This article needs additional citations for
verification. (September 2022) |
 | |
 | |
| Clinical data | |
|---|---|
| Other names | (-)-Trans-Δ9-tetrahydrocannabiphorol Δ9-THCP (C7)-Δ9-THC THC-Heptyl |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C23H34O2 |
| Molar mass | 342.523 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
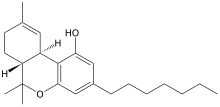
Tetrahydrocannabiphorol (THCP) is a potent phytocannabinoid, a CB1 and CB2 agonist which was known as a synthetic homologue of THC, [1] but for the first time in 2019 was isolated as a natural product in trace amounts from Cannabis sativa. [2] [3] It is structurally similar to Δ9- THC, the main active component of cannabis, but with the pentyl side chain extended to heptyl. Since it has a longer side chain, its cannabinoid effects are "far higher than Δ9-THC itself." Tetrahydrocannabiphorol has a reported binding affinity of 1.2 nM at CB1, approximately 33 times that of Δ9-THC (40 nM at CB1). [4]
THCP was studied by Roger Adams as early as 1942. [5]
Isomers
Delta-3-THCP

The Δ3/Δ6a(10a) isomer Δ3-THCP was synthesised in 1941, and was found to have around the same potency as Δ3-THC, unlike the hexyl homologue parahexyl which was significantly stronger. [6]
Delta-8-THCP

The Δ8 isomer is also known as a synthetic cannabinoid under the code name JWH-091. [7] [8] It's unconfirmed whether or not Δ8-THCP is found naturally in cannabis plants, but likely is due to Δ8-THC itself being a degraded form of Δ9-THC. [9] JWH-091 has approximately double the binding affinity at the CB1 receptor (22 nM ± 3.9 nM) in comparison to Δ9-THC (40.7 nM ± 1.7 nM) or Δ8-THC (44 nM ± 12 nM), [8] but appears significantly lower in vitro than the binding activity of Δ9-THCP (Ki = 1.2 nM) [4]
Natural occurrence in cannabis
The Δ9 isomer of THCP occurs naturally in cannabis, but in small amounts. A 2021 study reported the content of Δ9-THCP ranging from 0.0023% to 0.0136% (w/w) (approximately 0.02–0.13 mg/g) without correlation to THC percentage in Δ9-THC-dominant strains of cannabis; that study failed to detect THCP in CBD-dominant strains. [2] [10]
See also
- Nabilone
- Hexahydrocannabiphorol
- 1,2-Didehydro-3-oxo-THCO
- Delta-8-THC
- Parahexyl
- Perrottetinene
- Tetrahydrocannabivarin
- Tetrahydrocannabutol
- Tetrahydrocannabihexol
- THCP-O-acetate
References
- ^ Harvey DJ (March 1985). "Identification of hepatic metabolites of n-heptyl-delta-1-tetrahydrocannabinol in the mouse". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 15 (3): 187–197. doi: 10.3109/00498258509045349. PMID 2992174.
- ^ a b Bueno J, Greenbaum EA (February 2021). "(-)-trans-Δ9-Tetrahydrocannabiphorol Content of Cannabis sativa Inflorescence from Various Chemotypes". Journal of Natural Products. 84 (2): 531–536. doi: 10.1021/acs.jnatprod.0c01034. PMID 33565878. S2CID 231866062.
- ^ Linciano P, Russo F, Citti C, Tolomeo F, Paris R, Fulvio F, et al. (December 2021). "The novel heptyl phorolic acid cannabinoids content in different Cannabis sativa L. accessions". Talanta. 235: 122704. doi: 10.1016/j.talanta.2021.122704. hdl: 11380/1250339. PMID 34517579.
- ^ a b Citti C, Linciano P, Russo F, Luongo L, Iannotta M, Maione S, et al. (December 2019). "A novel phytocannabinoid isolated from Cannabis sativa L. with an in vivo cannabimimetic activity higher than Δ9-tetrahydrocannabinol: Δ9-Tetrahydrocannabiphorol". Scientific Reports. 9 (1): 20335. Bibcode: 2019NatSR...920335C. doi: 10.1038/s41598-019-56785-1. PMC 6937300. PMID 31889124.
