 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Pharmacokinetic data | |
| Elimination half-life | 2.5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
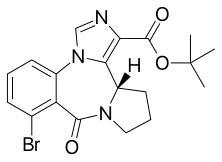
| Formula | C19H20BrN3O3 |
| Molar mass | 418.291 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Bretazenil (Ro16-6028) is an imidazopyrrolobenzodiazepine [1] anxiolytic drug which is derived from the benzodiazepine family, and was invented in 1988. It is most closely related in structure to the GABA antagonist flumazenil, although its effects are somewhat different. It is classified as a high-potency benzodiazepine due to its high affinity binding to benzodiazepine binding sites [2] [3] where it acts as a partial agonist. [4] Its profile as a partial agonist and preclinical trial data suggests that it may have a reduced adverse effect profile. [5] In particular bretazenil has been proposed to cause a less strong development of tolerance and withdrawal syndrome. [6] Bretazenil differs from traditional 1,4-benzodiazepines by being a partial agonist and because it binds to α1, α2, α3, α4, α5 and α6 subunit containing GABAA receptor benzodiazepine receptor complexes. 1,4-benzodiazepines bind only to α1, α2, α3 and α5 GABAA benzodiazepine receptor complexes. [1]
History
Bretazenil was originally developed as an anti-anxiety drug and has been studied for its use as an anticonvulsant [7] [8] but has never commercialised. It is a partial agonist for the benzodiazepine site of the GABAA receptors in the brain. David Nutt from the University of Bristol has suggested bretazenil as a possible base from which to make a better social drug, as it displays several of the positive effects of alcohol intoxication such as relaxation and sociability, but without the bad effects such as aggression, amnesia, nausea, loss of coordination, liver disease and brain damage. The effects of bretazenil can also be quickly reversed by the action of flumazenil, which is used as an antidote to benzodiazepine overdose, [9] in contrast to alcohol for which there is no effective and reliable antidote.[ citation needed]
Traditional benzodiazepines are associated with side effects such as drowsiness, physical dependence and abuse potential. It was hoped that bretazenil and other partial agonists would be an improvement on traditional benzodiazepines which are full agonists due to preclinical evidence that their side effect profile was less than that of full agonist benzodiazepines. For a variety of reasons however, bretazenil and other partial agonists such as pazinaclone and abecarnil were not clinically successful. However, research continues into other compounds with partial agonist and compounds which are selective for certain GABAA benzodiazepine receptor subtypes. [10]
Tolerance and dependence
In a study in rats, cross-tolerance between the benzodiazepine drug chlordiazepoxide and bretazenil has been demonstrated. [11] In a primate study bretazenil was found to be able to replace the full agonist diazepam in diazepam dependent primates without precipitating withdrawal effects, demonstrating cross tolerance between bretazenil and benzodiazepine agonists, whereas other partial agonists precipitated a withdrawal syndrome. The differences are likely due to differences in instrinsic properties between different benzodiazepine partial agonists. [12] Cross-tolerance has also been shown between bretazenil and full agonist benzodiazepines in rats. [13] In rats tolerance is slower to develop to the anticonvulsant effects compared to the benzodiazepine site full agonist diazepam. However, tolerance developed to the anticonvulsant effects of bretazenil partial agonist more quickly than they developed to imidazenil. [14]
Pharmacology
Bretazenil has a more broad spectrum of action than traditional benzodiazepines as it has been shown to have low affinity binding to α4 and α6 GABAA receptors in addition to acting on α1, α2, α3 and α5 subunits which traditional benzodiazepine drugs work on. The partial agonist imidazenil does not, however, act at these subunits. [15] [16] [17] 0.5mg of bretazenil is approximately equivalent in its psychomotor-impairing effect to 10 mg of diazepam. Bretazenil produces marked sedative-hypnotic effects when taken alone and when combined with alcohol. This human study also indicates that bretazenil is possibly more sedative than diazepam. The reason is unknown, but the study suggests the possibility that a full-agonist metabolite may be generated in humans but not animals previously tested or else that there are significant differences in benzodiazepine receptor population in animals and humans. [18]
In a study of monkeys bretazenil has been found to antagonize the effects of full agonist benzodiazepines. However, bretazenil has been found to enhance the effects of neurosteroids acting on the neurosteroid binding site of the GABAA receptor. [19] Another study found that bretazenil acted as an antagonist provoking withdrawal symptoms in monkeys who were physically dependent on the full agonist benzodiazepine triazolam. [20]
Partial agonists of benzodiazepine receptors have been proposed as a possible alternative to full agonists of the benzodiazepine site to overcome the problems of tolerance, dependence and withdrawal which limits the role of benzodiazepines in the treatment of anxiety, insomnia and epilepsy. Such adverse effects appear to be less problematic with bretazenil than full agonists. [21] Bretazenil has also been found to have less abuse potential than benzodiazepine full agonists such as diazepam and alprazolam, [22] [23] however long-term use of bretazenil would still be expected to result in dependence and addiction.[ citation needed]
Bretazenil alters the sleep EEG profile and causes a reduction in cortisol secretion and increases significantly the release of prolactin. [24] Bretazenil has effective hypnotic properties but impairs cognitive ability in humans. Bretazenil causes a reduction in the number of movements between sleep stages and delays movement into REM sleep. At a dosage of 0.5 mg of bretazenil REM sleep is decreased and stage 2 sleep is lengthened. [25]
See also
References
- ^ a b Pym LJ, Cook SM, Rosahl T, McKernan RM, Atack JR (November 2005). "Selective labelling of diazepam-insensitive GABAA receptors in vivo using [3H]Ro 15-4513". British Journal of Pharmacology. 146 (6): 817–25. doi: 10.1038/sj.bjp.0706392. PMC 1751217. PMID 16184188.
