(Redirected from
Alpha-Viniferin)
| |
| Names | |
|---|---|
|
Preferred IUPAC name
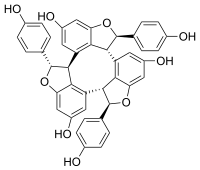
(1R,5bR,6R,10bS,11S,15bR)-1,6,11-Tris(4-hydroxyphenyl)-1,5b,6,10b,11,15b-hexahydrocyclonona[1,2,3-cd:4,5,6-c′d′:7,8,9-c′′d′′]tris([1]benzofuran)-4,9,14-triol | |
| Other names
α-Viniferin; (+)-α-Viniferin
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C42H30O9 | |
| Molar mass | 678.693 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
α-Viniferin is a stilbene trimer. It can be isolated from Caragana chamlagu [1] and from Caragana sinica [2] and from the stem bark of Dryobalanops aromatica. [3] It is also present in relation to resistance to Botrytis cinerea and Plasmopara viticola in Vitis vinifera and Vitis riparia. [4] It has been shown to inhibit acetylcholinesterase. [1]
References
- ^ a b Sung, Sang Hyun; Kang, So Young; Lee, Ki Yong; Park, Mi Jung; Kim, Jeong Hun; Park, Jong Hee; Kim, Young Chul; Kim, Jinwoong; Kim, Young Choong (2002). "(+)-α-Viniferin, a Stilbene Trimer from Caragana chamlague, Inhibits Acetylcholinesterase". Biological & Pharmaceutical Bulletin. 25 (1): 125–127. doi: 10.1248/bpb.25.125. PMID 11824541.
- ^ Shu, N; Zhou, H; Hu, C (2006). "Simultaneous determination of the contents of three stilbene oligomers in Caragana sinica collected in different seasons using an improved HPLC method". Biological & Pharmaceutical Bulletin. 29 (4): 608–12. doi: 10.1248/bpb.29.608. PMID 16595888.
- ^ Wibowo, A.; Ahmat, N.; Hamzah, A.S.; Sufian, A.S.; Ismail, N.H.; Ahmad, R.; Jaafar, F.M.; Takayama, H. (2011). "Malaysianol A, a new trimer resveratrol oligomer from the stem bark of Dryobalanops aromatica". Fitoterapia. 82 (4): 676–81. doi: 10.1016/j.fitote.2011.02.006. PMID 21338657.
- ^ Disease resistance of Vitis spp. and the production of the stress metabolites resveratrol, epsilon -viniferin, alpha -viniferin and pterostilbene. Langcake P, Physiological Plant Pathology, 1981, Vol. 18, No. 2, pages 213-226 ( abstract Archived 2014-12-10 at the Wayback Machine)
(Redirected from
Alpha-Viniferin)

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(1R,5bR,6R,10bS,11S,15bR)-1,6,11-Tris(4-hydroxyphenyl)-1,5b,6,10b,11,15b-hexahydrocyclonona[1,2,3-cd:4,5,6-c′d′:7,8,9-c′′d′′]tris([1]benzofuran)-4,9,14-triol | |
| Other names
α-Viniferin; (+)-α-Viniferin
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C42H30O9 | |
| Molar mass | 678.693 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
α-Viniferin is a stilbene trimer. It can be isolated from Caragana chamlagu [1] and from Caragana sinica [2] and from the stem bark of Dryobalanops aromatica. [3] It is also present in relation to resistance to Botrytis cinerea and Plasmopara viticola in Vitis vinifera and Vitis riparia. [4] It has been shown to inhibit acetylcholinesterase. [1]
References
- ^ a b Sung, Sang Hyun; Kang, So Young; Lee, Ki Yong; Park, Mi Jung; Kim, Jeong Hun; Park, Jong Hee; Kim, Young Chul; Kim, Jinwoong; Kim, Young Choong (2002). "(+)-α-Viniferin, a Stilbene Trimer from Caragana chamlague, Inhibits Acetylcholinesterase". Biological & Pharmaceutical Bulletin. 25 (1): 125–127. doi: 10.1248/bpb.25.125. PMID 11824541.
- ^ Shu, N; Zhou, H; Hu, C (2006). "Simultaneous determination of the contents of three stilbene oligomers in Caragana sinica collected in different seasons using an improved HPLC method". Biological & Pharmaceutical Bulletin. 29 (4): 608–12. doi: 10.1248/bpb.29.608. PMID 16595888.
- ^ Wibowo, A.; Ahmat, N.; Hamzah, A.S.; Sufian, A.S.; Ismail, N.H.; Ahmad, R.; Jaafar, F.M.; Takayama, H. (2011). "Malaysianol A, a new trimer resveratrol oligomer from the stem bark of Dryobalanops aromatica". Fitoterapia. 82 (4): 676–81. doi: 10.1016/j.fitote.2011.02.006. PMID 21338657.
- ^ Disease resistance of Vitis spp. and the production of the stress metabolites resveratrol, epsilon -viniferin, alpha -viniferin and pterostilbene. Langcake P, Physiological Plant Pathology, 1981, Vol. 18, No. 2, pages 213-226 ( abstract Archived 2014-12-10 at the Wayback Machine)