- ^ Adams R, Loewe S, Smith CM, McPhee WD (1942). "Tetrahydrocannabinol Homologs and Analogs with Marihuana Activity. XIII1". Journal of the American Chemical Society. 64 (3): 694–697. doi: 10.1021/ja01255a061.
- ^ Adams R, Loewe S, Jelinek C, Wolff H (July 1941). "Tetrahydrocannabinol Homologs with Marihuana Activity. IX". Journal of the American Chemical Society. 63 (7): 1971–1973. doi: 10.1021/ja01852a052.
- ^ Martin BR, Jefferson R, Winckler R, Wiley JL, Huffman JW, Crocker PJ, et al. (September 1999). "Manipulation of the tetrahydrocannabinol side chain delineates agonists, partial agonists, and antagonists". The Journal of Pharmacology and Experimental Therapeutics. 290 (3): 1065–1079. PMID 10454479.
- ^ a b Bow EW, Rimoldi JM (2016). "The Structure-Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation". Perspectives in Medicinal Chemistry. 8: 17–39. doi: 10.4137/PMC.S32171. PMC 4927043. PMID 27398024.
- ^ Hazekamp A, Fischedick JT, Díez ML, Lubbe A, Ruhaak RL (2010). "Chemistry of Cannabis". Comprehensive Natural Products II. pp. 1033–1084. doi: 10.1016/B978-008045382-8.00091-5. ISBN 978-0-08-045382-8.
- ^ Bueno J, Greenbaum EA (2021). "(-)- trans-Δ9-Tetrahydrocannabiphorol Content of Cannabis sativa Inflorescence from Various Chemotypes". Journal of Natural Products. 84 (2): 531–536. doi: 10.1021/acs.jnatprod.0c01034. PMID 33565878. S2CID 231866062. Retrieved 19 Feb 2024.
This article needs additional citations for
verification. (September 2022) |
 | |
 | |
| Clinical data | |
|---|---|
| Other names | (-)-Trans-Δ9-tetrahydrocannabiphorol Δ9-THCP (C7)-Δ9-THC THC-Heptyl |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C23H34O2 |
| Molar mass | 342.523 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Tetrahydrocannabiphorol (THCP) is a potent phytocannabinoid, a CB1 and CB2 agonist which was known as a synthetic homologue of THC, [1] but for the first time in 2019 was isolated as a natural product in trace amounts from Cannabis sativa. [2] [3] It is structurally similar to Δ9- THC, the main active component of cannabis, but with the pentyl side chain extended to heptyl. Since it has a longer side chain, its cannabinoid effects are "far higher than Δ9-THC itself." Tetrahydrocannabiphorol has a reported binding affinity of 1.2 nM at CB1, approximately 33 times that of Δ9-THC (40 nM at CB1). [4]
THCP was studied by Roger Adams as early as 1942. [5]
Isomers
Delta-3-THCP

The Δ3/Δ6a(10a) isomer Δ3-THCP was synthesised in 1941, and was found to have around the same potency as Δ3-THC, unlike the hexyl homologue parahexyl which was significantly stronger. [6]
Delta-8-THCP

The Δ8 isomer is also known as a synthetic cannabinoid under the code name JWH-091. [7] [8] It's unconfirmed whether or not Δ8-THCP is found naturally in cannabis plants, but likely is due to Δ8-THC itself being a degraded form of Δ9-THC. [9] JWH-091 has approximately double the binding affinity at the CB1 receptor (22 nM ± 3.9 nM) in comparison to Δ9-THC (40.7 nM ± 1.7 nM) or Δ8-THC (44 nM ± 12 nM), [8] but appears significantly lower in vitro than the binding activity of Δ9-THCP (Ki = 1.2 nM) [4]
Natural occurrence in cannabis
The Δ9 isomer of THCP occurs naturally in cannabis, but in small amounts. A 2021 study reported the content of Δ9-THCP ranging from 0.0023% to 0.0136% (w/w) (approximately 0.02–0.13 mg/g) without correlation to THC percentage in Δ9-THC-dominant strains of cannabis; that study failed to detect THCP in CBD-dominant strains. [2] [10]
See also
- Nabilone
- Hexahydrocannabiphorol
- 1,2-Didehydro-3-oxo-THCO
- Delta-8-THC
- Parahexyl
- Perrottetinene
- Tetrahydrocannabivarin
- Tetrahydrocannabutol
- Tetrahydrocannabihexol
- THCP-O-acetate
References
- ^ Harvey DJ (March 1985). "Identification of hepatic metabolites of n-heptyl-delta-1-tetrahydrocannabinol in the mouse". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 15 (3): 187–197. doi: 10.3109/00498258509045349. PMID 2992174.
