 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
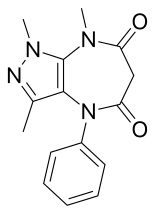
| Formula | C15H16N4O2 |
| Molar mass | 284.319 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Zomebazam [1] produced by Hoechst is a pyrazolodiazepinone derivative drug with anxiolytic properties. It is structurally related to razobazam and zometapine. [2]
Synthesis
The catalytic hydrogenation of N,2,5-trimethyl-4-phenyldiazenylpyrazol-3-amine [a] (1) over Raney nickel gives 4-amino-1,3-dimethyl-5-methylaminopyrazole [b] (2). Treatment with methyl malonyl chloride [c] (3) gives 4-α-ethoxycarbonylacetylamino-1,3-dimethyl-5-methylaminopyrazole [d] (4). Base-catalyzed lactamization gives (5). The Goldberg reaction completes the synthesis of zomebazam (6). [3] [4]

See also
References
- ^ US 3558605, "4-Aryl-5,6,7,8-tetrahydropyrazolo(3,4-B)-(1,5)diazepine-1H,4H-5,7-diones and medicaments containing same"
- ^ "Zomebazam". psychotropics.dk. 2003. Retrieved 7 December 2009.
- ^ Renger B (1985). "Direkte N-Arylierung von Amiden: Eine Verbesserung der Goldberg-Reaktion". Synthesis. 1985 (9): 856–560. doi: 10.1055/s-1985-31364. S2CID 93397774.
- ^ US 4302468, Rackur G, Hoffmann I, issued 1981, assigned to Hoechst Aktiengesellschaft
Notes
- ^ CID:136203602
- ^ CID:10219477
- ^ CAS# [37517-81-0]
- ^ CID:20561101
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C15H16N4O2 |
| Molar mass | 284.319 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Zomebazam [1] produced by Hoechst is a pyrazolodiazepinone derivative drug with anxiolytic properties. It is structurally related to razobazam and zometapine. [2]
Synthesis
The catalytic hydrogenation of N,2,5-trimethyl-4-phenyldiazenylpyrazol-3-amine [a] (1) over Raney nickel gives 4-amino-1,3-dimethyl-5-methylaminopyrazole [b] (2). Treatment with methyl malonyl chloride [c] (3) gives 4-α-ethoxycarbonylacetylamino-1,3-dimethyl-5-methylaminopyrazole [d] (4). Base-catalyzed lactamization gives (5). The Goldberg reaction completes the synthesis of zomebazam (6). [3] [4]

See also
References
- ^ US 3558605, "4-Aryl-5,6,7,8-tetrahydropyrazolo(3,4-B)-(1,5)diazepine-1H,4H-5,7-diones and medicaments containing same"
- ^ "Zomebazam". psychotropics.dk. 2003. Retrieved 7 December 2009.
- ^ Renger B (1985). "Direkte N-Arylierung von Amiden: Eine Verbesserung der Goldberg-Reaktion". Synthesis. 1985 (9): 856–560. doi: 10.1055/s-1985-31364. S2CID 93397774.
- ^ US 4302468, Rackur G, Hoffmann I, issued 1981, assigned to Hoechst Aktiengesellschaft
Notes
- ^ CID:136203602
- ^ CID:10219477
- ^ CAS# [37517-81-0]
- ^ CID:20561101