| This is the
talk page for discussing improvements to the
Sucrose article. This is not a forum for general discussion of the article's subject. |
Article policies
|
| Find sources: Google ( books · news · scholar · free images · WP refs) · FENS · JSTOR · TWL |
|
| This It is of interest to the following WikiProjects: | ||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
|
This article links to one or more target anchors that no longer exist.
Please help fix the broken anchors. You can remove this template after fixing the problems. |
Reporting errors |
Here is the link removed:
| ImageFile3 = Sucrose ball-and-stick.gif | ImageName3 = Ball-and-stick model of sucrose molecule
It was annoying and can make it almost impossible to read the text, adds absolutely nothing, and is not even a very good quality image. There is no pedagogic value to such an image. Certainly an external link to a manipulable 3D graphic such as https://chemapps.stolaf.edu/jmol/jmol.php?model=sucrose might be appropriate. Perhaps I am oversensitive about this. I personally cannot read text next to a moving image. I figure that if it is that disturbing to me, it might be to others. I would like to find out what research has been done on this, but I don't have any at this moment. There must be some somewhere.
Hansonrstolaf ( talk) 03:20, 26 April 2018 (UTC)
—
This animation was a shock to me. They are a major usability issue for distractible people. I've never seen one like this on Wikipedia before and hope it does not become a trend. Unwanted animations are one of the worst things about the web, and the lack of them and other distractions is one of the great things about Wikipedia. — Preceding unsigned comment added by 2001:569:529F:1F00:C4DD:2BE2:EB3:B43A ( talk) 18:53, 10 January 2022 (UTC)
It says the boiling point of sucrose is ".dec" what does that mean?
- Under decomposition. Meaning the saccharose gets destroyed while boiling. -- 188.103.229.169 ( talk) 21:49, 14 July 2013 (UTC)
It means that before it can get to a boiling point, where liquid would be in equilibrium with vapor at one atmosphere of pressure, it is destroyed. Hansonrstolaf ( talk) 03:37, 26 April 2018 (UTC)
I combined comments of the same repeated topic to one area where it can be concentrated. I hope no one minds. If so I do apologize in advance I am still learning. -- Doctorkc ( talk) 07:45, 28 January 2010 (UTC)
People, and in fact most other mammals except members of the cat family, will gladly accept a food sweetened with sucrose, even if they aren't hungry.
Does anyone know why cats are unique? Do they dislike the taste of sucrose, or do they only refuse sweetened food if they're not hungry? --
Bkell 07:58, 10 Feb 2004 (UTC)
- faulty gene on a feline ancestor - search on Washington Post site. GraemeLeggett 15:58, 28 July 2005 (UTC)
http://www.plosgenetics.org/article/info:doi/10.1371/journal.pgen.0010003 - it explains it all AndrewFlorea ( talk) 13:46, 18 October 2009 (UTC)
Erm
My cat does... but I suppose that doesn't make for a good reference...
- a local vet says that they simply don't taste it, or don't taste it very strongly: if they're going after something sweet it's probably because it has fat in it as well (i.e. melted ice cream). i guess if they don't have enzymes to break sugars down they don't need the taste receptors either. — Clarknova 18:53, 17 Oct 2004 (UTC)
Dude, Cats are just too smart, that's why.
-Tim
- Wrong-My cat ate a sugar solution! Scorpionman 17:43, 23 January 2006 (UTC)
Some users were saying above that cats can't taste sucrose. Well, I highly disagree. I made a saturated sugar solution, poured the incredibly thick syrup on the ground, and my cat saw it and started licking it up! Can anyone explain that?
Scorpionman
02:35, 21 January 2006 (UTC)
ur retarded if you think that just because you put some sugar water on the floor and your cat ate it means that they can taste anything. if you put just normal water on the floor the cat would eat it too.
Do you want to stick the other chemical name on?
[beta]-D-Fructofuranosyl [alpha]-D-glucopyranoside
Source: http://www.chem.qmul.ac.uk/iupac/2carb/app.html
Phil If you feel a change is needed, feel free to make it yourself! Wikipedia is a wiki, so anyone — including you — can edit any article by clicking the edit this page tab at the top of the page. You don't even need to log in, although there are several reasons why you might want to. Wikipedia convention is to be bold and not be afraid of making mistakes. If you're not sure how editing works, have a look at How to edit a page, or try out the Sandbox to test your editing skills. New contributors are always welcome. -- fvw * 01:13, 2004 Dec 22 (UTC)
Some of the comments don't seem to be relevant to wikipedia? -- Doctorkc ( talk) 07:52, 28 January 2010 (UTC)
When I was researching Maltose ,Something struck me as odd about the chemical formula. I came over here and found out what was wrong, the same fomula for this is repeated over there. So which is the right one?
- The empirical formulas are the same. Note that the full names differ. GraemeLeggett 20:27, 20 August 2005 (UTC)
Hmm... isn't that an alpha-alpha linkage in the picture?
