 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.037.767 |
| Chemical and physical data | |
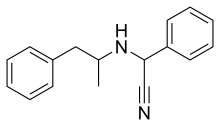
| Formula | C17H18N2 |
| Molar mass | 250.345 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Amfetaminil (also known as amphetaminil, N-cyanobenzylamphetamine, [1] and AN-1; brand name Aponeuron) is a stimulant drug derived from amphetamine, which was developed in the 1970s and used for the treatment of obesity, [2] ADHD, [3] [4] and narcolepsy. [5] It has largely been withdrawn from clinical use following problems with abuse. [6] The drug is a prodrug to amphetamine. [7] [8]
Amfetaminil is a molecule with two stereogenic centers. Thus, four different stereoisomers exist:
- (R)-2-[(R)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile ( CAS number 478392-08-4)
- (S)-2-[(S)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile (CAS number 478392-12-0)
- (R)-2-[(S)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile (CAS number 478392-10-8)
- (S)-2-[(R)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile (CAS number 478392-14-2)

Schiff base formation between amphetamine (1) and benzaldehyde (2) gives benzalamphetamine [2980-02-1] (3). Nucleophilic attack of cyanide anion on the imine (c.f. Strecker reaction) gives amfetaminil (4). Finally, reaction with nitrous acid gives (5). The rearrangement to a Sydnone then occurs to give CID:88166659 (6). Feprosidnine is sans the phenyl group.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 13–. ISBN 978-94-011-4439-1.
- ^ Harris LS (June 1986). "The stimulants and hallucinogens under consideration: a brief overview of their chemistry and pharmacology". Drug and Alcohol Dependence. 17 (2–3): 107–18. doi: 10.1016/0376-8716(86)90002-5. PMID 2874966.
- ^ Meyer-Probst B, Vehreschild T (August 1976). "[Influencing the lack of concentration in hyperkinetic school children with Aponeuron]". Psychiatrie, Neurologie, und Medizinische Psychologie (in German). 28 (8): 491–9. PMID 1005547.
- ^ Paclt I, Florian J, Brunclíková J, Růzicková I (May 1996). "[Effect of Aponeuron in the treatment of children with hyperkinetic syndrome]". Ceska a Slovenska Psychiatrie (in Czech). 92 (Suppl 1): 41–57. PMID 8768943.
- ^ Schlesser JL (1991). Drugs Available Abroad - A Guide to Therapeutic Drugs Approved Outside the US. Detroit: MEDEX Books.
- ^ Winter E (September 1976). "[Drug abuse and dependence of the amphetamine type with special regard to Amphetaminil (Aponeuron(R))]". Psychiatrie, Neurologie, und Medizinische Psychologie (in German). 28 (9): 513–25. PMID 1005549.
- ^ Dasgupta A (2 July 2012). Resolving Erroneous Reports in Toxicology and Therapeutic Drug Monitoring: A Comprehensive Guide. John Wiley & Sons. pp. 96–. ISBN 978-1-118-34785-0.
- ^ AHC Media, LLC (17 March 2014). Pediatric Trauma Care II: A clinical reference for physicians and nurses caring for the acutely injured child. AHC Media, LLC. pp. 118–. ISBN 978-1-934863-59-6.
- ^ Yashunskii VG, Gorkin VZ, Mashkovskii MD, Altshuler RA, Veryovkina IV, Kholodov LE (October 1971). "Synthesis and pharmacological effects of some alkyl-, aryl-, and aralkylsydnonimines". Journal of Medicinal Chemistry. 14 (10): 1013–5. doi: 10.1021/jm00292a042. PMID 5165569.
- ^ Klosa J (August 1975). "[The stability of amphetaminil. Syntheses with amphetaminil (author's transl)]". Arzneimittel-Forschung (in German). 25 (8): 1252–8. PMID 1242355.
- ^ Klosa J (1975). "[On the crystallisation of amphetaminil base into its hydrochloride salt (author's transl)]". Arzneimittel-Forschung (in German). 25 (12): 1863–4. PMID 1243655.
- ^ Beyer KH, Strassner W, Klinge D. "Amphetaminil [α-(1-benzylethylamino)-α-phenylacetonitrile]". Deutsche Apotheker Zeitung. 111 (19): 677–680.
- ^ Kholodov LE (1967). "Sydnones and sydnonimines. XXXV. Salts and exocyclic derivatives of some sydnonimines". Zhurnal Organicheskoi Khimii. 3 (8): 1513–1518.
