| |
| Names | |
|---|---|
|
Preferred IUPAC name
(2E)-N-[2-(5-Hydroxy-1H-indol-3-yl)ethyl]-3-(4-hydroxy-3-methoxyphenyl)prop-2-enamide | |
| Other names
Moschamine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C20H20N2O4 | |
| Molar mass | 352.390 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
N-Feruloylserotonin an alkaloid and polyphenol found in safflower seed. Chemically, it is an amide formed between serotonin and ferulic acid. It has in vitro anti-atherogenic activity. [1]
Serotonin derivatives found in safflower seeds
N-Feruloylserotonin and N-(p-coumaroyl)serotonin are natural products that can be found in the extract of safflower seeds (Carthamus tinctorius). [2] These natural products have been isolated and studied to investigate their antioxidant effects. [3] These polyphenols have been utilized in traditional Chinese medicine and other eastern medicine practices to have strong antioxidant effects, chemotherapeutic effects, and atherosclerosis attenuation. [1] [4] It has been found that N-(p-coumaroyl) and N- feruloyl serotonin can suppress the expression of matrix metalloproteinases MMP3/13 and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS), thus attenuating cartilage degradation. [2]
Biosynthesis
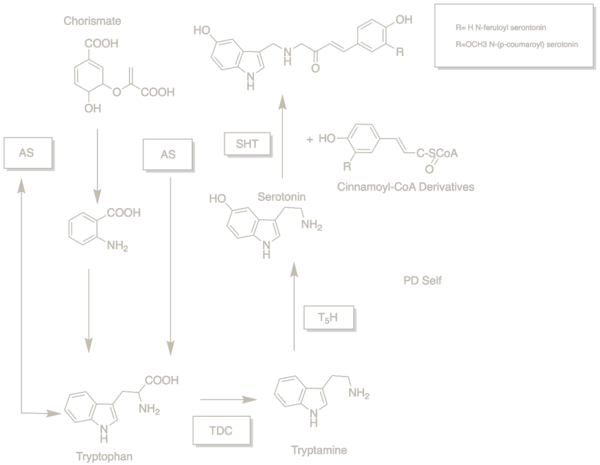
The biosynthetic pathway of N-feruloylserotonin and N-(p-coumaroyl)serotonin has been reported. [5] In plants, the enzyme anthranilate synthase (AS) is composed of two subunits that modulate the production or suppression of tryptophan from chorismate. [6] Tryptophan is then decarboxylated by tryptophan decarboxylase (TDC) into tryptamine. [5] Tryptamine 5-hydroxylase (T5H) then hydroxylates tryptamine into serotonin. [7] [8] Serotonin, the precursor to N-(p-coumaroyl) and N-feruloylserotonin, is found in the seeds of the safflower plant. [9] [10] [11] Hydroxycinnamic acids are then transferred to serotonin from hydroxycinnamoyl-CoA esters by hydroxycinnamoyl-CoA: serotonin N-(hydroxycinnamoyl)transferase (SHT). [6]
References
- ^ a b Takimoto T, Suzuki K, Arisaka H, Murata T, Ozaki H, Koyama N (October 2011). "Effect of N-(p-coumaroyl)serotonin and N-feruloylserotonin, major anti-atherogenic polyphenols in safflower seed, on vasodilation, proliferation and migration of vascular smooth muscle cells". Molecular Nutrition & Food Research. 55 (10): 1561–1571. doi: 10.1002/mnfr.201000545. PMID 21648068.
- ^ a b Han SJ, Lim MJ, Lee KM, Oh E, Shin YS, Kim S, et al. (March 2021). "Safflower Seed Extract Attenuates the Development of Osteoarthritis by Blocking NF-κB Signaling". Pharmaceuticals. 14 (3): 258. doi: 10.3390/ph14030258. PMC 7999399. PMID 33809253.