- Yeah, I'm confused. I thought alpha-fructose has the alcohol group (OH) upside in the anomeric carbon 83.44.34.89 ( talk) 21:54, 1 March 2008 (UTC)
- It is correct. The fructose unit is "upside down" relative to how it is generally shown. alpha/beta is an example of a relative stereochemical descriptor (chemistry). "beta" means "on the same side as the CH2OH group" (defining "D"); "alpha" means "on the opposite side of the CH2OH group, when "D". (Actually, it is a bit more complicated than that. "beta" means on the opposite side of the vertical line of a Fischer projection relative to the last oxygen atom of the chain -- the one on the center defining L or D. [ [1]] So L/D gets you the absolute configuration of the entire sugar, and alpha/beta gets you the relative orientation of the anomeric center relative to that. Hansonrstolaf ( talk) 03:42, 26 April 2018 (UTC)
Sucrose is composed of two simple sugars: fructose and glucose. Now, I can't count how many times I've heard the phrase "sugar is bad for you". What part of sucrose is bad: the fructose, or the glucose? Or both? Scorpionman 18:59, 27 October 2005 (UTC)
- You're not thinking.. Glucose is a sugar, fructose is a sugar, sucrose is a sugar. Sugar is bad for you. 219.77.98.28 09:18, 12 February 2006 (UTC)
What a nice topic for me to share with i have diabetes and i am 18 years and i love sugar in its solid state or dissolved phase but that was before i have diabetes 8 years ago i used to take my milk with eight tea spoons of sugar in the morning and the rest of the meals high sugar food cake, dounts, i even take my soup with sugar...am sickooo i know that and the messed up part now i can't taste it anymore and am crazy i made all the internet researches but i couldn't find a replacment for a something has the same taste of sugar altough i study pharmachy but i gave up....so does anyone know a replacment for sugar but without causing my diabetes worse —Preceding
unsigned comment added by
41.235.109.242 (
talk)
01:14, 16 October 2008 (UTC)
Type One diabetes is a big topic. Control of blood sugar levels is so important to reduce long-term effects of diabetes. The other health topic related to sucrose is tooth decay. As a practicing dentist, I think I have a perspective. The health problems of sucrose should be divided into separate topics: Diabetes, type one and two; tooth decay; etc. And one more comment: sugar can't be all bad. People who work hard physically can sure use it. And without the sweetness of mother's milk would a baby take longer to learn to suckle? Everything has its place. —Preceding unsigned comment added by LFlagg ( talk • contribs) 00:21, 17 March 2010 (UTC)
Your structure is wrong - it shows D glucose and L-fructose - should be D-glucose and D-fructose. Good picture is available on commons. See the difference:

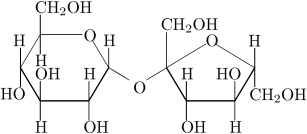
- You are correct. I've fixed the mistake. (In the future, you're welcome to do it yourself.) Edgar181 21:41, 4 January 2006 (UTC)
- Should the "sucrose.png" be removed, and "saccharose.png" renamed, then? Or at least swop the names? I don't know how to do it. Remember to swop the names in this talk page if you do. 219.77.98.28 09:20, 12 February 2006 (UTC)
Doesn't this diagram show a (1,5) link between alpha-D-glucose and beta-D-fructose (which is missing an "O" on the 2 carbon? Or, if you've reversed the order of the fructose carbons to read counter clockwise, then isn't the 6 carbon pointing in the wrong direction and you're showing an alpha-fructose? Please correct me if I'm wrong, but I think that this diagram is incorrect in a few ways. Thanks. Christophe, Brooklyn NY. —Preceding unsigned comment added by 141.149.61.111 ( talk) 14:11, 24 November 2008 (UTC)
I removed the statement, "Studies have also shown that tumors, especially cancers, consume most of the sugar people eat." There is a grain of truth there, in that many cancer cells take up glucose at a faster rate than the cells of surrounding tissues, making it possible to locate metastases by using PET scanning to detect concentrations of radioactively tagged glucose. That's entirely different, though, from saying that cancer consumes most of the sugar people eat, a statement that is self-evidently untrue for the vast majority of people who don't even have cancer. Since the true statement is really about glucose, not sucrose, and is far more relevant to articles about cancer and radiology than to one about sugar, I thought it better to omit the reference than to correct it. Paul Turner 16:51, 25 November 2005 (UTC)
I am skeptical that "your cleansing source online" [2] is a reliable source to include in this article. I would like to see some references to medical journal articles about sugar "leeching nutrients from the body because it is so nutriet poor". Anyone care to give opinions? -- Syrthiss 14:39, 8 December 2005 (UTC)
- That is a commercial site selling a colon cleansing program in which one receives supervision and instructions from someone calling himself Jos-hua Medicine Man. I took out the link and the information taken directly from it. Paul Turner 10:48, 14 December 2005 (UTC)
- lol I hadn't seen that. Ok, good call on both our parts. :) -- Syrthiss 13:23, 14 December 2005 (UTC)
I changed the sentence reading "As such it is common in many processed and junk foods" to "As such it is common in many processed and so-called "junk foods"." I feel this is slightly more neutral. Joeylawn 02:59, 15 February 2006 (UTC)
I think the safety diamonds have recently been updated for sucrose. The new bottle in the lab I work in is rated health-1, flamability-2, and reactivity-1. I wonder what they're thinking of when they say sucrose is a "slight" health risk? "Warning: may cause obesity if consumed in large amounts"? -- AaronM 12:45, 16 August 2006 (UTC)
- Yeah I was wondering why would something that is consumed by just about everyone on Earth would "cause irritation but only minor residual injury". Someone mind checking this out? BeefRendang 13:31, 3 May 2007 (UTC)
I am not a chemist, but I thought sucrose contains two glucose molecules, not one glucose and one fructose, as the article claims? Is this an error or am I mistaken? 24.20.118.3 16:55, 9 February 2007 (UTC)Dan
- You're mistaken! See for instance http://www.elmhurst.edu/~chm/vchembook/546sucrose.html
Specifically, the disaccharide consisting of two dextrose (glucose) molecules is maltose. Groogle 01:21, 17 February 2007 (UTC)
Is Sucrose actually combustible? I thought it simple decomposed but I only have three years of high school backing me up so there's a good chance I'm wrong. If it can is there something that must happen like direct flame or no flame and only heat?
- There is an amazingly fun way to test this one out yourself - all you need is a bag of sugar and a flame! Inhuman14 ( talk) 01:25, 17 December 2007 (UTC)
- The sugar mill in Savannah, Georgia blew up the other day, killing 12 workers. Sugar dust is a dangerous explosive !! That should settle the issue. —Preceding unsigned comment added by 208.63.237.3 ( talk) 05:25, 9 March 2008 (UTC)
I read somewhere years ago that sugar (sucrose) is very similar in chemical structure to alcohol, and thus has similar combustibility. Some words to this effect in the article would be nice. — Loadmaster ( talk) 14:37, 20 April 2009 (UTC)
- The similarity goes no further than having the same constituents: carbon, hyrogen and oxygen. The structure of simple alcohols ( methanol, ethanol, propanol, butanol, etc - click the links to see the structures) is completely different. You may be thinking of a more complex structure with an -OH radical - such a structure may well be called an alcohol as well, however an unqualified reference to alcohol would usually be understood to mean one of the short-chain simple forms, as found in methylated sprits or alcoholic beverages. -- Timberframe ( talk) 14:59, 20 April 2009 (UTC)
The article does not specify what the main sources of sucrose are. Is it the suger of sugar cane? Or are there other important sources of the substance? —Preceding unsigned comment added by 86.129.156.173 ( talk) 23:00, 22 November 2007 (UTC)
- The natural origin of sucrose is living-plant photosynthesis, energized by visible light -- whereby (typically) sunlight, water and carbon dioxide (from the air) are reacted.
- Does anyone know of any other radiant-energy process other than visible-light photosynthesis which can produce sucrose or some other simple hydrocarbon ? Such a process could provide an energy storage, transport and recovery mechanism far superior to hydrogen, I think !
