 | |
| Legal status | |
|---|---|
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C15H15N |
| Molar mass | 209.292 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| Site | IC50 (nM) | Action | Ref |
|---|---|---|---|
| SERT | 83 nM | Blocker | [1] |
| NET | 6 nM | Blocker | [1] |
| DAT | 38 nM | Blocker | [1] |
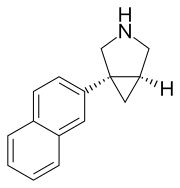
Centanafadine ( INN) (former developmental code name EB-1020) is a serotonin-norepinephrine-dopamine reuptake inhibitor (SNDRI) that began its development with Euthymics Bioscience after they acquired DOV Pharmaceutical. It was developed as a treatment for attention-deficit hyperactivity disorder (ADHD) and inhibits the reuptake of norepinephrine, dopamine, and serotonin with an IC50 ratio of 1:6:14, respectively. [1] [2] [3] [4] In 2011, Euthymics Bioscience spun off its development of centanafadine to a new company called Neurovance. [5] [6] In March 2017, Otsuka Pharmaceutical acquired Neurovance and the rights to centanafadine. [7] As of January 2018, Otsuka's pipeline indicates it is in Phase II and III clinical trials for a number of different applications to medical conditions. [8] [9] [10]
- ^ a b c d e "Neurovance's EB-1020 SR for Adult ADHD Shows Stimulant-Like Efficacy and Good Tolerability in Phase 2a Trial" (PDF). Neurovance. Retrieved 14 January 2018.
- ^ "3-Neurotransmitters, 1-Molecule: Optimized Ratios". Neurovance.
- ^ "EB-1020, a Non-Stimulant Norepinephrine and Dopamine - Preferring Reuptake Inhibitor for the Treatment of Adult ADHD" (PDF). Neurovance. Archived from the original (PDF) on 2015-11-17. Retrieved 2015-11-14.
- ^ Bymaster FP, Golembiowska K, Kowalska M, Choi YK, Tarazi FI (June 2012). "Pharmacological characterization of the norepinephrine and dopamine reuptake inhibitor EB-1020: implications for treatment of attention-deficit hyperactivity disorder". Synapse. 66 (6): 522–32. doi: 10.1002/syn.21538. PMID 22298359. S2CID 38850652.
- ^ "Euthymics". Ethismos Research Inc. Retrieved 14 January 2018.
- ^ "EUTHYMICS BIOSCIENCE, INC. PRESENTS DATA THAT SUPPORT ADVANCING EB-1020 INTO CLINICAL TRIALS FOR ADULT ADHD" (PDF). Neurovance. December 7, 2011. Retrieved 14 January 2018.
- ^ "Otsuka Pharmaceutical to Acquire Neurovance, Inc". Otsuka. Retrieved 14 January 2018.
- ^ "Otsuka U.S. Research & Development Programs". Otsuka U.S. Otsuka. Retrieved 14 January 2018.[ permanent dead link]
-
^ Otsuka Pharmaceutical Development & Commercialization, Inc. (2021-09-17).
"A Phase 3, Randomized, Double-blind, Multicenter, Placebo-controlled, Parallel-group Trial Evaluating the Efficacy, Safety, and Tolerability of Centanafadine Sustained-release Tablets in Adults With Attention-deficit/Hyperactivity Disorder".
{{ cite journal}}: Cite journal requires|journal=( help) - ^ Gunduz-Bruce H (2018-09-26). "SAGE-217 in major depressive disorder: a multicenter, randomized, double-blind, Phase 2 placebo-controlled trial". doi: 10.26226/morressier.5b68175eb56e9b005965c44b. S2CID 266120058. Retrieved 2023-06-26.
 | |
| Legal status | |
|---|---|
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C15H15N |
| Molar mass | 209.292 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| Site | IC50 (nM) | Action | Ref |
|---|---|---|---|
| SERT | 83 nM | Blocker | [1] |
| NET | 6 nM | Blocker | [1] |
| DAT | 38 nM | Blocker | [1] |
Centanafadine ( INN) (former developmental code name EB-1020) is a serotonin-norepinephrine-dopamine reuptake inhibitor (SNDRI) that began its development with Euthymics Bioscience after they acquired DOV Pharmaceutical. It was developed as a treatment for attention-deficit hyperactivity disorder (ADHD) and inhibits the reuptake of norepinephrine, dopamine, and serotonin with an IC50 ratio of 1:6:14, respectively. [1] [2] [3] [4] In 2011, Euthymics Bioscience spun off its development of centanafadine to a new company called Neurovance. [5] [6] In March 2017, Otsuka Pharmaceutical acquired Neurovance and the rights to centanafadine. [7] As of January 2018, Otsuka's pipeline indicates it is in Phase II and III clinical trials for a number of different applications to medical conditions. [8] [9] [10]
- ^ a b c d e "Neurovance's EB-1020 SR for Adult ADHD Shows Stimulant-Like Efficacy and Good Tolerability in Phase 2a Trial" (PDF). Neurovance. Retrieved 14 January 2018.
- ^ "3-Neurotransmitters, 1-Molecule: Optimized Ratios". Neurovance.
- ^ "EB-1020, a Non-Stimulant Norepinephrine and Dopamine - Preferring Reuptake Inhibitor for the Treatment of Adult ADHD" (PDF). Neurovance. Archived from the original (PDF) on 2015-11-17. Retrieved 2015-11-14.
- ^ Bymaster FP, Golembiowska K, Kowalska M, Choi YK, Tarazi FI (June 2012). "Pharmacological characterization of the norepinephrine and dopamine reuptake inhibitor EB-1020: implications for treatment of attention-deficit hyperactivity disorder". Synapse. 66 (6): 522–32. doi: 10.1002/syn.21538. PMID 22298359. S2CID 38850652.
- ^ "Euthymics". Ethismos Research Inc. Retrieved 14 January 2018.
- ^ "EUTHYMICS BIOSCIENCE, INC. PRESENTS DATA THAT SUPPORT ADVANCING EB-1020 INTO CLINICAL TRIALS FOR ADULT ADHD" (PDF). Neurovance. December 7, 2011. Retrieved 14 January 2018.
- ^ "Otsuka Pharmaceutical to Acquire Neurovance, Inc". Otsuka. Retrieved 14 January 2018.
- ^ "Otsuka U.S. Research & Development Programs". Otsuka U.S. Otsuka. Retrieved 14 January 2018.[ permanent dead link]
-
^ Otsuka Pharmaceutical Development & Commercialization, Inc. (2021-09-17).
"A Phase 3, Randomized, Double-blind, Multicenter, Placebo-controlled, Parallel-group Trial Evaluating the Efficacy, Safety, and Tolerability of Centanafadine Sustained-release Tablets in Adults With Attention-deficit/Hyperactivity Disorder".
{{ cite journal}}: Cite journal requires|journal=( help) - ^ Gunduz-Bruce H (2018-09-26). "SAGE-217 in major depressive disorder: a multicenter, randomized, double-blind, Phase 2 placebo-controlled trial". doi: 10.26226/morressier.5b68175eb56e9b005965c44b. S2CID 266120058. Retrieved 2023-06-26.