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.037.767 |
| Chemical and physical data | |
| Formula | C17H18N2 |
| Molar mass | 250.345 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Amfetaminil (also known as amphetaminil, N-cyanobenzylamphetamine, [1] and AN-1; brand name Aponeuron) is a stimulant drug derived from amphetamine, which was developed in the 1970s and used for the treatment of obesity, [2] ADHD, [3] [4] and narcolepsy. [5] It has largely been withdrawn from clinical use following problems with abuse. [6] The drug is a prodrug to amphetamine. [7] [8]
Amfetaminil is a molecule with two stereogenic centers. Thus, four different stereoisomers exist:
- (R)-2-[(R)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile ( CAS number 478392-08-4)
- (S)-2-[(S)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile (CAS number 478392-12-0)
- (R)-2-[(S)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile (CAS number 478392-10-8)
- (S)-2-[(R)-1-Phenylpropan-2-ylamino]-2-phenylacetonitrile (CAS number 478392-14-2)

Schiff base formation between amphetamine (1) and benzaldehyde (2) gives benzalamphetamine [2980-02-1] (3). Nucleophilic attack of cyanide anion on the imine (c.f. Strecker reaction) gives amfetaminil (4). Finally, reaction with nitrous acid gives (5). The rearrangement to a Sydnone then occurs to give CID:88166659 (6). Feprosidnine is sans the phenyl group.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 13–. ISBN 978-94-011-4439-1.
- ^ Harris LS (June 1986). "The stimulants and hallucinogens under consideration: a brief overview of their chemistry and pharmacology". Drug and Alcohol Dependence. 17 (2–3): 107–18. doi: 10.1016/0376-8716(86)90002-5. PMID 2874966.
- ^ Meyer-Probst B, Vehreschild T (August 1976). "[Influencing the lack of concentration in hyperkinetic school children with Aponeuron]". Psychiatrie, Neurologie, und Medizinische Psychologie (in German). 28 (8): 491–9. PMID 1005547.
- ^ Paclt I, Florian J, Brunclíková J, Růzicková I (May 1996). "[Effect of Aponeuron in the treatment of children with hyperkinetic syndrome]". Ceska a Slovenska Psychiatrie (in Czech). 92 (Suppl 1): 41–57. PMID 8768943.
- ^ Schlesser JL (1991). Drugs Available Abroad - A Guide to Therapeutic Drugs Approved Outside the US. Detroit: MEDEX Books.
- ^ Winter E (September 1976). "[Drug abuse and dependence of the amphetamine type with special regard to Amphetaminil (Aponeuron(R))]". Psychiatrie, Neurologie, und Medizinische Psychologie (in German). 28 (9): 513–25. PMID 1005549.
- ^ Dasgupta A (2 July 2012). Resolving Erroneous Reports in Toxicology and Therapeutic Drug Monitoring: A Comprehensive Guide. John Wiley & Sons. pp. 96–. ISBN 978-1-118-34785-0.
- ^ AHC Media, LLC (17 March 2014). Pediatric Trauma Care II: A clinical reference for physicians and nurses caring for the acutely injured child. AHC Media, LLC. pp. 118–. ISBN 978-1-934863-59-6.
- ^ Yashunskii VG, Gorkin VZ, Mashkovskii MD, Altshuler RA, Veryovkina IV, Kholodov LE (October 1971). "Synthesis and pharmacological effects of some alkyl-, aryl-, and aralkylsydnonimines". Journal of Medicinal Chemistry. 14 (10): 1013–5. doi: 10.1021/jm00292a042. PMID 5165569.
- ^ Klosa J (August 1975). "[The stability of amphetaminil. Syntheses with amphetaminil (author's transl)]". Arzneimittel-Forschung (in German). 25 (8): 1252–8. PMID 1242355.
- ^ Klosa J (1975). "[On the crystallisation of amphetaminil base into its hydrochloride salt (author's transl)]". Arzneimittel-Forschung (in German). 25 (12): 1863–4. PMID 1243655.
- ^ Beyer KH, Strassner W, Klinge D. "Amphetaminil [α-(1-benzylethylamino)-α-phenylacetonitrile]". Deutsche Apotheker Zeitung. 111 (19): 677–680.
- ^ Kholodov LE (1967). "Sydnones and sydnonimines. XXXV. Salts and exocyclic derivatives of some sydnonimines". Zhurnal Organicheskoi Khimii. 3 (8): 1513–1518.
