 | |
 | |
| Combination of | |
|---|---|
| Estradiol valerate | Estrogen |
| Cyproterone acetate | Progestogen |
| Clinical data | |
| Trade names | Climen, Femilar |
| Other names | EV/CPA |
|
Routes of administration | By mouth |
| Drug class | Estrogen; Progestogen |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
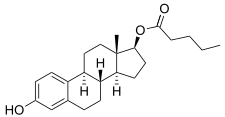
Estradiol valerate/cyproterone acetate (EV/CPA), sold under the brand names Climen and Femilar among others, is a combination product of estradiol valerate (EV), an estrogen, and cyproterone acetate (CPA), a progestogen, which is used in menopausal hormone therapy and as a birth control pill to prevent pregnancy. [1] It is taken by mouth. [1] Climen, which is used in menopausal hormone therapy, is a sequential preparation that contains 2 mg estradiol valerate and 1 mg CPA. [2] [3] [4] It was the first product for use in menopausal hormone therapy containing CPA to be marketed [3] and is available in more than 40 countries. [2] Femilar, which is an estradiol-containing birth control pill, contains 1 to 2 mg estradiol valerate and 1 to 2 mg CPA, and has been approved for use in Finland since 1993. [5]
CPA alone has been found to suppress ovulation in 3 of 5 women at a dose of 0.5 mg/day and in 5 of 5 women at a dose of 1 mg/day. [6] [7] [8] [9] [10] [11] Ovulation inhibition with Femilar occurred in 94.4% of 108 women during the third treatment cycle in one study and in almost 100% of 26 women over 12 treatment cycles in another study (except for one woman who ovulated during her first treatment cycle). [5] [12] [1]
See also
References
- ^ a b c Hirvonen E, Stenman UH, Mälkönen M, Rasi V, Vartiainen E, Ylöstalo P (October 1988). "New natural oestradiol/cyproterone acetate oral contraceptive for pre-menopausal women". Maturitas. 10 (3): 201–213. doi: 10.1016/0378-5122(88)90023-0. PMID 2972897.
- ^ a b Husmann F (1997). "Clinical Experiences with a Combination of Estradiol Valerate and Cyproterone Acetate for Hormone Replacement". Women's Health and Menopause. Medical Science Symposia Series. Vol. 11. Springer. pp. 257–261. doi: 10.1007/978-94-011-5560-1_38. ISBN 978-94-010-6343-2. ISSN 0928-9550.
- ^ a b Schneider HP (November 2003). "Androgens and antiandrogens". Annals of the New York Academy of Sciences. 997 (1): 292–306. Bibcode: 2003NYASA.997..292S. doi: 10.1196/annals.1290.033. PMID 14644837. S2CID 8400556.
- ^ Schneider HP (December 2000). "The role of antiandrogens in hormone replacement therapy". Climacteric. 3 (Suppl 2): 21–27. PMID 11379383.
- ^ a b Fruzzetti F, Trémollieres F, Bitzer J (May 2012). "An overview of the development of combined oral contraceptives containing estradiol: focus on estradiol valerate/dienogest". Gynecological Endocrinology. 28 (5): 400–408. doi: 10.3109/09513590.2012.662547. PMC 3399636. PMID 22468839.
- ^ Endrikat J, Gerlinger C, Richard S, Rosenbaum P, Düsterberg B (December 2011). "Ovulation inhibition doses of progestins: a systematic review of the available literature and of marketed preparations worldwide". Contraception. 84 (6): 549–557. doi: 10.1016/j.contraception.2011.04.009. PMID 22078182.
- ^ Spona J, Schneider WH, Bieglmayer C, Schroeder R, Pirker R (1979). "Ovulation inhibition with different doses of levonorgestrel and other progestogens: clinical and experimental investigations". Acta Obstetricia et Gynecologica Scandinavica. Supplement. 88: 7–15. doi: 10.3109/00016347909157223. PMID 393050. S2CID 30486799.
- ^ Spona J, Huber J, Schmidt JB (July 1986). "[Inhibition of ovulation with 35 micrograms of ethinyl estradiol and 2 mg of cyproterone acetate (Diane 35)]" [Inhibition of ovulation with 35 micrograms of ethinyl estradiol and 2 mg of cyproterone acetate (Diane 35)]. Geburtshilfe und Frauenheilkunde (in German). 46 (7): 435–438. doi: 10.1055/s-2008-1026659. PMID 3093307. S2CID 260156932.
- ^ Spona J, Huber J (1987). "Efficacy of low-dose oral contraceptives containing levonorgestrel, gestoden and cyproterone acetate". Gynecologic and Obstetric Investigation. 23 (3): 184–193. doi: 10.1159/000298860. PMID 2954886.
- ^ Spona J, Huber J, Schmidt JB (1987-12-31). "Ovulation inhibitory effect of SH B 209 AE (Diane-35)—a new antiandrogen-estrogen combination". In Schindler AE (ed.). Antiandrogen-Estrogen Therapy for Signs of Androgenization. Berlin, Boston: De Gruyter. pp. 51–58. doi: 10.1515/9783110866902-006. ISBN 9783110866902.
- ^ Hammerstein J (1990). "Antiandrogens: Clinical Aspects". Hair and Hair Diseases. Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 827–886. doi: 10.1007/978-3-642-74612-3_35. ISBN 978-3-642-74614-7.
