| |
| Names | |
|---|---|
|
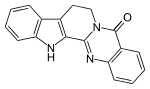
Preferred IUPAC name
8,13-Hydroindolo[2′,3′:3,4]pyrido[2,1-b]quinazolin-5(7H)-one | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.163.752 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C18H13N3O | |
| Molar mass | 287.322 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Rutecarpine or rutaecarpine is a COX-2 inhibitor isolated from Tetradium ruticarpum, a tree native to China. [1] It is classified as a non-basic alkaloid. [2]
In contrast to synthetic COX-2 inhibitors like etoricoxib and celecoxib, rutecarpine does not appear to cause negative effects on the cardiovascular system. [3]
Metabolism
Microsome studies suggest that rutaecarpine may be at least a weak inhibitor of CYP1A2, CYP2C9, CYP2C19, CYP2E1, and CYP3A4 enzymes. [4] [5] At the same time, it is believed to be a strong inducer of CYP1A2 and CYP1A1. [6]
Rutecarpine metabolism is complex and proceeds along several routes, primarily involving the addition of a single hydroxyl group by CYP3A4. Six monohydroxylated and four dihydroxylated metabolites have been identified. To a much lesser extent, rutecarpine may be metabolized by CYP2C9 and CYP1A2, according to liver microsome studies. [7]
Supplementation
Rutecarpine has been shown to decrease the overall bioavailability of caffeine in rats by up to 80 percent, [8] likely through induction of enzymes CYP1A2 and CYP2E1. [9]
References
- ^ Moon, T. C.; Murakami, M.; Kudo, I.; Son, K. H.; Kim, H. P.; Kang, S. S.; Chang, H. W. (1999). "A new class of COX-2 inhibitor, rutaecarpine from Evodia rutaecarpa". Inflammation Research. 48 (12): 621–625. doi: 10.1007/s000110050512. PMID 10669112. S2CID 19555209.
- ^ Manske, R. H. F. (1950). "Sources of alkaloids and their isolation". In Manske, R. H. F.; Holmes, H. L. (eds.). The Alkaloids: Chemistry and Physiology. Vol. 1. Academic Press. pp. 1–14. doi: 10.1016/S1876-0813(08)60184-0. ISBN 978-0-12-469501-6. S2CID 82529003.
- ^ Jia, Sujie; Hu, Changping (2010). "Pharmacological effects of rutaecarpine as a cardiovascular protective agent". Molecules. 15 (3): 1873–1881. doi: 10.3390/molecules15031873. PMC 6257227. PMID 20336017. S2CID 21968872.
- ^ Zhang, Fang-Liang; He, Xin; Zhai, Yi-Ran; He, Li-Na; Zhang, Si-Chao; Wang, Li-Li; Yang, Ai-Hong; An, Li-Jun (2 November 2015). "Mechanism-based inhibition of CYPs and RMs-induced hepatoxicity by rutaecarpine". Xenobiotica. 45 (11): 978–989. doi: 10.3109/00498254.2015.1038742. PMID 26053557. S2CID 6293291.
- ^ Ueng, Yune-Fang; Jan, Woan-Ching; Lin, Lie-Chwen; Chen, Ta-Liang; Guengerich, F. Peter; Chen, Chieh-Fu (1 March 2002). "The Alkaloid Rutaecarpine Is a Selective Inhibitor of Cytochrome P450 1A in Mouse and Human Liver Microsomes". Drug Metabolism and Disposition. 30 (3): 349–353. doi: 10.1124/dmd.30.3.349. PMID 11854157.
- ^ Ueng, Yune-Fang; Wang, Jong-Jing; Lin, Lie-Chwen; Park, Sang Shin; Chen, Chieh-Fu (November 2001). "Induction of cytochrome P450-dependent monooxygenase in mouse liver and kidney by rutaecarpine, an alkaloid of the herbal drug Evodia rutaecarpa". Life Sciences. 70 (2): 207–217. doi: 10.1016/S0024-3205(01)01390-X. PMID 11787945.
- ^ Lee, Seung; Son, Jong-Keun; Jeong, Byeong; Jeong, Tae-Cheon; Chang, Hyeon; Lee, Eung-Seok; Jahng, Yurngdong (6 February 2008). "Progress in the Studies on Rutaecarpine". Molecules. 13 (2): 272–300. doi: 10.3390/molecules13020272. PMC 6245441. PMID 18305418.
- ^ Estari, Rohit Kumar; Dong, Jin; Chan, William K.; Park, Miki Susanto; Zhou, Zhu (1 December 2021). "Time effect of rutaecarpine on caffeine pharmacokinetics in rats". Biochemistry and Biophysics Reports. 28: 101121. doi: 10.1016/j.bbrep.2021.101121. PMC 8429912. PMID 34527815.