- ^ Giusti P, Guidetti G, Costa E, Guidotti A (June 1991). "The preferential antagonism of pentylenetetrazole proconflict responses differentiates a class of anxiolytic benzodiazepines with potential antipanic action". The Journal of Pharmacology and Experimental Therapeutics. 257 (3): 1062–8. PMID 1675286.
- ^ Finn DA, Gee KW (November 1993). "A comparison of Ro 16-6028 with benzodiazepine receptor 'full agonists' on GABAA receptor function". European Journal of Pharmacology. 247 (3): 233–7. doi: 10.1016/0922-4106(93)90190-K. PMID 7905829.
- ^ Haefely W, Facklam M, Schoch P, Martin JR, Bonetti EP, Moreau JL, et al. (1992). "Partial agonists of benzodiazepine receptors for the treatment of epilepsy, sleep, and anxiety disorders". Advances in Biochemical Psychopharmacology. 47: 379–94. PMID 1324584.
- ^ Kunovac JL, Stahl SM (December 1995). "Future directions in anxiolytic pharmacotherapy". The Psychiatric Clinics of North America. 18 (4): 895–909. doi: 10.1016/S0193-953X(18)30030-3. PMID 8748388.
- ^ Płaźnik A (1995). "Pharmacology of tolerance to benzodiazepine receptor ligands". Polish Journal of Pharmacology. 47 (6): 489–99. PMID 8868371.
- ^ Cole JC, Rodgers RJ (December 1993). "An ethological analysis of the effects of chlordiazepoxide and bretazenil (Ro 16-6028) in the murine elevated plus-maze". Behavioural Pharmacology. 4 (6): 573–580. doi: 10.1097/00008877-199312000-00003. PMID 11224226.
- ^ Brabcová R, Kubová H, Velísek L, Mares P (1993). "Effects of a benzodiazepine, bretazenil (Ro 16-6028), on rhythmic metrazol EEG activity: comparison with standard anticonvulsants". Epilepsia. 34 (6): 1135–40. doi: 10.1111/j.1528-1157.1993.tb02146.x. PMID 8243369. S2CID 24157328.
- ^ Nutt DJ (May 2006). "Alcohol alternatives--a goal for psychopharmacology?". Journal of Psychopharmacology. 20 (3): 318–20. doi: 10.1177/0269881106063042. PMID 16574703. S2CID 44290147.
- ^ Atack JR (May 2005). "The benzodiazepine binding site of GABA(A) receptors as a target for the development of novel anxiolytics". Expert Opinion on Investigational Drugs. 14 (5): 601–18. doi: 10.1517/13543784.14.5.601. PMID 15926867. S2CID 22793644.
- ^ Bronson ME (1993). "Tolerance/cross-tolerance to the discriminative stimulus effects of chlordiazepoxide and bretazenil". Molecular and Chemical Neuropathology. 18 (1–2): 85–98. doi: 10.1007/BF03160023. PMID 8385466.
- ^ Martin JR, Jenck F, Moreau JL (October 1995). "Comparison of benzodiazepine receptor ligands with partial agonistic, antagonistic or partial inverse agonistic properties in precipitating withdrawal in squirrel monkeys". The Journal of Pharmacology and Experimental Therapeutics. 275 (1): 405–11. PMID 7562578.