. —Preceding unsigned comment added by 208.63.237.3 ( talk) 05:35, 9 March 2008 (UTC)
- Hello. Even though these were old posts, I too found it odd that there was no mention of the natural occurrence and sources of sucrose. I have now put up a new section called "Sources" with a little general information. RhinoMind ( talk) 18:54, 10 August 2015 (UTC)
What's chewcowenthia? A google search comes up with nothing. —Preceding unsigned comment added by 65.35.236.126 ( talk) 00:33, 4 April 2008 (UTC)
The picture of sucrose only has 21 Hydrogens. The two pictures on this talk page each have 22 Hydrogens. The missing one is attached to glucose-carbon 5. The carbon immediately below the non-cyclic carbon. —Preceding unsigned comment added by 69.49.55.240 ( talk) 01:08, 29 April 2008 (UTC)
In structural formulas it is common to not show all the hydrogens. Unless a carbon has a formal positive charge it is assumed to be completely saturated with hydrogens. — Preceding unsigned comment added by 24.127.234.20 ( talk) 23:04, 9 June 2011 (UTC)
I am making ice cream. Why does fructose interfere with the crystaline structure of ice water, but sucrose does not? I would like to know more about how sucrose acts in ice crystals, and chemical applications in food production. —Preceding unsigned comment added by Stevo517 ( talk • contribs) 06:05, 6 August 2008 (UTC)
Why is the arrow 1↔2 two-sided? -- kupirijo ( talk) 22:52, 21 October 2008 (UTC)
- Although most soft drinks in the USA are now made with high fructose corn syrup, not sucrose, this makes little functional difference, since high fructose corn syrup contains fructose and glucose in a similar ratio to that produced metabolically from sucrose.
This statement is patently wrong. Yes, HFCS contains fructose and glucose in similar ratios, but as sucrose is a disaccharide and its breakdown is limited by the availability of sucrase, the availability of fructose (which requires the liver enzymes fructokinase, triokinase, and to some extent, aldolase B) is therefore regulated.
With HFCS, no such regulation exists and the liver produces these enzymes in vast quantities to break down the flood of free fructose.
So clearly, there is a major functional difference. —Preceding unsigned comment added by 72.204.3.74 ( talk) 17:08, 6 July 2009 (UTC)
This is a valid point, why hasn't a change been made to the main article? —Preceding
unsigned comment added by
128.255.5.66 (
talk)
15:53, 15 December 2009 (UTC)
Almost all of sugar is about sucrose. Those few parts that are not about sucrose duplicate the content in carbohydrates. So readers are not getting the best presentation, and editors are duplicating their efforts. I understand that sugar has many uses and many cultural impacts.-- Smokefoot ( talk) 22:15, 3 September 2009 (UTC)
- Generally Agree but - the sugar page should be maintained as an entry point to other pages dealing with properties and uses of sugars (saccharides). Sugar, in general parlance, means nothing more than "table sugar" or sucrose. This is what the vast majority of people mean by sugar when they do a search or look-up. Wikipedia is targeted at everyone so a general description page with entry points makes sense to me. Chemical structures are not warranted on the sugar page. Please comment. Codwiki ( talk) 20:38, 8 December 2009 (UTC)
- Oppose Whilst there may be duplication of content, Sugar is a term used for any small carbohydrate of which sucrose is one such sugar. As Sugar states however there are many different sugars and sucrose deserves a separate article particularly for its function in plants and as being the most generic sugar. I don't have the time but the biochemistry section of sucrose could really have a lot more detail. Smartse ( talk) 23:22, 3 September 2009 (UTC)
- Thanks. Excellent points: (i) some duplication of content is no reason for a merger and (ii) sugar is a term for any small carbohydrate. Small sugars are however covered in carbohydrate, an article that is pretty good and covers all sugars with a natural bias toward sucrose. In terms of your interest in "small sugars", these are are covered not only in carbohydrate, but also in disaccharide and trisaccharide. I guess an alternative interpretation would be to strip out of sugar the detailed focus on sucrose.-- Smokefoot ( talk) 02:22, 4 September 2009 (UTC)
- Also opposed. Sugar is a general term, just as is carbohydrate or disaccharide, but the article is mostly about a specific sugar. If anything, it would be better to strip the current sugar article to the general definition and move specifics to more relevant locations. So perhaps move the relevant text to table sugar or sucrose and keep sugar generic. This seems analogous to a situation like gas versus gasoline or petrol. So, I think a more logical solution is a better thought out separation between the two rather than a merge.-- Glycoform ( talk) 05:04, 4 September 2009 (UTC)
- Thanks also. Here are the IUPAC definitions
- Sugar: "A loose term applied to monosaccharides and lower oligosaccharides."
- Carbohydrate: "Originally, compounds such as aldoses and ketoses having the stoichiometric formula Cn(H2O)n, hence 'hydrates of carbon'. The generic term carbohydrate includes monosaccharides, oligosaccharides and polysaccharides as well as substances derived from monosaccharides by reduction ... by oxidation ... by replacement of one or more hydroxy group(s) .... It also includes derivatives of these compounds."