- ^ Noh, Keumhan; Seo, Young Min; Lee, Sang Kyu; Bista, Sudeep R.; Kang, Mi Jeong; Jahng, Yurngdong; Kim, Eunyoung; Kang, Wonku; Jeong, Tae Cheon (January 2011). "Effects of rutaecarpine on the metabolism and urinary excretion of caffeine in rats". Archives of Pharmacal Research. 34 (1): 119–125. doi: 10.1007/s12272-011-0114-3. PMID 21468923. S2CID 44752343.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
8,13-Hydroindolo[2′,3′:3,4]pyrido[2,1-b]quinazolin-5(7H)-one | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.163.752 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C18H13N3O | |
| Molar mass | 287.322 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Rutecarpine or rutaecarpine is a COX-2 inhibitor isolated from Tetradium ruticarpum, a tree native to China. [1] It is classified as a non-basic alkaloid. [2]
In contrast to synthetic COX-2 inhibitors like etoricoxib and celecoxib, rutecarpine does not appear to cause negative effects on the cardiovascular system. [3]
Metabolism
Microsome studies suggest that rutaecarpine may be at least a weak inhibitor of CYP1A2, CYP2C9, CYP2C19, CYP2E1, and CYP3A4 enzymes. [4] [5] At the same time, it is believed to be a strong inducer of CYP1A2 and CYP1A1. [6]
Rutecarpine metabolism is complex and proceeds along several routes, primarily involving the addition of a single hydroxyl group by CYP3A4. Six monohydroxylated and four dihydroxylated metabolites have been identified. To a much lesser extent, rutecarpine may be metabolized by CYP2C9 and CYP1A2, according to liver microsome studies. [7]
Supplementation
Rutecarpine has been shown to decrease the overall bioavailability of caffeine in rats by up to 80 percent, [8] likely through induction of enzymes CYP1A2 and CYP2E1. [9]
References
- ^ Moon, T. C.; Murakami, M.; Kudo, I.; Son, K. H.; Kim, H. P.; Kang, S. S.; Chang, H. W. (1999). "A new class of COX-2 inhibitor, rutaecarpine from Evodia rutaecarpa". Inflammation Research. 48 (12): 621–625. doi: 10.1007/s000110050512. PMID 10669112. S2CID 19555209.
- ^ Manske, R. H. F. (1950). "Sources of alkaloids and their isolation". In Manske, R. H. F.; Holmes, H. L. (eds.). The Alkaloids: Chemistry and Physiology. Vol. 1. Academic Press. pp. 1–14. doi: 10.1016/S1876-0813(08)60184-0. ISBN 978-0-12-469501-6. S2CID 82529003.
- ^ Jia, Sujie; Hu, Changping (2010). "Pharmacological effects of rutaecarpine as a cardiovascular protective agent". Molecules. 15 (3): 1873–1881. doi: 10.3390/molecules15031873. PMC 6257227. PMID 20336017. S2CID 21968872.
- ^ Zhang, Fang-Liang; He, Xin; Zhai, Yi-Ran; He, Li-Na; Zhang, Si-Chao; Wang, Li-Li; Yang, Ai-Hong; An, Li-Jun (2 November 2015). "Mechanism-based inhibition of CYPs and RMs-induced hepatoxicity by rutaecarpine". Xenobiotica. 45 (11): 978–989. doi: 10.3109/00498254.2015.1038742. PMID 26053557. S2CID 6293291.
- ^ Ueng, Yune-Fang; Jan, Woan-Ching; Lin, Lie-Chwen; Chen, Ta-Liang; Guengerich, F. Peter; Chen, Chieh-Fu (1 March 2002). "The Alkaloid Rutaecarpine Is a Selective Inhibitor of Cytochrome P450 1A in Mouse and Human Liver Microsomes". Drug Metabolism and Disposition. 30 (3): 349–353. doi: 10.1124/dmd.30.3.349. PMID 11854157.
- ^ Ueng, Yune-Fang; Wang, Jong-Jing; Lin, Lie-Chwen; Park, Sang Shin; Chen, Chieh-Fu (November 2001). "Induction of cytochrome P450-dependent monooxygenase in mouse liver and kidney by rutaecarpine, an alkaloid of the herbal drug Evodia rutaecarpa". Life Sciences. 70 (2): 207–217. doi: 10.1016/S0024-3205(01)01390-X. PMID 11787945.
- ^ Lee, Seung; Son, Jong-Keun; Jeong, Byeong; Jeong, Tae-Cheon; Chang, Hyeon; Lee, Eung-Seok; Jahng, Yurngdong (6 February 2008). "Progress in the Studies on Rutaecarpine". Molecules. 13 (2): 272–300. doi: 10.3390/molecules13020272. PMC 6245441. PMID 18305418.
- ^ Estari, Rohit Kumar; Dong, Jin; Chan, William K.; Park, Miki Susanto; Zhou, Zhu (1 December 2021). "Time effect of rutaecarpine on caffeine pharmacokinetics in rats". Biochemistry and Biophysics Reports. 28: 101121. doi: 10.1016/j.bbrep.2021.101121. PMC 8429912. PMID 34527815.
- ^ Noh, Keumhan; Seo, Young Min; Lee, Sang Kyu; Bista, Sudeep R.; Kang, Mi Jeong; Jahng, Yurngdong; Kim, Eunyoung; Kang, Wonku; Jeong, Tae Cheon (January 2011). "Effects of rutaecarpine on the metabolism and urinary excretion of caffeine in rats". Archives of Pharmacal Research. 34 (1): 119–125. doi: 10.1007/s12272-011-0114-3. PMID 21468923. S2CID 44752343.