- ^ Bronson ME (Jun–Jul 1995). "Chronic bretazenil produces tolerance to chlordiazepoxide, midazolam, and abecarnil". Pharmacology, Biochemistry, and Behavior. 51 (2–3): 481–90. doi: 10.1016/0091-3057(95)00014-N. PMID 7667373. S2CID 26419284.
- ^ Auta J, Giusti P, Guidotti A, Costa E (September 1994). "Imidazenil, a partial positive allosteric modulator of GABAA receptors, exhibits low tolerance and dependence liabilities in the rat". The Journal of Pharmacology and Experimental Therapeutics. 270 (3): 1262–9. PMID 7932179.
- ^ Brown N, Kerby J, Bonnert TP, Whiting PJ, Wafford KA (August 2002). "Pharmacological characterization of a novel cell line expressing human alpha(4)beta(3)delta GABA(A) receptors". British Journal of Pharmacology. 136 (7): 965–74. doi: 10.1038/sj.bjp.0704795. PMC 1573424. PMID 12145096.
- ^ Witkin JM, Acri JB, Gleeson S, Barrett JE (January 1997). "Blockade of behavioral effects of bretazenil by flumazenil and ZK 93,426 in pigeons". Pharmacology, Biochemistry, and Behavior. 56 (1): 1–7. doi: 10.1016/S0091-3057(96)00120-7. PMID 8981602. S2CID 36088521.
- ^ Knoflach F, Benke D, Wang Y, Scheurer L, Lüddens H, Hamilton BJ, et al. (November 1996). "Pharmacological modulation of the diazepam-insensitive recombinant gamma-aminobutyric acidA receptors alpha 4 beta 2 gamma 2 and alpha 6 beta 2 gamma 2". Molecular Pharmacology. 50 (5): 1253–61. PMID 8913357.
- ^ van Steveninck AL, Gieschke R, Schoemaker RC, Roncari G, Tuk B, Pieters MS, et al. (June 1996). "Pharmacokinetic and pharmacodynamic interactions of bretazenil and diazepam with alcohol". British Journal of Clinical Pharmacology. 41 (6): 565–73. doi: 10.1046/j.1365-2125.1996.38514.x. PMC 2042631. PMID 8799523.
- ^ McMahon LR, France CP (February 2006). "Differential behavioral effects of low efficacy positive GABAA modulators in combination with benzodiazepines and a neuroactive steroid in rhesus monkeys". British Journal of Pharmacology. 147 (3): 260–8. doi: 10.1038/sj.bjp.0706550. PMC 1751295. PMID 16331290.
- ^ Weerts EM, Ator NA, Kaminski BJ, Griffiths RR (September 2005). "Comparison of the behavioral effects of bretazenil and flumazenil in triazolam-dependent and non-dependent baboons". European Journal of Pharmacology. 519 (1–2): 103–13. doi: 10.1016/j.ejphar.2005.06.038. PMID 16129429.
- ^ Schoch P, Moreau JL, Martin JR, Haefely WE (1993). "Aspects of benzodiazepine receptor structure and function with relevance to drug tolerance and dependence". Biochemical Society Symposium. 59: 121–34. PMID 7910739.
- ^ Busto U, Kaplan HL, Zawertailo L, Sellers EM (April 1994). "Pharmacologic effects and abuse liability of bretazenil, diazepam, and alprazolam in humans". Clinical Pharmacology and Therapeutics. 55 (4): 451–63. doi: 10.1038/clpt.1994.55. PMID 8162672. S2CID 21357076.
- ^ Sellers EM, Kaplan HL, Busto UE, et al. (1992). "Abuse liability of bretazenil and other partial agonists". Clinical Neuropharmacology. 15 Suppl 1 Pt A: 409A. doi: 10.1097/00002826-199201001-00213. PMID 1354047. S2CID 40095405.
- ^ Guldner J, Trachsel L, Kratschmayr C, Rothe B, Holsboer F, Steiger A (November 1995). "Bretazenil modulates sleep EEG and nocturnal hormone secretion in normal men". Psychopharmacology. 122 (2): 115–21. doi: 10.1007/BF02246085. PMID 8848526. S2CID 34247989.