- We can shift most sucrose-specific material from sugar to sucrose, that aspect most editors would agree to, but what remains in sugar will be carbohydrate. My guess is that 99.9% of readers looking up sugar will want to read about table sugar (a redirect to sucrose) and will be uninterested-in/frustrated-by our nuanced definitions. But maybe we deal with that issue later after realigning sugar and sucrose content.-- Smokefoot ( talk) 12:55, 4 September 2009 (UTC)
- Oppose I agree with Smartse
case closed. Thanks for the input, even if I didnt get my way. I moved the sucrose-specific material (the majority of the content) from sugar to sucrose. Remaining at sugar is a modest but respectable article about several sugars. I still think that sugar should be merged with carbohydrate since they are colloguial and technical terms for the same thing. But we can debate that point at another time. -- Smokefoot ( talk) 00:18, 18 September 2009 (UTC)
- Oppose —Preceding unsigned comment added by 71.205.160.222 ( talk) 22:43, 20 October 2009 (UTC)
- Oppose Let's make a white powders page and merge all of them. Galactose doesn't redirect to sugar and neither should sucrose. Sugar is the stuff that's used in cooking, a pop-culture phenomenon, an industry. Sucrose is used in labs and to explain chemical processes and is one of I'm sure thousands of molecular forms of sugar. Two completely different things similar in a few ways...two completely different pages. It's like the travesty on the salt page. It's about table salt!! Unbelievable. —Preceding unsigned comment added by Tnotejack ( talk • contribs) 01:16, 22 October 2009 (UTC)
- Strongly Oppose Sugar and Sucrose are not the same thing. This is a article about a CHEMICAL COMPOUND. Sugar should be an article mainly as a cooking ingredient. Tangerine! ( talk) 04:00, 3 December 2009 (UTC)
- [adding to a closed debate] oppose but the two articles sugar and sucrose are still a messy overlap. We need to where we are talking about the culinary stuff and where about the chemicial compound. 112.118.167.5 ( talk) 14:15, 6 June 2011 (UTC)
- Oppose Didn't knew this was a closed vote/discussion? Is it? Anyway, I oppose for reasons I have explained in my new post below called Article splitting?. I actually suggest splitting this article in two. Please go and read my post and reply there. RhinoMind ( talk) 19:24, 10 August 2015 (UTC)
It says on this page that sucrose has a high GI. I tried to find a source to verify this and couldn't - most websites put the GI of table sugar at anywhere between 43-61, putting it in the low or medium category. can anyone find a definite figure for the GI of sucrose? Michael1244 ( talk) 00:47, 9 January 2010 (UTC)
I don't see the point of that table, showing the ratio of sugar to fat consumed has increased tells us nothing, it's totally biased. Fat intake has increased in the Western World in recent decades, just to a slightly lesser extent than carbs. Obesity has increased because people are eating more of everything, not because the roportions of different types of food has changed. 82.5.217.254 ( talk) 13:57, 16 February 2010 (UTC)
this article claims fructose is the primary sugar in pears and grapes. Grapes contain glucose: a ripe grape contains just as much fructose as glucose. see Webster's 1913 entry for grape sugar and for glucose at http://dictionary.die.net/glucose, and also http://www.extension.iastate.edu/NR/rdonlyres/A647BBD4-08D5-494B-A55B-680667E6C342/56373/compositionofgrapes.pdf
Also, the grapes page does not mention which sugar is in them. (though glucose says grape sugar is a synonym - no source.) I would propose correcting this one ( sucrose) for grapes, or deleting the grapes statement altogether since it doesn't relate to sucrose, (though it may be valid for pears) and possible adding a line to grapes regarding glucose, though I don't know where it fits in context. -David HappySneezy ( talk) 03:40, 13 June 2011 (UTC)
The article claims that caster sugar, and similar, are merely regular cane sucrose but with much smaller crystals. Try caramelizing both types and you'll find that the difference is more than cosmetic.
Are there any isomers of sucrose that are levorotatory? 173.17.169.171 ( talk) 09:57, 2 March 2012 (UTC)
What's the speed constant of socrose hydrolyzation reaction ? — Preceding unsigned comment added by JoannaAnn ( talk • contribs) 14:01, 11 April 2012 (UTC)
It seems that all the content of that section is based upon religious issues rather then anything regarding ethics. Religion and ethics are not necessarily the same thing. 76.170.170.58 ( talk) 09:47, 31 March 2013 (UTC)
Someone should do a write-up on the controversies surrounding the presumed melting point of sucrose. As can be seen at http://www.sciencedaily.com/releases/2011/07/110725123549.htm there is an assumption that sugar was always believed to have a melting point, and that this proof of decomposition was startling new information. That would be fascinating enough, yet but to quote the 1853 "Cyclopedia of Useful Arts & Manufactures":
"crystallized cane-sugar and barley-sugar consist of C12H11O11, but if heated to temperatures between 300 and 400 [degrees] they lose two equivalents of water, and become converted to caramel".
The apparent centuries-long *loss* of facts related to sugary non-meltability and its recent rediscovery would make a useful background for students of the scientific method. 76.21.5.244 ( talk) 11:48, 23 August 2013 (UTC)
Hello. I suggest we split this article in an article on "Sucrose" and another article on "Sugar refinement".
The subject of sucrose is much broader than sugar refinement alone and the article as it is, contains too much information on the refinement process and history. While it is important and interesting in itself, I dont think it should fill up so much space in an article on just sucrose. It is worthy of an article all on its own. Such a new page can also contain information on the refinement of other sugars, such as coconut and palm sugar for example. In addition it can also tell more about molasses perhaps. Post your comments below please. RhinoMind ( talk) 19:19, 10 August 2015 (UTC)
- After reading the old merger proposal above, some of the sugar refinement stuff in this article, could perhaps be merged with the Sugar page. Maybe. Just a thought. RhinoMind ( talk) 19:27, 10 August 2015 (UTC)
Note that I just corrected the paragraph about HFCS health concerns. ( diff) While the previous version did not say anything strictly inaccurate - "there are claims" that HFCS is especially nasty, and the industry disputes these claims - it was extremely misleading because it ignored the very large preponderance of scientific and medical opinion, which agrees with the industry that HFCS is not especially nasty. This also brings the paragraph in line with the main high fructose corn syrup article, which correctly describes the consensus (although IMO it still gives too much weight to a few really shitty advocacy studies, but whatever.)
Also, the only "citation" here previously was a half-assed reference to a TV documentary - "'The man who made us fat', TV series narrated by Jacques Peritti, 2012?". Heh. 174.93.32.178 ( talk) 19:32, 27 September 2015 (UTC)
Hello fellow Wikipedians,
I have just modified one external link on Sucrose. Please take a moment to review my edit. If you have any questions, or need the bot to ignore the links, or the page altogether, please visit this simple FaQ for additional information. I made the following changes:
- Corrected formatting/usage for http://www.abcvitaminslife.com/HealthFacts/Article429.aspx
When you have finished reviewing my changes, please set the checked parameter below to true or failed to let others know (documentation at {{
Sourcecheck}}).
This message was posted before February 2018.
After February 2018, "External links modified" talk page sections are no longer generated or monitored by InternetArchiveBot. No special action is required regarding these talk page notices, other than
regular verification using the archive tool instructions below. Editors
have permission to delete these "External links modified" talk page sections if they want to de-clutter talk pages, but see the
RfC before doing mass systematic removals. This message is updated dynamically through the template {{
source check}} (last update: 5 June 2024).
- If you have discovered URLs which were erroneously considered dead by the bot, you can report them with this tool.
- If you found an error with any archives or the URLs themselves, you can fix them with this tool.
