| |
| Names | |
|---|---|
|
Preferred IUPAC name
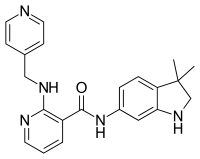
N-(3,3-Dimethyl-2,3-dihydro-1H-indol-6-yl)-2-{[(pyridin-4-yl)methyl]amino}pyridine-3-carboxamide | |
| Other names
AMG 706
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C22H23N5O | |
| Molar mass | 373.460 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Motesanib (AMG 706) is an experimental drug candidate originally developed by Amgen [1] but later investigated by the Takeda Pharmaceutical Company. It is an orally administered small molecule belonging to angiokinase inhibitor class which acts as an antagonist of VEGF receptors, platelet-derived growth factor receptors, and stem cell factor receptors. [2] It is used as the phosphate salt motesanib diphosphate. After clinical trials in thyroid cancer, non-small cell lung cancer, gastrointestinal stromal cancer, colorectal cancer, and breast cancer, the drug was not found to show sufficient efficacy for further development, and development was abandoned by Takeda. [3]
Motesanib was originally investigated for effectiveness against advanced nonsquamous non-small-cell lung cancer (NSCLC), with Phase II trials indicating an effectiveness comparable to bevacizumab when they were both used in combination with paclitaxel/ carboplatin. [4] However a later and more detailed Phase III trial failed to show any benefit for the treatment of NSCLC. [2] [5] A second Phase III trial was started in 2012, [6] which focused on patients from Asian backgrounds (performed on the basis of subgroup analysis) [7] however this also failed to meet its primary endpoint. [8]
The drug has undergone a Phase II evaluation as first-line therapy for breast cancer [2] however this study found no evidence to support further investigation. [9] Phase II testing against persistent or recurrent ovarian, fallopian tube and primary peritoneal carcinomas was also unsuccessful. [10] Two phase II clinical trials for thyroid cancer showed promising results. [11] [12] [13]
-
^ Stafford, edited by Rongshi Li, Jeffrey A. (2009). "Chapter 5. Discovery of Motesanib".
Kinase inhibitor drugs. Hoboken, N.J.: Wiley. pp.
113–130.
doi:
10.1002/9780470524961.ch5.
ISBN
978-0-470-27829-1.
{{ cite book}}:|first1=has generic name ( help)CS1 maint: multiple names: authors list ( link) - ^ a b c "Amgen and Takeda's NSCLC Drug Fails in Phase III Study". 30 Mar 2011.
- ^ "Motesanib". AdisInsight. Retrieved 1 February 2017.
- ^ Blumenschein Jr, G. R.; Kabbinavar, F.; Menon, H.; Mok, T. S. K.; Stephenson, J.; Beck, J. T.; Lakshmaiah, K.; Reckamp, K.; Hei, Y.- J.; Kracht, K.; Sun, Y.- N.; Sikorski, R.; Schwartzberg, L. (14 February 2011). "A phase II, multicenter, open-label randomized study of motesanib or bevacizumab in combination with paclitaxel and carboplatin for advanced nonsquamous non-small-cell lung cancer". Annals of Oncology. 22 (9): 2057–2067. doi: 10.1093/annonc/mdq731. PMID 21321086.
- ^ Scagliotti, G. V.; Vynnychenko, I.; Park, K.; Ichinose, Y.; Kubota, K.; Blackhall, F.; Pirker, R.; Galiulin, R.; Ciuleanu, T.-E.; Sydorenko, O.; Dediu, M.; Papai-Szekely, Z.; Banaclocha, N. M.; McCoy, S.; Yao, B.; Hei, Y.-j.; Galimi, F.; Spigel, D. R. (2 July 2012). "International, Randomized, Placebo-Controlled, Double-Blind Phase III Study of Motesanib Plus Carboplatin/Paclitaxel in Patients With Advanced Nonsquamous Non-Small-Cell Lung Cancer: MONET1" (PDF). Journal of Clinical Oncology. 30 (23): 2829–2836. doi: 10.1200/JCO.2011.41.4987. hdl: 2318/118283. PMID 22753922.
- ^ "Takeda Initiates Phase 3 Trial of Motesanib in Japan and Additional Asian Countries". Takeda Pharmaceutical Company Limited. Retrieved 19 February 2015.
