endorphin was unknowingly determined 10 years earlier, when Li and colleagues analyzed the sequence of another neuropeptide produced in the pituitary gland, γ-lipotropin. They noticed that the C-terminus region of this neuropeptide was similar to that of some enkephalins, suggesting that it may have a similar function to these neuropeptides. The C-terminal sequence of γ-lipotropin turned out to be the primary sequence of the β-endorphin. [6]
References
- ^
a
b
c
d
e
f
g Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 7: Neuropeptides". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 184, 190, 192.
ISBN
978-0-07-148127-4.
Opioid Peptides
β-Endorphin (also a pituitary hormone) ...
Opioid peptides are encoded by three distinct genes. These precursors include POMC, from which the opioid peptide β-endorphin and several nonopioid peptides are derived, as discussed earlier; proenkephalin, from which met-enkephalin and leu-enkephalin are derived; and prodynorphin, which is the precursor of dynorphin and related peptides. Although they come from different precursors, opioid peptides share significant amino acid sequence identity. Specifically, all of the well-validated endogenous opioids contain the same four N-terminal amino acids (Tyr-Gly-Gly-Phe), followed by either Met or Leu ... Among endogenous opioid peptides, β-endorphin binds preferentially to μ receptors. ... Shared opioid peptide sequences. Although they vary in length from as few as five amino acids (enkephalins) to as many as 31 (β-endorphin), the endogenous opioid peptides shown here contain a shared N-terminal sequence followed by either Met or Leu. - ^ Li Y, Lefever MR, Muthu D, Bidlack JM, Bilsky EJ, Polt R (February 2012). "Opioid glycopeptide analgesics derived from endogenous enkephalins and endorphins". Future Medicinal Chemistry. 4 (2). Table 1: Endogenous opioid peptides. doi: 10.4155/fmc.11.195. PMC 3306179. PMID 22300099.
- ^ Pilozzi A, Carro C, Huang X (December 2020). "Roles of β-Endorphin in Stress, Behavior, Neuroinflammation, and Brain Energy Metabolism". International Journal of Molecular Sciences. 22 (1): 338. doi: 10.3390/ijms22010338. PMC 7796446. PMID 33396962.
- ^ DBGET
- ^ a b c Veening JG, Barendregt HP (January 2015). "The effects of beta-endorphin: state change modification". Fluids and Barriers of the CNS. 12: 3. doi: 10.1186/2045-8118-12-3. PMC 4429837. PMID 25879522.
- ^ a b c d e f Smyth DG (May 2016). "60 YEARS OF POMC: Lipotropin and beta-endorphin: a perspective". Journal of Molecular Endocrinology. 56 (4): T13-25. doi: 10.1530/JME-16-0033. PMID 26903509.
- ^ a b c d e Dalayeun JF, Norès JM, Bergal S (1993). "Physiology of beta-endorphins. A close-up view and a review of the literature". Biomedicine & Pharmacotherapy. 47 (8): 311–20. doi: 10.1016/0753-3322(93)90080-5. PMID 7520295.
- ^ a b Foley KM, Kourides IA, Inturrisi CE, Kaiko RF, Zaroulis CG, Posner JB, Houde RW, Li CH (October 1979). "beta-Endorphin: analgesic and hormonal effects in humans". Proceedings of the National Academy of Sciences of the United States of America. 76 (10): 5377–81. Bibcode: 1979PNAS...76.5377F. doi: 10.1073/pnas.76.10.5377. PMC 413146. PMID 291954.
- ^
a
b Borsodi A, Caló G, Chavkin C, Christie MJ, Civelli O, Cox BM, Devi LA, Evans C, Henderson G, Höllt V, Kieffer B, Kitchen I, Kreek MJ, Liu-Chen LY, Meunier JC, Portoghese PS, Shippenberg TS, Simon EJ, Toll L, Traynor JR, Ueda H, Wong YH (15 March 2017).
"Opioid receptors: μ receptor". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 26 May 2017.
