 | |
| Clinical data | |
|---|---|
| AHFS/ Drugs.com | International Drug Names |
| License data |
|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.051.024 |
| Chemical and physical data | |
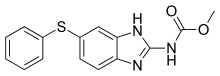
| Formula | C15H13N3O2S |
| Molar mass | 299.35 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Fenbendazole (also known as Fenben) is a broad spectrum benzimidazole anthelmintic used against gastrointestinal parasites including: giardia, roundworms, hookworms, whipworms, the tapeworm genus Taenia (but not effective against Dipylidium caninum, a common dog tapeworm), pinworms, aelurostrongylus, paragonimiasis, strongyles, and strongyloides that can be administered to sheep, cattle, horses, fish, dogs, cats, rabbits, most reptiles, freshwater shrimp tanks as planaria and hydra treatments, as well as seals. [1]
Drug interactions
Drug interactions may occur if salicylanilides such as dibromsalan and niclosamide are co-administered. Abortions in cattle and death in sheep have been reported after using these medications together. [2] Abortions in domestic ruminants have been associated with concurrent use of anti-trematode therapeutic agents. [3]
Toxicity
Fenbendazole is poorly absorbed from the gastrointestinal tract in most species. The LD50 in laboratory animals exceeds 10 g/kg when administered orally. [2] Given its low toxicity and high tolerability among different species, the possibility of repurposing it as a drug to treat cancer in humans has been proposed. [4]
Metabolism
Fenbendazole is metabolized in the liver to oxfendazole, which is anthelmintic too; oxfendazole partially gets reduced back to fenbendazole in the liver and rumen. [5] [6] Also, fenbendazole itself is an active metabolite of another anthelmintic drug, febantel. [7]
See also
References
- ^ "Fenbendazole: From MD Anderson to Joe Tippens". Institute of Integrative BioOncology. Retrieved 2023-10-19.
- ^ a b Plumb DC (2005). Plumb's veterinary drug handbook (Fifth ed.). Stockholm, Wis.: PhrmaVet. ISBN 978-0-8138-0518-4.
- ^ Sarangi LN, Tharani N, Polapally S, Rana SK, Thodangala N, Bahekar VS, et al. (March 2021). "Infectious bovine abortions: observations from an organized dairy herd". Brazilian Journal of Microbiology. 52 (1): 439–448. doi: 10.1007/s42770-020-00414-x. PMC 7966683. PMID 33415719.
-
^ Dogra N, Kumar A, Mukhopadhyay T (August 2018).
"Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death by modulating multiple cellular pathways". Scientific Reports. 8 (1): 11926.
Bibcode:
2018NatSR...811926D.
doi:
10.1038/s41598-018-30158-6.
PMC
6085345.
PMID
30093705.
Fenbendazole is known to have a high safety margin and most species tolerate it very well. It has very low degree of toxicity and high degree of safety in experimental animals. In this study, we show that fenbendazole (FZ) exhibits a moderate microtubule depolymerizing activity towards human cancer cells, but possesses a potent antitumor effect as evident from in vitro and in vivo experiments.
- ^ Junquera P (2015-07-26). "Fenbendazole, Anthelmintic for Veterinary Use on Cattle, Sheep, Goats, Pig, Poultry, Horses, Dogs and Cats Against Roundworms and Tapeworms". PARASITIPEDIA. Retrieved 2015-09-08.
- ^ Junquera P (2015-07-26). "Oxfendazole, Anthelmintic for Veterinary Use on Cattle, Sheep, Goats, Horses, Dogs and Cats Against Roundworms and Tapeworms". PARASITIPEDIA. Retrieved 2015-09-08.
- ^ Junquera P (2015-07-26). "Febantel for Veterinary Use on Dogs, Cats, Cattle, Sheep, Goats, Pig and Poultry Against Roundworms and Tapeworms". PARASITIPEDIA. Retrieved 2015-09-08.
 | |
| Clinical data | |
|---|---|
| AHFS/ Drugs.com | International Drug Names |
| License data |
|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.051.024 |
| Chemical and physical data | |
| Formula | C15H13N3O2S |
| Molar mass | 299.35 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Fenbendazole (also known as Fenben) is a broad spectrum benzimidazole anthelmintic used against gastrointestinal parasites including: giardia, roundworms, hookworms, whipworms, the tapeworm genus Taenia (but not effective against Dipylidium caninum, a common dog tapeworm), pinworms, aelurostrongylus, paragonimiasis, strongyles, and strongyloides that can be administered to sheep, cattle, horses, fish, dogs, cats, rabbits, most reptiles, freshwater shrimp tanks as planaria and hydra treatments, as well as seals. [1]
Drug interactions
Drug interactions may occur if salicylanilides such as dibromsalan and niclosamide are co-administered. Abortions in cattle and death in sheep have been reported after using these medications together. [2] Abortions in domestic ruminants have been associated with concurrent use of anti-trematode therapeutic agents. [3]
Toxicity
Fenbendazole is poorly absorbed from the gastrointestinal tract in most species. The LD50 in laboratory animals exceeds 10 g/kg when administered orally. [2] Given its low toxicity and high tolerability among different species, the possibility of repurposing it as a drug to treat cancer in humans has been proposed. [4]
Metabolism
Fenbendazole is metabolized in the liver to oxfendazole, which is anthelmintic too; oxfendazole partially gets reduced back to fenbendazole in the liver and rumen. [5] [6] Also, fenbendazole itself is an active metabolite of another anthelmintic drug, febantel. [7]
See also
References
- ^ "Fenbendazole: From MD Anderson to Joe Tippens". Institute of Integrative BioOncology. Retrieved 2023-10-19.
- ^ a b Plumb DC (2005). Plumb's veterinary drug handbook (Fifth ed.). Stockholm, Wis.: PhrmaVet. ISBN 978-0-8138-0518-4.
- ^ Sarangi LN, Tharani N, Polapally S, Rana SK, Thodangala N, Bahekar VS, et al. (March 2021). "Infectious bovine abortions: observations from an organized dairy herd". Brazilian Journal of Microbiology. 52 (1): 439–448. doi: 10.1007/s42770-020-00414-x. PMC 7966683. PMID 33415719.
-
^ Dogra N, Kumar A, Mukhopadhyay T (August 2018).
"Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death by modulating multiple cellular pathways". Scientific Reports. 8 (1): 11926.
Bibcode:
2018NatSR...811926D.
doi:
10.1038/s41598-018-30158-6.
PMC
6085345.
PMID
30093705.
Fenbendazole is known to have a high safety margin and most species tolerate it very well. It has very low degree of toxicity and high degree of safety in experimental animals. In this study, we show that fenbendazole (FZ) exhibits a moderate microtubule depolymerizing activity towards human cancer cells, but possesses a potent antitumor effect as evident from in vitro and in vivo experiments.
- ^ Junquera P (2015-07-26). "Fenbendazole, Anthelmintic for Veterinary Use on Cattle, Sheep, Goats, Pig, Poultry, Horses, Dogs and Cats Against Roundworms and Tapeworms". PARASITIPEDIA. Retrieved 2015-09-08.
- ^ Junquera P (2015-07-26). "Oxfendazole, Anthelmintic for Veterinary Use on Cattle, Sheep, Goats, Horses, Dogs and Cats Against Roundworms and Tapeworms". PARASITIPEDIA. Retrieved 2015-09-08.
- ^ Junquera P (2015-07-26). "Febantel for Veterinary Use on Dogs, Cats, Cattle, Sheep, Goats, Pig and Poultry Against Roundworms and Tapeworms". PARASITIPEDIA. Retrieved 2015-09-08.