- ^ Kubota, K.; Ichinose, Y.; Scagliotti, G.; Spigel, D.; Kim, J. H.; Shinkai, T.; Takeda, K.; Kim, S.- W.; Hsia, T.- C.; Li, R. K.; Tiangco, B. J.; Yau, S.; Lim, W.- T.; Yao, B.; Hei, Y.- J.; Park, K. (13 January 2014). "Phase III study (MONET1) of motesanib plus carboplatin/paclitaxel in patients with advanced nonsquamous nonsmall-cell lung cancer (NSCLC): Asian subgroup analysis". Annals of Oncology. 25 (2): 529–536. doi: 10.1093/annonc/mdt552. hdl: 2318/150328. PMID 24419239.
- ^ "Takeda Announces Phase 3 MONET-A Study Evaluating Motesanib (AMG 706) in Patients with Advanced Non-Squamous Non-Small Cell Lung Cancer Does Not Meet Primary Endpoint". Takeda Pharmaceutical Company Limited. Retrieved 19 February 2015.
- ^ Martin, Miguel; Roche, Henri; Pinter, Tamas; Crown, John; Kennedy, M John; Provencher, Louise; Priou, Frank; Eiermann, Wolfgang; Adrover, Encarna; Lang, Istvan; Ramos, Manuel; Latreille, Jean; Jagiełło-Gruszfeld, Agnieszka; Pienkowski, Tadeusz; Alba, Emilio; Snyder, Raymond; Almel, Sachin; Rolski, Janusz; Munoz, Montserrat; Moroose, Rebecca; Hurvitz, Sara; Baños, Ana; Adewoye, Henry; Hei, Yong-Jiang; Lindsay, Mary-Ann; Rupin, Matthieu; Cabaribere, David; Lemmerick, Yasmin; Mackey, John R (April 2011). "Motesanib, or open-label bevacizumab, in combination with paclitaxel, as first-line treatment for HER2-negative locally recurrent or metastatic breast cancer: a phase 2, randomised, double-blind, placebo-controlled study". The Lancet Oncology. 12 (4): 369–376. doi: 10.1016/S1470-2045(11)70037-7. PMID 21429799.
- ^ Schilder, R.J.; Sill, M.W.; Lankes, H.A.; Gold, M.A.; Mannel, R.S.; Modesitt, S.C.; Hanjani, P.; Bonebrake, A.J.; Sood, A.K.; Godwin, A.K.; Hu, W.; Alpaugh, R.K. (April 2013). "A phase II evaluation of motesanib (AMG 706) in the treatment of persistent or recurrent ovarian, fallopian tube and primary peritoneal carcinomas: A Gynecologic Oncology Group study". Gynecologic Oncology. 129 (1): 86–91. doi: 10.1016/j.ygyno.2013.01.006. PMC 3712785. PMID 23321064.
- ^ Motesanib Diphosphate Provides Anticancer Activity Among Patients with Progressive Thyroid Cancer, CancerConnect.com
- ^ Schlumberger, M. J.; Elisei, R.; Bastholt, L.; Wirth, L. J.; Martins, R. G.; Locati, L. D.; Jarzab, B.; Pacini, F.; Daumerie, C.; Droz, J.-P.; Eschenberg, M. J.; Sun, Y.-N.; Juan, T.; Stepan, D. E.; Sherman, S. I. (29 June 2009). "Phase II Study of Safety and Efficacy of Motesanib in Patients With Progressive or Symptomatic, Advanced or Metastatic Medullary Thyroid Cancer". Journal of Clinical Oncology. 27 (23): 3794–3801. doi: 10.1200/JCO.2008.18.7815. PMID 19564535.