Principal endogenous agonists (Human)
β-endorphin (POMC, P01189), [Met]enkephalin (PENK, P01210), [Leu]enkephalin (PENK, P01210) ...
Comments: β-Endorphin is the highest potency endogenous ligand - ^ Livingston KE, Traynor JR (2018). "Allostery at opioid receptors: modulation with small molecule ligands". British Journal of Pharmacology. 175 (14): 2846–2856. doi: 10.1111/bph.13823. PMC 6016636. PMID 28419415.
- ^ Al-Hasani R, Bruchas MR (December 2011). "Molecular mechanisms of opioid receptor-dependent signaling and behavior". Anesthesiology. 115 (6): 1363–81. doi: 10.1097/ALN.0b013e318238bba6. PMC 3698859. PMID 22020140.
- ^ Yamada M, Inanobe A, Kurachi Y (December 1998). "G protein regulation of potassium ion channels". Pharmacological Reviews. 50 (4): 723–60. PMID 9860808.
- ^ Reuveny E, Slesinger PA, Inglese J, Morales JM, Iñiguez-Liuhi JA, Lefkowitz RJ, Bourne HR, Jan YN, Jan LY (July 1994). "Activation of the Cloned Muscarinic Potassium Channel by G Protein βγ Subunits". Nature. 370 (6485): 143–146. Bibcode: 1994Natur.370..143R. doi: 10.1038/370143a0. PMID 8022483. S2CID 4345632.
- ^ Kosten TR, George TP (July 2002). "The neurobiology of opioid dependence: implications for treatment". Science & Practice Perspectives. 1 (1): 13–20. doi: 10.1151/spp021113. PMC 2851054. PMID 18567959.
- ^ a b c d Sprouse-Blum AS, Smith G, Sugai D, Parsa FD (March 2010). "Understanding endorphins and their importance in pain management". Hawaii Medical Journal. 69 (3): 70–1. PMC 3104618. PMID 20397507.
- ^ a b c d Luan YH, Wang D, Yu Q, Chai XQ (February 2017). "Action of β-endorphin and nonsteroidal anti-inflammatory drugs, and the possible effects of nonsteroidal anti-inflammatory drugs on β-endorphin". Journal of Clinical Anesthesia. 37: 123–128. doi: 10.1016/j.jclinane.2016.12.016. PMID 28235500.
- ^ Plein LM, Rittner HL (2018). "Opioids and the immune system – friend or foe". British Journal of Pharmacology. 175 (14): 2717–2725. doi: 10.1111/bph.13750. PMC 6016673. PMID 28213891.
- ^ Loh HH, Tseng LF, Wei E, Li CH (August 1976). "beta-endorphin is a potent analgesic agent". Proceedings of the National Academy of Sciences of the United States of America. 73 (8): 2895–8. Bibcode: 1976PNAS...73.2895L. doi: 10.1073/pnas.73.8.2895. PMC 430793. PMID 8780.
- ^ a b Harber VJ, Sutton JR (March–April 1984). "Endorphins and exercise". Sports Medicine. 1 (2): 154–71. doi: 10.2165/00007256-198401020-00004. PMID 6091217. S2CID 6435497.
- ^ Goldberg J (19 February 2014). "Exercise and Depression". WebMD. Retrieved 14 July 2014.
- ^ "Addicted to the Sun". hms.harvard.edu. 19 June 2014. Retrieved 29 August 2023.
- ^ Li CH, Chung D (April 1976). "Isolation and structure of an untriakontapeptide with opiate activity from camel pituitary glands". Proceedings of the National Academy of Sciences of the United States of America. 73 (4): 1145–8. Bibcode: 1976PNAS...73.1145L. doi: 10.1073/pnas.73.4.1145. PMC 430217. PMID 1063395.
External links
- CID 16132316 from PubChem – β-endorphin
- CID 3081525 from PubChem – β-endorphin (1-9)
- CID 133304 from PubChem – β-endorphin (2-9)
- β-endorphin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)