- ^ Gieschke R, Cluydts R, Dingemanse J, De Roeck J, De Cock W (November 1994). "Effects of bretazenil vs. zolpidem and placebo on experimentally induced sleep disturbance in healthy volunteers". Methods and Findings in Experimental and Clinical Pharmacology. 16 (9): 667–75. PMID 7746029.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Pharmacokinetic data | |
| Elimination half-life | 2.5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C19H20BrN3O3 |
| Molar mass | 418.291 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Bretazenil (Ro16-6028) is an imidazopyrrolobenzodiazepine [1] anxiolytic drug which is derived from the benzodiazepine family, and was invented in 1988. It is most closely related in structure to the GABA antagonist flumazenil, although its effects are somewhat different. It is classified as a high-potency benzodiazepine due to its high affinity binding to benzodiazepine binding sites [2] [3] where it acts as a partial agonist. [4] Its profile as a partial agonist and preclinical trial data suggests that it may have a reduced adverse effect profile. [5] In particular bretazenil has been proposed to cause a less strong development of tolerance and withdrawal syndrome. [6] Bretazenil differs from traditional 1,4-benzodiazepines by being a partial agonist and because it binds to α1, α2, α3, α4, α5 and α6 subunit containing GABAA receptor benzodiazepine receptor complexes. 1,4-benzodiazepines bind only to α1, α2, α3 and α5 GABAA benzodiazepine receptor complexes. [1]
History
Bretazenil was originally developed as an anti-anxiety drug and has been studied for its use as an anticonvulsant [7] [8] but has never commercialised. It is a partial agonist for the benzodiazepine site of the GABAA receptors in the brain. David Nutt from the University of Bristol has suggested bretazenil as a possible base from which to make a better social drug, as it displays several of the positive effects of alcohol intoxication such as relaxation and sociability, but without the bad effects such as aggression, amnesia, nausea, loss of coordination, liver disease and brain damage. The effects of bretazenil can also be quickly reversed by the action of flumazenil, which is used as an antidote to benzodiazepine overdose, [9] in contrast to alcohol for which there is no effective and reliable antidote.[ citation needed]
Traditional benzodiazepines are associated with side effects such as drowsiness, physical dependence and abuse potential. It was hoped that bretazenil and other partial agonists would be an improvement on traditional benzodiazepines which are full agonists due to preclinical evidence that their side effect profile was less than that of full agonist benzodiazepines. For a variety of reasons however, bretazenil and other partial agonists such as pazinaclone and abecarnil were not clinically successful. However, research continues into other compounds with partial agonist and compounds which are selective for certain GABAA benzodiazepine receptor subtypes. [10]
Tolerance and dependence
In a study in rats, cross-tolerance between the benzodiazepine drug chlordiazepoxide and bretazenil has been demonstrated. [11] In a primate study bretazenil was found to be able to replace the full agonist diazepam in diazepam dependent primates without precipitating withdrawal effects, demonstrating cross tolerance between bretazenil and benzodiazepine agonists, whereas other partial agonists precipitated a withdrawal syndrome. The differences are likely due to differences in instrinsic properties between different benzodiazepine partial agonists. [12] Cross-tolerance has also been shown between bretazenil and full agonist benzodiazepines in rats. [13] In rats tolerance is slower to develop to the anticonvulsant effects compared to the benzodiazepine site full agonist diazepam. However, tolerance developed to the anticonvulsant effects of bretazenil partial agonist more quickly than they developed to imidazenil. [14]
Pharmacology
Bretazenil has a more broad spectrum of action than traditional benzodiazepines as it has been shown to have low affinity binding to α4 and α6 GABAA receptors in addition to acting on α1, α2, α3 and α5 subunits which traditional benzodiazepine drugs work on. The partial agonist imidazenil does not, however, act at these subunits. [15] [16] [17] 0.5mg of bretazenil is approximately equivalent in its psychomotor-impairing effect to 10 mg of diazepam. Bretazenil produces marked sedative-hypnotic effects when taken alone and when combined with alcohol. This human study also indicates that bretazenil is possibly more sedative than diazepam. The reason is unknown, but the study suggests the possibility that a full-agonist metabolite may be generated in humans but not animals previously tested or else that there are significant differences in benzodiazepine receptor population in animals and humans. [18]
In a study of monkeys bretazenil has been found to antagonize the effects of full agonist benzodiazepines. However, bretazenil has been found to enhance the effects of neurosteroids acting on the neurosteroid binding site of the GABAA receptor. [19] Another study found that bretazenil acted as an antagonist provoking withdrawal symptoms in monkeys who were physically dependent on the full agonist benzodiazepine triazolam. [20]
Partial agonists of benzodiazepine receptors have been proposed as a possible alternative to full agonists of the benzodiazepine site to overcome the problems of tolerance, dependence and withdrawal which limits the role of benzodiazepines in the treatment of anxiety, insomnia and epilepsy. Such adverse effects appear to be less problematic with bretazenil than full agonists. [21] Bretazenil has also been found to have less abuse potential than benzodiazepine full agonists such as diazepam and alprazolam, [22] [23] however long-term use of bretazenil would still be expected to result in dependence and addiction.[ citation needed]
Bretazenil alters the sleep EEG profile and causes a reduction in cortisol secretion and increases significantly the release of prolactin. [24] Bretazenil has effective hypnotic properties but impairs cognitive ability in humans. Bretazenil causes a reduction in the number of movements between sleep stages and delays movement into REM sleep. At a dosage of 0.5 mg of bretazenil REM sleep is decreased and stage 2 sleep is lengthened. [25]
See also
References
- ^ a b Pym LJ, Cook SM, Rosahl T, McKernan RM, Atack JR (November 2005). "Selective labelling of diazepam-insensitive GABAA receptors in vivo using [3H]Ro 15-4513". British Journal of Pharmacology. 146 (6): 817–25. doi: 10.1038/sj.bjp.0706392. PMC 1751217. PMID 16184188.