- ^ Kanehira T, Takekoshi S, Nagata H, Matsuzaki K, Kambayashi Y, Osamura RY, Homma T (November 2003). "A novel and potent biological antioxidant, Kinobeon A, from cell culture of safflower". Life Sciences. 74 (1): 87–97. doi: 10.1016/j.lfs.2003.06.033. PMID 14575815.
- ^ Kang S, Kang K, Lee K, Back K (November 2007). "Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants". Plant Cell Reports. 26 (11): 2009–2015. doi: 10.1007/s00299-007-0405-9. PMID 17639402.
- ^ a b Kang K, Kang S, Lee K, Park M, Back K (June 2008). "Enzymatic features of serotonin biosynthetic enzymes and serotonin biosynthesis in plants". Plant Signaling & Behavior. 3 (6): 389–390. doi: 10.4161/psb.3.6.5401. PMC 2634310. PMID 19704574.
- ^ a b Radwanski ER, Last RL (July 1995). "Tryptophan biosynthesis and metabolism: biochemical and molecular genetics". The Plant Cell. 7 (7): 921–934. doi: 10.1105/tpc.7.7.921. PMC 160888. PMID 7640526.
- ^ Facchini PJ, Huber-Allanach KL, Tari LW (May 2000). "Plant aromatic L-amino acid decarboxylases: evolution, biochemistry, regulation, and metabolic engineering applications". Phytochemistry. 54 (2): 121–138. doi: 10.1016/s0031-9422(00)00050-9. PMID 10872203.
- ^ Kang S, Kang K, Lee K, Back K (December 2007). "Characterization of rice tryptophan decarboxylases and their direct involvement in serotonin biosynthesis in transgenic rice". Planta. 227 (1): 263–272. doi: 10.1007/s00425-007-0614-z. PMID 17763868.
- ^ Bowden K, Brown BG, Batty JE (November 1954). "5-Hydroxytryptamine: its occurrence in cowhage". Nature. 174 (4437): 925–926. doi: 10.1038/174925a0. PMID 13214042.
- ^ Kushiro T, Nambara E, McCourt P (March 2003). "Hormone evolution: The key to signalling". Nature. 422 (6928): 122. doi: 10.1093/aob/mcg113. PMC 4243638.
- ^ Vigliante I, Mannino G, Maffei ME (March 2019). "Chemical Characterization and DNA Fingerprinting of Griffonia simplicifolia Baill". Molecules. 24 (6): E1032. doi: 10.3390/molecules24061032. PMC 6472197. PMID 30875930.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(2E)-N-[2-(5-Hydroxy-1H-indol-3-yl)ethyl]-3-(4-hydroxy-3-methoxyphenyl)prop-2-enamide | |
| Other names
Moschamine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C20H20N2O4 | |
| Molar mass | 352.390 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
N-Feruloylserotonin an alkaloid and polyphenol found in safflower seed. Chemically, it is an amide formed between serotonin and ferulic acid. It has in vitro anti-atherogenic activity. [1]
Serotonin derivatives found in safflower seeds
N-Feruloylserotonin and N-(p-coumaroyl)serotonin are natural products that can be found in the extract of safflower seeds (Carthamus tinctorius). [2] These natural products have been isolated and studied to investigate their antioxidant effects. [3] These polyphenols have been utilized in traditional Chinese medicine and other eastern medicine practices to have strong antioxidant effects, chemotherapeutic effects, and atherosclerosis attenuation. [1] [4] It has been found that N-(p-coumaroyl) and N- feruloyl serotonin can suppress the expression of matrix metalloproteinases MMP3/13 and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS), thus attenuating cartilage degradation. [2]
Biosynthesis
The biosynthetic pathway of N-feruloylserotonin and N-(p-coumaroyl)serotonin has been reported. [5] In plants, the enzyme anthranilate synthase (AS) is composed of two subunits that modulate the production or suppression of tryptophan from chorismate. [6] Tryptophan is then decarboxylated by tryptophan decarboxylase (TDC) into tryptamine. [5] Tryptamine 5-hydroxylase (T5H) then hydroxylates tryptamine into serotonin. [7] [8] Serotonin, the precursor to N-(p-coumaroyl) and N-feruloylserotonin, is found in the seeds of the safflower plant. [9] [10] [11] Hydroxycinnamic acids are then transferred to serotonin from hydroxycinnamoyl-CoA esters by hydroxycinnamoyl-CoA: serotonin N-(hydroxycinnamoyl)transferase (SHT). [6]
References
- ^ a b Takimoto T, Suzuki K, Arisaka H, Murata T, Ozaki H, Koyama N (October 2011). "Effect of N-(p-coumaroyl)serotonin and N-feruloylserotonin, major anti-atherogenic polyphenols in safflower seed, on vasodilation, proliferation and migration of vascular smooth muscle cells". Molecular Nutrition & Food Research. 55 (10): 1561–1571. doi: 10.1002/mnfr.201000545. PMID 21648068.
