 | |
| Clinical data | |
|---|---|
|
Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
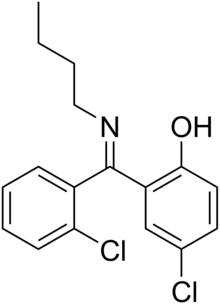
| Formula | C17H17Cl2NO |
| Molar mass | 322.23 g·mol−1 |
Fengabine (SL-79,229) is a drug which was investigated as an antidepressant but was never marketed. [1] [2] Its mechanism of action is unknown, but its antidepressant effects are reversed by GABAA receptor antagonists like bicuculline and it has hence been labeled as GABAergic; however, it does not actually bind to GABA receptors, nor does it inhibit GABA-T. [1] [2] In clinical trials, fengabine's efficacy was comparable to that of the tricyclic antidepressants, but with a more rapid onset of action and much less side effects. [3] [4] [5] Notably, fengabine lacks any sedative effects. [4]
See also
References
- ^ a b Lloyd KG, Zivkovic B, Sanger D, Depoortere H, Bartholini G (April 1987). "Fengabine, a novel antidepressant GABAergic agent. I. Activity in models for antidepressant drugs and psychopharmacological profile". The Journal of Pharmacology and Experimental Therapeutics. 241 (1): 245–50. PMID 3033203.
- ^ a b Scatton B, Lloyd KG, Zivkovic B, et al. (April 1987). "Fengabine, a novel antidepressant GABAergic agent. II. Effect on cerebral noradrenergic, serotonergic and GABAergic transmission in the rat". The Journal of Pharmacology and Experimental Therapeutics. 241 (1): 251–7. PMID 3033204.
- ^ Magni G, Garreau M, Orofiamma B, Palminteri R (1989). "Fengabine, a new GABAmimetic agent in the treatment of depressive disorders: an overview of six double-blind studies versus tricyclics". Neuropsychobiology. 20 (3): 126–31. doi: 10.1159/000118485. PMID 2668780.
- ^ a b Nielsen NP, Cesana B, Zizolfi S, Ascalone V, Priore P, Morselli PL (November 1990). "Therapeutic effects of fengabine, a new GABAergic agent, in depressed outpatients: a double-blind study versus clomipramine". Acta Psychiatrica Scandinavica. 82 (5): 366–71. doi: 10.1111/j.1600-0447.1990.tb01402.x. PMID 2281807. S2CID 44534975.
- ^ Fairweather DB, Kerr JS, Hilton S, Hindmarch I (March 1993). "A placebo controlled double-blind evaluation of the pharmacodynamics of fengabine vs amitriptyline following single and multiple doses in elderly volunteers". British Journal of Clinical Pharmacology. 35 (3): 278–83. doi: 10.1111/j.1365-2125.1993.tb05695.x. PMC 1381575. PMID 8471403.
 | |
| Clinical data | |
|---|---|
|
Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C17H17Cl2NO |
| Molar mass | 322.23 g·mol−1 |
Fengabine (SL-79,229) is a drug which was investigated as an antidepressant but was never marketed. [1] [2] Its mechanism of action is unknown, but its antidepressant effects are reversed by GABAA receptor antagonists like bicuculline and it has hence been labeled as GABAergic; however, it does not actually bind to GABA receptors, nor does it inhibit GABA-T. [1] [2] In clinical trials, fengabine's efficacy was comparable to that of the tricyclic antidepressants, but with a more rapid onset of action and much less side effects. [3] [4] [5] Notably, fengabine lacks any sedative effects. [4]
See also
References
- ^ a b Lloyd KG, Zivkovic B, Sanger D, Depoortere H, Bartholini G (April 1987). "Fengabine, a novel antidepressant GABAergic agent. I. Activity in models for antidepressant drugs and psychopharmacological profile". The Journal of Pharmacology and Experimental Therapeutics. 241 (1): 245–50. PMID 3033203.
- ^ a b Scatton B, Lloyd KG, Zivkovic B, et al. (April 1987). "Fengabine, a novel antidepressant GABAergic agent. II. Effect on cerebral noradrenergic, serotonergic and GABAergic transmission in the rat". The Journal of Pharmacology and Experimental Therapeutics. 241 (1): 251–7. PMID 3033204.
- ^ Magni G, Garreau M, Orofiamma B, Palminteri R (1989). "Fengabine, a new GABAmimetic agent in the treatment of depressive disorders: an overview of six double-blind studies versus tricyclics". Neuropsychobiology. 20 (3): 126–31. doi: 10.1159/000118485. PMID 2668780.
- ^ a b Nielsen NP, Cesana B, Zizolfi S, Ascalone V, Priore P, Morselli PL (November 1990). "Therapeutic effects of fengabine, a new GABAergic agent, in depressed outpatients: a double-blind study versus clomipramine". Acta Psychiatrica Scandinavica. 82 (5): 366–71. doi: 10.1111/j.1600-0447.1990.tb01402.x. PMID 2281807. S2CID 44534975.
- ^ Fairweather DB, Kerr JS, Hilton S, Hindmarch I (March 1993). "A placebo controlled double-blind evaluation of the pharmacodynamics of fengabine vs amitriptyline following single and multiple doses in elderly volunteers". British Journal of Clinical Pharmacology. 35 (3): 278–83. doi: 10.1111/j.1365-2125.1993.tb05695.x. PMC 1381575. PMID 8471403.