- ^ Hirvonen E, Allonen H, Anttila M, Kulmala Y, Ranta T, Rautiainen H, et al. (January 1995). "Oral contraceptive containing natural estradiol for premenopausal women". Maturitas. 21 (1): 27–32. doi: 10.1016/0378-5122(94)00856-3. PMID 7731379.
 | |
 | |
| Combination of | |
|---|---|
| Estradiol valerate | Estrogen |
| Cyproterone acetate | Progestogen |
| Clinical data | |
| Trade names | Climen, Femilar |
| Other names | EV/CPA |
|
Routes of administration | By mouth |
| Drug class | Estrogen; Progestogen |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
Estradiol valerate/cyproterone acetate (EV/CPA), sold under the brand names Climen and Femilar among others, is a combination product of estradiol valerate (EV), an estrogen, and cyproterone acetate (CPA), a progestogen, which is used in menopausal hormone therapy and as a birth control pill to prevent pregnancy. [1] It is taken by mouth. [1] Climen, which is used in menopausal hormone therapy, is a sequential preparation that contains 2 mg estradiol valerate and 1 mg CPA. [2] [3] [4] It was the first product for use in menopausal hormone therapy containing CPA to be marketed [3] and is available in more than 40 countries. [2] Femilar, which is an estradiol-containing birth control pill, contains 1 to 2 mg estradiol valerate and 1 to 2 mg CPA, and has been approved for use in Finland since 1993. [5]
CPA alone has been found to suppress ovulation in 3 of 5 women at a dose of 0.5 mg/day and in 5 of 5 women at a dose of 1 mg/day. [6] [7] [8] [9] [10] [11] Ovulation inhibition with Femilar occurred in 94.4% of 108 women during the third treatment cycle in one study and in almost 100% of 26 women over 12 treatment cycles in another study (except for one woman who ovulated during her first treatment cycle). [5] [12] [1]
See also
References
- ^ a b c Hirvonen E, Stenman UH, Mälkönen M, Rasi V, Vartiainen E, Ylöstalo P (October 1988). "New natural oestradiol/cyproterone acetate oral contraceptive for pre-menopausal women". Maturitas. 10 (3): 201–213. doi: 10.1016/0378-5122(88)90023-0. PMID 2972897.
- ^ a b Husmann F (1997). "Clinical Experiences with a Combination of Estradiol Valerate and Cyproterone Acetate for Hormone Replacement". Women's Health and Menopause. Medical Science Symposia Series. Vol. 11. Springer. pp. 257–261. doi: 10.1007/978-94-011-5560-1_38. ISBN 978-94-010-6343-2. ISSN 0928-9550.
- ^ a b Schneider HP (November 2003). "Androgens and antiandrogens". Annals of the New York Academy of Sciences. 997 (1): 292–306. Bibcode: 2003NYASA.997..292S. doi: 10.1196/annals.1290.033. PMID 14644837. S2CID 8400556.
- ^ Schneider HP (December 2000). "The role of antiandrogens in hormone replacement therapy". Climacteric. 3 (Suppl 2): 21–27. PMID 11379383.
- ^ a b Fruzzetti F, Trémollieres F, Bitzer J (May 2012). "An overview of the development of combined oral contraceptives containing estradiol: focus on estradiol valerate/dienogest". Gynecological Endocrinology. 28 (5): 400–408. doi: 10.3109/09513590.2012.662547. PMC 3399636. PMID 22468839.
- ^ Endrikat J, Gerlinger C, Richard S, Rosenbaum P, Düsterberg B (December 2011). "Ovulation inhibition doses of progestins: a systematic review of the available literature and of marketed preparations worldwide". Contraception. 84 (6): 549–557. doi: 10.1016/j.contraception.2011.04.009. PMID 22078182.
- ^ Spona J, Schneider WH, Bieglmayer C, Schroeder R, Pirker R (1979). "Ovulation inhibition with different doses of levonorgestrel and other progestogens: clinical and experimental investigations". Acta Obstetricia et Gynecologica Scandinavica. Supplement. 88: 7–15. doi: 10.3109/00016347909157223. PMID 393050. S2CID 30486799.
- ^ Spona J, Huber J, Schmidt JB (July 1986). "[Inhibition of ovulation with 35 micrograms of ethinyl estradiol and 2 mg of cyproterone acetate (Diane 35)]" [Inhibition of ovulation with 35 micrograms of ethinyl estradiol and 2 mg of cyproterone acetate (Diane 35)]. Geburtshilfe und Frauenheilkunde (in German). 46 (7): 435–438. doi: 10.1055/s-2008-1026659. PMID 3093307. S2CID 260156932.
- ^ Spona J, Huber J (1987). "Efficacy of low-dose oral contraceptives containing levonorgestrel, gestoden and cyproterone acetate". Gynecologic and Obstetric Investigation. 23 (3): 184–193. doi: 10.1159/000298860. PMID 2954886.
- ^ Spona J, Huber J, Schmidt JB (1987-12-31). "Ovulation inhibitory effect of SH B 209 AE (Diane-35)—a new antiandrogen-estrogen combination". In Schindler AE (ed.). Antiandrogen-Estrogen Therapy for Signs of Androgenization. Berlin, Boston: De Gruyter. pp. 51–58. doi: 10.1515/9783110866902-006. ISBN 9783110866902.
- ^ Hammerstein J (1990). "Antiandrogens: Clinical Aspects". Hair and Hair Diseases. Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 827–886. doi: 10.1007/978-3-642-74612-3_35. ISBN 978-3-642-74614-7.
- ^ Hirvonen E, Allonen H, Anttila M, Kulmala Y, Ranta T, Rautiainen H, et al. (January 1995). "Oral contraceptive containing natural estradiol for premenopausal women". Maturitas. 21 (1): 27–32. doi: 10.1016/0378-5122(94)00856-3. PMID 7731379.