- ^ Giusti P, Guidetti G, Costa E, Guidotti A (June 1991). "The preferential antagonism of pentylenetetrazole proconflict responses differentiates a class of anxiolytic benzodiazepines with potential antipanic action". The Journal of Pharmacology and Experimental Therapeutics. 257 (3): 1062–8. PMID 1675286.
- ^ Finn DA, Gee KW (November 1993). "A comparison of Ro 16-6028 with benzodiazepine receptor 'full agonists' on GABAA receptor function". European Journal of Pharmacology. 247 (3): 233–7. doi: 10.1016/0922-4106(93)90190-K. PMID 7905829.
- ^ Haefely W, Facklam M, Schoch P, Martin JR, Bonetti EP, Moreau JL, et al. (1992). "Partial agonists of benzodiazepine receptors for the treatment of epilepsy, sleep, and anxiety disorders". Advances in Biochemical Psychopharmacology. 47: 379–94. PMID 1324584.
- ^ Kunovac JL, Stahl SM (December 1995). "Future directions in anxiolytic pharmacotherapy". The Psychiatric Clinics of North America. 18 (4): 895–909. doi: 10.1016/S0193-953X(18)30030-3. PMID 8748388.
- ^ Płaźnik A (1995). "Pharmacology of tolerance to benzodiazepine receptor ligands". Polish Journal of Pharmacology. 47 (6): 489–99. PMID 8868371.
- ^ Cole JC, Rodgers RJ (December 1993). "An ethological analysis of the effects of chlordiazepoxide and bretazenil (Ro 16-6028) in the murine elevated plus-maze". Behavioural Pharmacology. 4 (6): 573–580. doi: 10.1097/00008877-199312000-00003. PMID 11224226.
- ^ Brabcová R, Kubová H, Velísek L, Mares P (1993). "Effects of a benzodiazepine, bretazenil (Ro 16-6028), on rhythmic metrazol EEG activity: comparison with standard anticonvulsants". Epilepsia. 34 (6): 1135–40. doi: 10.1111/j.1528-1157.1993.tb02146.x. PMID 8243369. S2CID 24157328.
- ^ Nutt DJ (May 2006). "Alcohol alternatives--a goal for psychopharmacology?". Journal of Psychopharmacology. 20 (3): 318–20. doi: 10.1177/0269881106063042. PMID 16574703. S2CID 44290147.
- ^ Atack JR (May 2005). "The benzodiazepine binding site of GABA(A) receptors as a target for the development of novel anxiolytics". Expert Opinion on Investigational Drugs. 14 (5): 601–18. doi: 10.1517/13543784.14.5.601. PMID 15926867. S2CID 22793644.
- ^ Bronson ME (1993). "Tolerance/cross-tolerance to the discriminative stimulus effects of chlordiazepoxide and bretazenil". Molecular and Chemical Neuropathology. 18 (1–2): 85–98. doi: 10.1007/BF03160023. PMID 8385466.
- ^ Martin JR, Jenck F, Moreau JL (October 1995). "Comparison of benzodiazepine receptor ligands with partial agonistic, antagonistic or partial inverse agonistic properties in precipitating withdrawal in squirrel monkeys". The Journal of Pharmacology and Experimental Therapeutics. 275 (1): 405–11. PMID 7562578.