Cheers.— cyberbot II Talk to my owner:Online 12:36, 4 April 2016 (UTC)
The comment(s) below were originally left at Talk:Sucrose/Comments, and are posted here for posterity. Following several discussions in past years, these subpages are now deprecated. The comments may be irrelevant or outdated; if so, please feel free to remove this section.
| This article is in need of urgent upgrading. I am no expert but have a layman's interest in scientific advances in the study of glucose/sucrose/fructose in the human diet. Even I can see that the comments on the adverse health effects of the fructose component of sucrose are way off the mark. Recent studies have identified Fructose as a major culprit in causing insulin resistance, diabeties, obesity, artery/heart disease etc. The Wikipedia article on Fructose contains much of the required information; it just needs to be worked into the Sucrose article by someone who knows what they are doing. 121.45.110.194 ( talk) 09:40, 28 January 2008 (UTC) |
Last edited at 09:40, 28 January 2008 (UTC). Substituted at 07:14, 30 April 2016 (UTC)
On the nutrient label of any edible product, why is sugar the only nutrient ingredient without a daily nutrient value? Thank you M.cook — Preceding unsigned comment added by 65.94.195.17 ( talk) 18:06, 20 June 2016 (UTC)
Can someone publish the magnetic susceptibility of sucrose in the chembox section #2 physical properties? I'm having a hard time finding such simple data. If someone has a magnetic susceptibility balance it would be just a short calculation that could benefit everyone! Standard magnetic susceptibility units are either cm3/mol or cm3/g. Remember to fill in the correct units or place the data here and I can place it in the physical properties box for us. TerpeneOtto ( talk) 04:47, 7 December 2016 (UTC)
Someone asked me why sugar is so soluble in water. I had previously told them that ionic bonds generally resulted in things that were soluble in water, whereas covalent bonds generally resulted in things that were relatively insoluble in water. But they brought up sugar, and I guessed that its solubility had s.t. to do with the oxygens. I came here looking for an answer, but the problem is not discussed. Could someone who knows more about such things than I do add a paragraph on that? Looking elsewhere, it appears that it has to do with the OH groups on sugar, which are somewhat polar (as are the OH bonds in water). But a fuller explanation would be...well, fuller. Mcswell ( talk) 02:24, 1 May 2018 (UTC)
"digestion releases about 4 small calories per gram"
Doesn't a gram contain 4 regular kcal? Perhaps a historic unit like "small" should be avoided. -- J7n ( talk) 05:03, 24 February 2021 (UTC)
I see that the source is : "Prices from 1960 to 2010 are ICE spot sugar prices. Prices from 2011 onward are ICE contract 11 nearby futures prices. Data compiled by the USDA.". It would be useful if the graph could be annotated to say whether the prices are for 1 January, 30 June, maximum for the year, average for the year, or what. Exbrum ( talk) 10:55, 2 April 2021 (UTC)
The image currently* describing sucrose on the Main page shows a (#1C to #5C) linkage of glucose to fructose respectively. But it should show (#1C to #2C) linkage of glucose to fructose respectively.
- -> At time of writing that is 19 November 2021; 12:45 IST 2402:3A80:1818:654B:0:6:9D67:9B01 ( talk) 19:16, 18 November 2021 (UTC)
At the end of the summary article*, the chemical formula is malformed when seen in Android 10. Perhaps the author omitted trailing "end sub" digit delimiters in angle brackets. *Typically what shows up when there's an embedded ref. in another article Nikevich 22:38, 12 January 2022 (UTC)
- B-Class vital articles
- Wikipedia level-5 vital articles
- Wikipedia vital articles in Biology and health sciences
- B-Class level-5 vital articles
- Wikipedia level-5 vital articles in Biology and health sciences
- B-Class vital articles in Biology and health sciences
- B-Class chemicals articles
- High-importance chemicals articles
- B-Class Food and drink articles
- Mid-importance Food and drink articles
- WikiProject Food and drink articles
- B-Class Molecular Biology articles
- Unknown-importance Molecular Biology articles
- B-Class MCB articles
- Low-importance MCB articles
- WikiProject Molecular and Cellular Biology articles
- All WikiProject Molecular Biology pages
| This is the
talk page for discussing improvements to the
Sucrose article. This is not a forum for general discussion of the article's subject. |
Article policies
|
| Find sources: Google ( books · news · scholar · free images · WP refs) · FENS · JSTOR · TWL |
|
| This It is of interest to the following WikiProjects: | ||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
|
This article links to one or more target anchors that no longer exist.
Please help fix the broken anchors. You can remove this template after fixing the problems. |
Reporting errors |
Here is the link removed:
| ImageFile3 = Sucrose ball-and-stick.gif | ImageName3 = Ball-and-stick model of sucrose molecule
It was annoying and can make it almost impossible to read the text, adds absolutely nothing, and is not even a very good quality image. There is no pedagogic value to such an image. Certainly an external link to a manipulable 3D graphic such as https://chemapps.stolaf.edu/jmol/jmol.php?model=sucrose might be appropriate. Perhaps I am oversensitive about this. I personally cannot read text next to a moving image. I figure that if it is that disturbing to me, it might be to others. I would like to find out what research has been done on this, but I don't have any at this moment. There must be some somewhere.
Hansonrstolaf ( talk) 03:20, 26 April 2018 (UTC)
—
This animation was a shock to me. They are a major usability issue for distractible people. I've never seen one like this on Wikipedia before and hope it does not become a trend. Unwanted animations are one of the worst things about the web, and the lack of them and other distractions is one of the great things about Wikipedia. — Preceding unsigned comment added by 2001:569:529F:1F00:C4DD:2BE2:EB3:B43A ( talk) 18:53, 10 January 2022 (UTC)
It says the boiling point of sucrose is ".dec" what does that mean?
- Under decomposition. Meaning the saccharose gets destroyed while boiling. -- 188.103.229.169 ( talk) 21:49, 14 July 2013 (UTC)
It means that before it can get to a boiling point, where liquid would be in equilibrium with vapor at one atmosphere of pressure, it is destroyed. Hansonrstolaf ( talk) 03:37, 26 April 2018 (UTC)
I combined comments of the same repeated topic to one area where it can be concentrated. I hope no one minds. If so I do apologize in advance I am still learning. -- Doctorkc ( talk) 07:45, 28 January 2010 (UTC)
People, and in fact most other mammals except members of the cat family, will gladly accept a food sweetened with sucrose, even if they aren't hungry.