- ^ Sherman, Steven I.; Wirth, Lori J.; Droz, Jean-Pierre; Hofmann, Michael; Bastholt, Lars; Martins, Renato G.; Licitra, Lisa; Eschenberg, Michael J.; Sun, Yu-Nien; Juan, Todd; Stepan, Daniel E.; Schlumberger, Martin J. (3 July 2008). "Motesanib Diphosphate in Progressive Differentiated Thyroid Cancer". New England Journal of Medicine. 359 (1): 31–42. doi: 10.1056/NEJMoa075853. hdl: 2318/100325. PMID 18596272.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
N-(3,3-Dimethyl-2,3-dihydro-1H-indol-6-yl)-2-{[(pyridin-4-yl)methyl]amino}pyridine-3-carboxamide | |
| Other names
AMG 706
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C22H23N5O | |
| Molar mass | 373.460 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Motesanib (AMG 706) is an experimental drug candidate originally developed by Amgen [1] but later investigated by the Takeda Pharmaceutical Company. It is an orally administered small molecule belonging to angiokinase inhibitor class which acts as an antagonist of VEGF receptors, platelet-derived growth factor receptors, and stem cell factor receptors. [2] It is used as the phosphate salt motesanib diphosphate. After clinical trials in thyroid cancer, non-small cell lung cancer, gastrointestinal stromal cancer, colorectal cancer, and breast cancer, the drug was not found to show sufficient efficacy for further development, and development was abandoned by Takeda. [3]
Motesanib was originally investigated for effectiveness against advanced nonsquamous non-small-cell lung cancer (NSCLC), with Phase II trials indicating an effectiveness comparable to bevacizumab when they were both used in combination with paclitaxel/ carboplatin. [4] However a later and more detailed Phase III trial failed to show any benefit for the treatment of NSCLC. [2] [5] A second Phase III trial was started in 2012, [6] which focused on patients from Asian backgrounds (performed on the basis of subgroup analysis) [7] however this also failed to meet its primary endpoint. [8]
The drug has undergone a Phase II evaluation as first-line therapy for breast cancer [2] however this study found no evidence to support further investigation. [9] Phase II testing against persistent or recurrent ovarian, fallopian tube and primary peritoneal carcinomas was also unsuccessful. [10] Two phase II clinical trials for thyroid cancer showed promising results. [11] [12] [13]
-
^ Stafford, edited by Rongshi Li, Jeffrey A. (2009). "Chapter 5. Discovery of Motesanib".
Kinase inhibitor drugs. Hoboken, N.J.: Wiley. pp.
113–130.
doi:
10.1002/9780470524961.ch5.
ISBN
978-0-470-27829-1.
{{ cite book}}:|first1=has generic name ( help)CS1 maint: multiple names: authors list ( link) - ^ a b c "Amgen and Takeda's NSCLC Drug Fails in Phase III Study". 30 Mar 2011.
- ^ "Motesanib". AdisInsight. Retrieved 1 February 2017.
- ^ Blumenschein Jr, G. R.; Kabbinavar, F.; Menon, H.; Mok, T. S. K.; Stephenson, J.; Beck, J. T.; Lakshmaiah, K.; Reckamp, K.; Hei, Y.- J.; Kracht, K.; Sun, Y.- N.; Sikorski, R.; Schwartzberg, L. (14 February 2011). "A phase II, multicenter, open-label randomized study of motesanib or bevacizumab in combination with paclitaxel and carboplatin for advanced nonsquamous non-small-cell lung cancer". Annals of Oncology. 22 (9): 2057–2067. doi: 10.1093/annonc/mdq731. PMID 21321086.
- ^ Scagliotti, G. V.; Vynnychenko, I.; Park, K.; Ichinose, Y.; Kubota, K.; Blackhall, F.; Pirker, R.; Galiulin, R.; Ciuleanu, T.-E.; Sydorenko, O.; Dediu, M.; Papai-Szekely, Z.; Banaclocha, N. M.; McCoy, S.; Yao, B.; Hei, Y.-j.; Galimi, F.; Spigel, D. R. (2 July 2012). "International, Randomized, Placebo-Controlled, Double-Blind Phase III Study of Motesanib Plus Carboplatin/Paclitaxel in Patients With Advanced Nonsquamous Non-Small-Cell Lung Cancer: MONET1" (PDF). Journal of Clinical Oncology. 30 (23): 2829–2836. doi: 10.1200/JCO.2011.41.4987. hdl: 2318/118283. PMID 22753922.
- ^ "Takeda Initiates Phase 3 Trial of Motesanib in Japan and Additional Asian Countries". Takeda Pharmaceutical Company Limited. Retrieved 19 February 2015.