- ^ Bronson ME (Jun–Jul 1995). "Chronic bretazenil produces tolerance to chlordiazepoxide, midazolam, and abecarnil". Pharmacology, Biochemistry, and Behavior. 51 (2–3): 481–90. doi: 10.1016/0091-3057(95)00014-N. PMID 7667373. S2CID 26419284.
- ^ Auta J, Giusti P, Guidotti A, Costa E (September 1994). "Imidazenil, a partial positive allosteric modulator of GABAA receptors, exhibits low tolerance and dependence liabilities in the rat". The Journal of Pharmacology and Experimental Therapeutics. 270 (3): 1262–9. PMID 7932179.
- ^ Brown N, Kerby J, Bonnert TP, Whiting PJ, Wafford KA (August 2002). "Pharmacological characterization of a novel cell line expressing human alpha(4)beta(3)delta GABA(A) receptors". British Journal of Pharmacology. 136 (7): 965–74. doi: 10.1038/sj.bjp.0704795. PMC 1573424. PMID 12145096.
- ^ Witkin JM, Acri JB, Gleeson S, Barrett JE (January 1997). "Blockade of behavioral effects of bretazenil by flumazenil and ZK 93,426 in pigeons". Pharmacology, Biochemistry, and Behavior. 56 (1): 1–7. doi: 10.1016/S0091-3057(96)00120-7. PMID 8981602. S2CID 36088521.
- ^ Knoflach F, Benke D, Wang Y, Scheurer L, Lüddens H, Hamilton BJ, et al. (November 1996). "Pharmacological modulation of the diazepam-insensitive recombinant gamma-aminobutyric acidA receptors alpha 4 beta 2 gamma 2 and alpha 6 beta 2 gamma 2". Molecular Pharmacology. 50 (5): 1253–61. PMID 8913357.
- ^ van Steveninck AL, Gieschke R, Schoemaker RC, Roncari G, Tuk B, Pieters MS, et al. (June 1996). "Pharmacokinetic and pharmacodynamic interactions of bretazenil and diazepam with alcohol". British Journal of Clinical Pharmacology. 41 (6): 565–73. doi: 10.1046/j.1365-2125.1996.38514.x. PMC 2042631. PMID 8799523.
- ^ McMahon LR, France CP (February 2006). "Differential behavioral effects of low efficacy positive GABAA modulators in combination with benzodiazepines and a neuroactive steroid in rhesus monkeys". British Journal of Pharmacology. 147 (3): 260–8. doi: 10.1038/sj.bjp.0706550. PMC 1751295. PMID 16331290.
- ^ Weerts EM, Ator NA, Kaminski BJ, Griffiths RR (September 2005). "Comparison of the behavioral effects of bretazenil and flumazenil in triazolam-dependent and non-dependent baboons". European Journal of Pharmacology. 519 (1–2): 103–13. doi: 10.1016/j.ejphar.2005.06.038. PMID 16129429.
- ^ Schoch P, Moreau JL, Martin JR, Haefely WE (1993). "Aspects of benzodiazepine receptor structure and function with relevance to drug tolerance and dependence". Biochemical Society Symposium. 59: 121–34. PMID 7910739.
- ^ Busto U, Kaplan HL, Zawertailo L, Sellers EM (April 1994). "Pharmacologic effects and abuse liability of bretazenil, diazepam, and alprazolam in humans". Clinical Pharmacology and Therapeutics. 55 (4): 451–63. doi: 10.1038/clpt.1994.55. PMID 8162672. S2CID 21357076.
- ^ Sellers EM, Kaplan HL, Busto UE, et al. (1992). "Abuse liability of bretazenil and other partial agonists". Clinical Neuropharmacology. 15 Suppl 1 Pt A: 409A. doi: 10.1097/00002826-199201001-00213. PMID 1354047. S2CID 40095405.
- ^ Guldner J, Trachsel L, Kratschmayr C, Rothe B, Holsboer F, Steiger A (November 1995). "Bretazenil modulates sleep EEG and nocturnal hormone secretion in normal men". Psychopharmacology. 122 (2): 115–21. doi: 10.1007/BF02246085. PMID 8848526. S2CID 34247989.
- ^ Gieschke R, Cluydts R, Dingemanse J, De Roeck J, De Cock W (November 1994). "Effects of bretazenil vs. zolpidem and placebo on experimentally induced sleep disturbance in healthy volunteers". Methods and Findings in Experimental and Clinical Pharmacology. 16 (9): 667–75. PMID 7746029.