Does anyone know why cats are unique? Do they dislike the taste of sucrose, or do they only refuse sweetened food if they're not hungry? --
Bkell 07:58, 10 Feb 2004 (UTC)
- faulty gene on a feline ancestor - search on Washington Post site. GraemeLeggett 15:58, 28 July 2005 (UTC)
http://www.plosgenetics.org/article/info:doi/10.1371/journal.pgen.0010003 - it explains it all AndrewFlorea ( talk) 13:46, 18 October 2009 (UTC)
Erm
My cat does... but I suppose that doesn't make for a good reference...
- a local vet says that they simply don't taste it, or don't taste it very strongly: if they're going after something sweet it's probably because it has fat in it as well (i.e. melted ice cream). i guess if they don't have enzymes to break sugars down they don't need the taste receptors either. — Clarknova 18:53, 17 Oct 2004 (UTC)
Dude, Cats are just too smart, that's why.
-Tim
- Wrong-My cat ate a sugar solution! Scorpionman 17:43, 23 January 2006 (UTC)
Some users were saying above that cats can't taste sucrose. Well, I highly disagree. I made a saturated sugar solution, poured the incredibly thick syrup on the ground, and my cat saw it and started licking it up! Can anyone explain that?
Scorpionman
02:35, 21 January 2006 (UTC)
ur retarded if you think that just because you put some sugar water on the floor and your cat ate it means that they can taste anything. if you put just normal water on the floor the cat would eat it too.
Do you want to stick the other chemical name on?
[beta]-D-Fructofuranosyl [alpha]-D-glucopyranoside
Source: http://www.chem.qmul.ac.uk/iupac/2carb/app.html
Phil If you feel a change is needed, feel free to make it yourself! Wikipedia is a wiki, so anyone — including you — can edit any article by clicking the edit this page tab at the top of the page. You don't even need to log in, although there are several reasons why you might want to. Wikipedia convention is to be bold and not be afraid of making mistakes. If you're not sure how editing works, have a look at How to edit a page, or try out the Sandbox to test your editing skills. New contributors are always welcome. -- fvw * 01:13, 2004 Dec 22 (UTC)
Some of the comments don't seem to be relevant to wikipedia? -- Doctorkc ( talk) 07:52, 28 January 2010 (UTC)
When I was researching Maltose ,Something struck me as odd about the chemical formula. I came over here and found out what was wrong, the same fomula for this is repeated over there. So which is the right one?
- The empirical formulas are the same. Note that the full names differ. GraemeLeggett 20:27, 20 August 2005 (UTC)
Hmm... isn't that an alpha-alpha linkage in the picture?
- Yeah, I'm confused. I thought alpha-fructose has the alcohol group (OH) upside in the anomeric carbon 83.44.34.89 ( talk) 21:54, 1 March 2008 (UTC)
- It is correct. The fructose unit is "upside down" relative to how it is generally shown. alpha/beta is an example of a relative stereochemical descriptor (chemistry). "beta" means "on the same side as the CH2OH group" (defining "D"); "alpha" means "on the opposite side of the CH2OH group, when "D". (Actually, it is a bit more complicated than that. "beta" means on the opposite side of the vertical line of a Fischer projection relative to the last oxygen atom of the chain -- the one on the center defining L or D. [ [1]] So L/D gets you the absolute configuration of the entire sugar, and alpha/beta gets you the relative orientation of the anomeric center relative to that. Hansonrstolaf ( talk) 03:42, 26 April 2018 (UTC)
Sucrose is composed of two simple sugars: fructose and glucose. Now, I can't count how many times I've heard the phrase "sugar is bad for you". What part of sucrose is bad: the fructose, or the glucose? Or both? Scorpionman 18:59, 27 October 2005 (UTC)
- You're not thinking.. Glucose is a sugar, fructose is a sugar, sucrose is a sugar. Sugar is bad for you. 219.77.98.28 09:18, 12 February 2006 (UTC)
What a nice topic for me to share with i have diabetes and i am 18 years and i love sugar in its solid state or dissolved phase but that was before i have diabetes 8 years ago i used to take my milk with eight tea spoons of sugar in the morning and the rest of the meals high sugar food cake, dounts, i even take my soup with sugar...am sickooo i know that and the messed up part now i can't taste it anymore and am crazy i made all the internet researches but i couldn't find a replacment for a something has the same taste of sugar altough i study pharmachy but i gave up....so does anyone know a replacment for sugar but without causing my diabetes worse —Preceding
unsigned comment added by
41.235.109.242 (
talk)
01:14, 16 October 2008 (UTC)
Type One diabetes is a big topic. Control of blood sugar levels is so important to reduce long-term effects of diabetes. The other health topic related to sucrose is tooth decay. As a practicing dentist, I think I have a perspective. The health problems of sucrose should be divided into separate topics: Diabetes, type one and two; tooth decay; etc. And one more comment: sugar can't be all bad. People who work hard physically can sure use it. And without the sweetness of mother's milk would a baby take longer to learn to suckle? Everything has its place. —Preceding unsigned comment added by LFlagg ( talk • contribs) 00:21, 17 March 2010 (UTC)
Your structure is wrong - it shows D glucose and L-fructose - should be D-glucose and D-fructose. Good picture is available on commons. See the difference:


- You are correct. I've fixed the mistake. (In the future, you're welcome to do it yourself.) Edgar181 21:41, 4 January 2006 (UTC)
- Should the "sucrose.png" be removed, and "saccharose.png" renamed, then? Or at least swop the names? I don't know how to do it. Remember to swop the names in this talk page if you do. 219.77.98.28 09:20, 12 February 2006 (UTC)
Doesn't this diagram show a (1,5) link between alpha-D-glucose and beta-D-fructose (which is missing an "O" on the 2 carbon? Or, if you've reversed the order of the fructose carbons to read counter clockwise, then isn't the 6 carbon pointing in the wrong direction and you're showing an alpha-fructose? Please correct me if I'm wrong, but I think that this diagram is incorrect in a few ways. Thanks. Christophe, Brooklyn NY. —Preceding unsigned comment added by 141.149.61.111 ( talk) 14:11, 24 November 2008 (UTC)
