 | |
| Clinical data | |
|---|---|
| Trade names | Vascor |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a699051 |
|
Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | Well absorbed |
| Protein binding | 99% |
| Metabolism | Hepatic, CYP3A4-mediated |
| Elimination half-life | 42 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
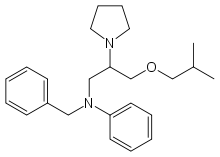
| Formula | C24H34N2O |
| Molar mass | 366.549 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Bepridil (trade name Vascor) is an diamine calcium channel blocker once used to treat angina pectoris. It is no longer sold in the United States.
It is nonselective. [1]
It has been discussed as a possible option in the treatment of atrial fibrillation. [2]
It has been implicated in causing ventricular arrhythmia ( torsades de pointes).
Ebola research
In June 2015 a research paper [3] was published finding bepridil to result in a 100% survival rate for mice exposed to ebola during an experiment searching for potential pharmaceutical ebola treatments; indicating its potential use in future ebola research and therapy. [4]
SARS-CoV-2 research
A research paper [5] showed that Bepridil inhibited cytopathogenic effects induced by SARS-CoV-2 in Vero E6 cells and in A549 cells in an in vitro assay.
References
- ^ Bezprozvanny I, Tsien RW (September 1995). "Voltage-dependent blockade of diverse types of voltage-gated Ca2+ channels expressed in Xenopus oocytes by the Ca2+ channel antagonist mibefradil (Ro 40-5967)". Molecular Pharmacology. 48 (3): 540–549. PMID 7565636.
- ^ Imai S, Saito F, Takase H, Enomoto M, Aoyama H, Yamaji S, et al. (May 2008). "Use of bepridil in combination with Ic antiarrhythmic agent in converting persistent atrial fibrillation to sinus rhythm". Circulation Journal. 72 (5): 709–715. doi: 10.1253/circj.72.709. PMID 18441448.
- ^ Johansen LM, DeWald LE, Shoemaker CJ, Hoffstrom BG, Lear-Rooney CM, Stossel A, et al. (June 2015). "A screen of approved drugs and molecular probes identifies therapeutics with anti-Ebola virus activity". Science Translational Medicine. 7 (290): 290ra89. doi: 10.1126/scitranslmed.aaa5597. PMID 26041706.
- ^ Cha AE (4 June 2015). "Zoloft as Ebola cure? Antidepressant is one of a number of promising drugs being looked at by scientists". Washington Post.
- ^ Vatansever EC, Yang KS, Drelich AK, Kratch KC, Cho CC, Kempaiah KR, et al. (March 2021). "Bepridil is potent against SARS-CoV-2 in vitro". Proceedings of the National Academy of Sciences of the United States of America. 118 (10): e2012201118. Bibcode: 2021PNAS..11812201V. doi: 10.1073/pnas.2012201118. PMC 7958448. PMID 33597253.
External links
 | |
| Clinical data | |
|---|---|
| Trade names | Vascor |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a699051 |
|
Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | Well absorbed |
| Protein binding | 99% |
| Metabolism | Hepatic, CYP3A4-mediated |
| Elimination half-life | 42 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C24H34N2O |
| Molar mass | 366.549 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Bepridil (trade name Vascor) is an diamine calcium channel blocker once used to treat angina pectoris. It is no longer sold in the United States.
It is nonselective. [1]
It has been discussed as a possible option in the treatment of atrial fibrillation. [2]
It has been implicated in causing ventricular arrhythmia ( torsades de pointes).
Ebola research
In June 2015 a research paper [3] was published finding bepridil to result in a 100% survival rate for mice exposed to ebola during an experiment searching for potential pharmaceutical ebola treatments; indicating its potential use in future ebola research and therapy. [4]
SARS-CoV-2 research
A research paper [5] showed that Bepridil inhibited cytopathogenic effects induced by SARS-CoV-2 in Vero E6 cells and in A549 cells in an in vitro assay.
References
- ^ Bezprozvanny I, Tsien RW (September 1995). "Voltage-dependent blockade of diverse types of voltage-gated Ca2+ channels expressed in Xenopus oocytes by the Ca2+ channel antagonist mibefradil (Ro 40-5967)". Molecular Pharmacology. 48 (3): 540–549. PMID 7565636.
- ^ Imai S, Saito F, Takase H, Enomoto M, Aoyama H, Yamaji S, et al. (May 2008). "Use of bepridil in combination with Ic antiarrhythmic agent in converting persistent atrial fibrillation to sinus rhythm". Circulation Journal. 72 (5): 709–715. doi: 10.1253/circj.72.709. PMID 18441448.
- ^ Johansen LM, DeWald LE, Shoemaker CJ, Hoffstrom BG, Lear-Rooney CM, Stossel A, et al. (June 2015). "A screen of approved drugs and molecular probes identifies therapeutics with anti-Ebola virus activity". Science Translational Medicine. 7 (290): 290ra89. doi: 10.1126/scitranslmed.aaa5597. PMID 26041706.
- ^ Cha AE (4 June 2015). "Zoloft as Ebola cure? Antidepressant is one of a number of promising drugs being looked at by scientists". Washington Post.
- ^ Vatansever EC, Yang KS, Drelich AK, Kratch KC, Cho CC, Kempaiah KR, et al. (March 2021). "Bepridil is potent against SARS-CoV-2 in vitro". Proceedings of the National Academy of Sciences of the United States of America. 118 (10): e2012201118. Bibcode: 2021PNAS..11812201V. doi: 10.1073/pnas.2012201118. PMC 7958448. PMID 33597253.