- ^ a b Han SJ, Lim MJ, Lee KM, Oh E, Shin YS, Kim S, et al. (March 2021). "Safflower Seed Extract Attenuates the Development of Osteoarthritis by Blocking NF-κB Signaling". Pharmaceuticals. 14 (3): 258. doi: 10.3390/ph14030258. PMC 7999399. PMID 33809253.
- ^ Kanehira T, Takekoshi S, Nagata H, Matsuzaki K, Kambayashi Y, Osamura RY, Homma T (November 2003). "A novel and potent biological antioxidant, Kinobeon A, from cell culture of safflower". Life Sciences. 74 (1): 87–97. doi: 10.1016/j.lfs.2003.06.033. PMID 14575815.
- ^ Kang S, Kang K, Lee K, Back K (November 2007). "Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants". Plant Cell Reports. 26 (11): 2009–2015. doi: 10.1007/s00299-007-0405-9. PMID 17639402.
- ^ a b Kang K, Kang S, Lee K, Park M, Back K (June 2008). "Enzymatic features of serotonin biosynthetic enzymes and serotonin biosynthesis in plants". Plant Signaling & Behavior. 3 (6): 389–390. doi: 10.4161/psb.3.6.5401. PMC 2634310. PMID 19704574.
- ^ a b Radwanski ER, Last RL (July 1995). "Tryptophan biosynthesis and metabolism: biochemical and molecular genetics". The Plant Cell. 7 (7): 921–934. doi: 10.1105/tpc.7.7.921. PMC 160888. PMID 7640526.
- ^ Facchini PJ, Huber-Allanach KL, Tari LW (May 2000). "Plant aromatic L-amino acid decarboxylases: evolution, biochemistry, regulation, and metabolic engineering applications". Phytochemistry. 54 (2): 121–138. doi: 10.1016/s0031-9422(00)00050-9. PMID 10872203.
- ^ Kang S, Kang K, Lee K, Back K (December 2007). "Characterization of rice tryptophan decarboxylases and their direct involvement in serotonin biosynthesis in transgenic rice". Planta. 227 (1): 263–272. doi: 10.1007/s00425-007-0614-z. PMID 17763868.
- ^ Bowden K, Brown BG, Batty JE (November 1954). "5-Hydroxytryptamine: its occurrence in cowhage". Nature. 174 (4437): 925–926. doi: 10.1038/174925a0. PMID 13214042.
- ^ Kushiro T, Nambara E, McCourt P (March 2003). "Hormone evolution: The key to signalling". Nature. 422 (6928): 122. doi: 10.1093/aob/mcg113. PMC 4243638.
- ^ Vigliante I, Mannino G, Maffei ME (March 2019). "Chemical Characterization and DNA Fingerprinting of Griffonia simplicifolia Baill". Molecules. 24 (6): E1032. doi: 10.3390/molecules24061032. PMC 6472197. PMID 30875930.