I removed the statement, "Studies have also shown that tumors, especially cancers, consume most of the sugar people eat." There is a grain of truth there, in that many cancer cells take up glucose at a faster rate than the cells of surrounding tissues, making it possible to locate metastases by using PET scanning to detect concentrations of radioactively tagged glucose. That's entirely different, though, from saying that cancer consumes most of the sugar people eat, a statement that is self-evidently untrue for the vast majority of people who don't even have cancer. Since the true statement is really about glucose, not sucrose, and is far more relevant to articles about cancer and radiology than to one about sugar, I thought it better to omit the reference than to correct it. Paul Turner 16:51, 25 November 2005 (UTC)
I am skeptical that "your cleansing source online" [2] is a reliable source to include in this article. I would like to see some references to medical journal articles about sugar "leeching nutrients from the body because it is so nutriet poor". Anyone care to give opinions? -- Syrthiss 14:39, 8 December 2005 (UTC)
- That is a commercial site selling a colon cleansing program in which one receives supervision and instructions from someone calling himself Jos-hua Medicine Man. I took out the link and the information taken directly from it. Paul Turner 10:48, 14 December 2005 (UTC)
- lol I hadn't seen that. Ok, good call on both our parts. :) -- Syrthiss 13:23, 14 December 2005 (UTC)
I changed the sentence reading "As such it is common in many processed and junk foods" to "As such it is common in many processed and so-called "junk foods"." I feel this is slightly more neutral. Joeylawn 02:59, 15 February 2006 (UTC)
I think the safety diamonds have recently been updated for sucrose. The new bottle in the lab I work in is rated health-1, flamability-2, and reactivity-1. I wonder what they're thinking of when they say sucrose is a "slight" health risk? "Warning: may cause obesity if consumed in large amounts"? -- AaronM 12:45, 16 August 2006 (UTC)
- Yeah I was wondering why would something that is consumed by just about everyone on Earth would "cause irritation but only minor residual injury". Someone mind checking this out? BeefRendang 13:31, 3 May 2007 (UTC)
I am not a chemist, but I thought sucrose contains two glucose molecules, not one glucose and one fructose, as the article claims? Is this an error or am I mistaken? 24.20.118.3 16:55, 9 February 2007 (UTC)Dan
- You're mistaken! See for instance http://www.elmhurst.edu/~chm/vchembook/546sucrose.html
Specifically, the disaccharide consisting of two dextrose (glucose) molecules is maltose. Groogle 01:21, 17 February 2007 (UTC)
Is Sucrose actually combustible? I thought it simple decomposed but I only have three years of high school backing me up so there's a good chance I'm wrong. If it can is there something that must happen like direct flame or no flame and only heat?
- There is an amazingly fun way to test this one out yourself - all you need is a bag of sugar and a flame! Inhuman14 ( talk) 01:25, 17 December 2007 (UTC)
- The sugar mill in Savannah, Georgia blew up the other day, killing 12 workers. Sugar dust is a dangerous explosive !! That should settle the issue. —Preceding unsigned comment added by 208.63.237.3 ( talk) 05:25, 9 March 2008 (UTC)
I read somewhere years ago that sugar (sucrose) is very similar in chemical structure to alcohol, and thus has similar combustibility. Some words to this effect in the article would be nice. — Loadmaster ( talk) 14:37, 20 April 2009 (UTC)
- The similarity goes no further than having the same constituents: carbon, hyrogen and oxygen. The structure of simple alcohols ( methanol, ethanol, propanol, butanol, etc - click the links to see the structures) is completely different. You may be thinking of a more complex structure with an -OH radical - such a structure may well be called an alcohol as well, however an unqualified reference to alcohol would usually be understood to mean one of the short-chain simple forms, as found in methylated sprits or alcoholic beverages. -- Timberframe ( talk) 14:59, 20 April 2009 (UTC)
The article does not specify what the main sources of sucrose are. Is it the suger of sugar cane? Or are there other important sources of the substance? —Preceding unsigned comment added by 86.129.156.173 ( talk) 23:00, 22 November 2007 (UTC)
- The natural origin of sucrose is living-plant photosynthesis, energized by visible light -- whereby (typically) sunlight, water and carbon dioxide (from the air) are reacted.
- Does anyone know of any other radiant-energy process other than visible-light photosynthesis which can produce sucrose or some other simple hydrocarbon ? Such a process could provide an energy storage, transport and recovery mechanism far superior to hydrogen, I think !
. —Preceding unsigned comment added by 208.63.237.3 ( talk) 05:35, 9 March 2008 (UTC)
- Hello. Even though these were old posts, I too found it odd that there was no mention of the natural occurrence and sources of sucrose. I have now put up a new section called "Sources" with a little general information. RhinoMind ( talk) 18:54, 10 August 2015 (UTC)
What's chewcowenthia? A google search comes up with nothing. —Preceding unsigned comment added by 65.35.236.126 ( talk) 00:33, 4 April 2008 (UTC)
The picture of sucrose only has 21 Hydrogens. The two pictures on this talk page each have 22 Hydrogens. The missing one is attached to glucose-carbon 5. The carbon immediately below the non-cyclic carbon. —Preceding unsigned comment added by 69.49.55.240 ( talk) 01:08, 29 April 2008 (UTC)
In structural formulas it is common to not show all the hydrogens. Unless a carbon has a formal positive charge it is assumed to be completely saturated with hydrogens. — Preceding unsigned comment added by 24.127.234.20 ( talk) 23:04, 9 June 2011 (UTC)
I am making ice cream. Why does fructose interfere with the crystaline structure of ice water, but sucrose does not? I would like to know more about how sucrose acts in ice crystals, and chemical applications in food production. —Preceding unsigned comment added by Stevo517 ( talk • contribs) 06:05, 6 August 2008 (UTC)
Why is the arrow 1↔2 two-sided? -- kupirijo ( talk) 22:52, 21 October 2008 (UTC)
- Although most soft drinks in the USA are now made with high fructose corn syrup, not sucrose, this makes little functional difference, since high fructose corn syrup contains fructose and glucose in a similar ratio to that produced metabolically from sucrose.
This statement is patently wrong. Yes, HFCS contains fructose and glucose in similar ratios, but as sucrose is a disaccharide and its breakdown is limited by the availability of sucrase, the availability of fructose (which requires the liver enzymes fructokinase, triokinase, and to some extent, aldolase B) is therefore regulated.
With HFCS, no such regulation exists and the liver produces these enzymes in vast quantities to break down the flood of free fructose.