- ^ Kubota, K.; Ichinose, Y.; Scagliotti, G.; Spigel, D.; Kim, J. H.; Shinkai, T.; Takeda, K.; Kim, S.- W.; Hsia, T.- C.; Li, R. K.; Tiangco, B. J.; Yau, S.; Lim, W.- T.; Yao, B.; Hei, Y.- J.; Park, K. (13 January 2014). "Phase III study (MONET1) of motesanib plus carboplatin/paclitaxel in patients with advanced nonsquamous nonsmall-cell lung cancer (NSCLC): Asian subgroup analysis". Annals of Oncology. 25 (2): 529–536. doi: 10.1093/annonc/mdt552. hdl: 2318/150328. PMID 24419239.
- ^ "Takeda Announces Phase 3 MONET-A Study Evaluating Motesanib (AMG 706) in Patients with Advanced Non-Squamous Non-Small Cell Lung Cancer Does Not Meet Primary Endpoint". Takeda Pharmaceutical Company Limited. Retrieved 19 February 2015.
- ^ Martin, Miguel; Roche, Henri; Pinter, Tamas; Crown, John; Kennedy, M John; Provencher, Louise; Priou, Frank; Eiermann, Wolfgang; Adrover, Encarna; Lang, Istvan; Ramos, Manuel; Latreille, Jean; Jagiełło-Gruszfeld, Agnieszka; Pienkowski, Tadeusz; Alba, Emilio; Snyder, Raymond; Almel, Sachin; Rolski, Janusz; Munoz, Montserrat; Moroose, Rebecca; Hurvitz, Sara; Baños, Ana; Adewoye, Henry; Hei, Yong-Jiang; Lindsay, Mary-Ann; Rupin, Matthieu; Cabaribere, David; Lemmerick, Yasmin; Mackey, John R (April 2011). "Motesanib, or open-label bevacizumab, in combination with paclitaxel, as first-line treatment for HER2-negative locally recurrent or metastatic breast cancer: a phase 2, randomised, double-blind, placebo-controlled study". The Lancet Oncology. 12 (4): 369–376. doi: 10.1016/S1470-2045(11)70037-7. PMID 21429799.
- ^ Schilder, R.J.; Sill, M.W.; Lankes, H.A.; Gold, M.A.; Mannel, R.S.; Modesitt, S.C.; Hanjani, P.; Bonebrake, A.J.; Sood, A.K.; Godwin, A.K.; Hu, W.; Alpaugh, R.K. (April 2013). "A phase II evaluation of motesanib (AMG 706) in the treatment of persistent or recurrent ovarian, fallopian tube and primary peritoneal carcinomas: A Gynecologic Oncology Group study". Gynecologic Oncology. 129 (1): 86–91. doi: 10.1016/j.ygyno.2013.01.006. PMC 3712785. PMID 23321064.
- ^ Motesanib Diphosphate Provides Anticancer Activity Among Patients with Progressive Thyroid Cancer, CancerConnect.com
- ^ Schlumberger, M. J.; Elisei, R.; Bastholt, L.; Wirth, L. J.; Martins, R. G.; Locati, L. D.; Jarzab, B.; Pacini, F.; Daumerie, C.; Droz, J.-P.; Eschenberg, M. J.; Sun, Y.-N.; Juan, T.; Stepan, D. E.; Sherman, S. I. (29 June 2009). "Phase II Study of Safety and Efficacy of Motesanib in Patients With Progressive or Symptomatic, Advanced or Metastatic Medullary Thyroid Cancer". Journal of Clinical Oncology. 27 (23): 3794–3801. doi: 10.1200/JCO.2008.18.7815. PMID 19564535.
- ^ Sherman, Steven I.; Wirth, Lori J.; Droz, Jean-Pierre; Hofmann, Michael; Bastholt, Lars; Martins, Renato G.; Licitra, Lisa; Eschenberg, Michael J.; Sun, Yu-Nien; Juan, Todd; Stepan, Daniel E.; Schlumberger, Martin J. (3 July 2008). "Motesanib Diphosphate in Progressive Differentiated Thyroid Cancer". New England Journal of Medicine. 359 (1): 31–42. doi: 10.1056/NEJMoa075853. hdl: 2318/100325. PMID 18596272.