- ^ a b Bueno J, Greenbaum EA (February 2021). "(-)-trans-Δ9-Tetrahydrocannabiphorol Content of Cannabis sativa Inflorescence from Various Chemotypes". Journal of Natural Products. 84 (2): 531–536. doi: 10.1021/acs.jnatprod.0c01034. PMID 33565878. S2CID 231866062.
- ^ Linciano P, Russo F, Citti C, Tolomeo F, Paris R, Fulvio F, et al. (December 2021). "The novel heptyl phorolic acid cannabinoids content in different Cannabis sativa L. accessions". Talanta. 235: 122704. doi: 10.1016/j.talanta.2021.122704. hdl: 11380/1250339. PMID 34517579.
- ^ a b Citti C, Linciano P, Russo F, Luongo L, Iannotta M, Maione S, et al. (December 2019). "A novel phytocannabinoid isolated from Cannabis sativa L. with an in vivo cannabimimetic activity higher than Δ9-tetrahydrocannabinol: Δ9-Tetrahydrocannabiphorol". Scientific Reports. 9 (1): 20335. Bibcode: 2019NatSR...920335C. doi: 10.1038/s41598-019-56785-1. PMC 6937300. PMID 31889124.
- ^ Adams R, Loewe S, Smith CM, McPhee WD (1942). "Tetrahydrocannabinol Homologs and Analogs with Marihuana Activity. XIII1". Journal of the American Chemical Society. 64 (3): 694–697. doi: 10.1021/ja01255a061.
- ^ Adams R, Loewe S, Jelinek C, Wolff H (July 1941). "Tetrahydrocannabinol Homologs with Marihuana Activity. IX". Journal of the American Chemical Society. 63 (7): 1971–1973. doi: 10.1021/ja01852a052.
- ^ Martin BR, Jefferson R, Winckler R, Wiley JL, Huffman JW, Crocker PJ, et al. (September 1999). "Manipulation of the tetrahydrocannabinol side chain delineates agonists, partial agonists, and antagonists". The Journal of Pharmacology and Experimental Therapeutics. 290 (3): 1065–1079. PMID 10454479.
- ^ a b Bow EW, Rimoldi JM (2016). "The Structure-Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation". Perspectives in Medicinal Chemistry. 8: 17–39. doi: 10.4137/PMC.S32171. PMC 4927043. PMID 27398024.
- ^ Hazekamp A, Fischedick JT, Díez ML, Lubbe A, Ruhaak RL (2010). "Chemistry of Cannabis". Comprehensive Natural Products II. pp. 1033–1084. doi: 10.1016/B978-008045382-8.00091-5. ISBN 978-0-08-045382-8.
- ^ Bueno J, Greenbaum EA (2021). "(-)- trans-Δ9-Tetrahydrocannabiphorol Content of Cannabis sativa Inflorescence from Various Chemotypes". Journal of Natural Products. 84 (2): 531–536. doi: 10.1021/acs.jnatprod.0c01034. PMID 33565878. S2CID 231866062. Retrieved 19 Feb 2024.