So clearly, there is a major functional difference. —Preceding unsigned comment added by 72.204.3.74 ( talk) 17:08, 6 July 2009 (UTC)
This is a valid point, why hasn't a change been made to the main article? —Preceding
unsigned comment added by
128.255.5.66 (
talk)
15:53, 15 December 2009 (UTC)
Almost all of sugar is about sucrose. Those few parts that are not about sucrose duplicate the content in carbohydrates. So readers are not getting the best presentation, and editors are duplicating their efforts. I understand that sugar has many uses and many cultural impacts.-- Smokefoot ( talk) 22:15, 3 September 2009 (UTC)
- Generally Agree but - the sugar page should be maintained as an entry point to other pages dealing with properties and uses of sugars (saccharides). Sugar, in general parlance, means nothing more than "table sugar" or sucrose. This is what the vast majority of people mean by sugar when they do a search or look-up. Wikipedia is targeted at everyone so a general description page with entry points makes sense to me. Chemical structures are not warranted on the sugar page. Please comment. Codwiki ( talk) 20:38, 8 December 2009 (UTC)
- Oppose Whilst there may be duplication of content, Sugar is a term used for any small carbohydrate of which sucrose is one such sugar. As Sugar states however there are many different sugars and sucrose deserves a separate article particularly for its function in plants and as being the most generic sugar. I don't have the time but the biochemistry section of sucrose could really have a lot more detail. Smartse ( talk) 23:22, 3 September 2009 (UTC)
- Thanks. Excellent points: (i) some duplication of content is no reason for a merger and (ii) sugar is a term for any small carbohydrate. Small sugars are however covered in carbohydrate, an article that is pretty good and covers all sugars with a natural bias toward sucrose. In terms of your interest in "small sugars", these are are covered not only in carbohydrate, but also in disaccharide and trisaccharide. I guess an alternative interpretation would be to strip out of sugar the detailed focus on sucrose.-- Smokefoot ( talk) 02:22, 4 September 2009 (UTC)
- Also opposed. Sugar is a general term, just as is carbohydrate or disaccharide, but the article is mostly about a specific sugar. If anything, it would be better to strip the current sugar article to the general definition and move specifics to more relevant locations. So perhaps move the relevant text to table sugar or sucrose and keep sugar generic. This seems analogous to a situation like gas versus gasoline or petrol. So, I think a more logical solution is a better thought out separation between the two rather than a merge.-- Glycoform ( talk) 05:04, 4 September 2009 (UTC)
- Thanks also. Here are the IUPAC definitions
- Sugar: "A loose term applied to monosaccharides and lower oligosaccharides."
- Carbohydrate: "Originally, compounds such as aldoses and ketoses having the stoichiometric formula Cn(H2O)n, hence 'hydrates of carbon'. The generic term carbohydrate includes monosaccharides, oligosaccharides and polysaccharides as well as substances derived from monosaccharides by reduction ... by oxidation ... by replacement of one or more hydroxy group(s) .... It also includes derivatives of these compounds."
- We can shift most sucrose-specific material from sugar to sucrose, that aspect most editors would agree to, but what remains in sugar will be carbohydrate. My guess is that 99.9% of readers looking up sugar will want to read about table sugar (a redirect to sucrose) and will be uninterested-in/frustrated-by our nuanced definitions. But maybe we deal with that issue later after realigning sugar and sucrose content.-- Smokefoot ( talk) 12:55, 4 September 2009 (UTC)
- Oppose I agree with Smartse
case closed. Thanks for the input, even if I didnt get my way. I moved the sucrose-specific material (the majority of the content) from sugar to sucrose. Remaining at sugar is a modest but respectable article about several sugars. I still think that sugar should be merged with carbohydrate since they are colloguial and technical terms for the same thing. But we can debate that point at another time. -- Smokefoot ( talk) 00:18, 18 September 2009 (UTC)
- Oppose —Preceding unsigned comment added by 71.205.160.222 ( talk) 22:43, 20 October 2009 (UTC)
- Oppose Let's make a white powders page and merge all of them. Galactose doesn't redirect to sugar and neither should sucrose. Sugar is the stuff that's used in cooking, a pop-culture phenomenon, an industry. Sucrose is used in labs and to explain chemical processes and is one of I'm sure thousands of molecular forms of sugar. Two completely different things similar in a few ways...two completely different pages. It's like the travesty on the salt page. It's about table salt!! Unbelievable. —Preceding unsigned comment added by Tnotejack ( talk • contribs) 01:16, 22 October 2009 (UTC)
- Strongly Oppose Sugar and Sucrose are not the same thing. This is a article about a CHEMICAL COMPOUND. Sugar should be an article mainly as a cooking ingredient. Tangerine! ( talk) 04:00, 3 December 2009 (UTC)
- [adding to a closed debate] oppose but the two articles sugar and sucrose are still a messy overlap. We need to where we are talking about the culinary stuff and where about the chemicial compound. 112.118.167.5 ( talk) 14:15, 6 June 2011 (UTC)
- Oppose Didn't knew this was a closed vote/discussion? Is it? Anyway, I oppose for reasons I have explained in my new post below called Article splitting?. I actually suggest splitting this article in two. Please go and read my post and reply there. RhinoMind ( talk) 19:24, 10 August 2015 (UTC)
It says on this page that sucrose has a high GI. I tried to find a source to verify this and couldn't - most websites put the GI of table sugar at anywhere between 43-61, putting it in the low or medium category. can anyone find a definite figure for the GI of sucrose? Michael1244 ( talk) 00:47, 9 January 2010 (UTC)
I don't see the point of that table, showing the ratio of sugar to fat consumed has increased tells us nothing, it's totally biased. Fat intake has increased in the Western World in recent decades, just to a slightly lesser extent than carbs. Obesity has increased because people are eating more of everything, not because the roportions of different types of food has changed. 82.5.217.254 ( talk) 13:57, 16 February 2010 (UTC)
this article claims fructose is the primary sugar in pears and grapes. Grapes contain glucose: a ripe grape contains just as much fructose as glucose. see Webster's 1913 entry for grape sugar and for glucose at http://dictionary.die.net/glucose, and also http://www.extension.iastate.edu/NR/rdonlyres/A647BBD4-08D5-494B-A55B-680667E6C342/56373/compositionofgrapes.pdf
Also, the grapes page does not mention which sugar is in them. (though glucose says grape sugar is a synonym - no source.) I would propose correcting this one ( sucrose) for grapes, or deleting the grapes statement altogether since it doesn't relate to sucrose, (though it may be valid for pears) and possible adding a line to grapes regarding glucose, though I don't know where it fits in context. -David HappySneezy ( talk) 03:40, 13 June 2011 (UTC)
The article claims that caster sugar, and similar, are merely regular cane sucrose but with much smaller crystals. Try caramelizing both types and you'll find that the difference is more than cosmetic.
Are there any isomers of sucrose that are levorotatory? 173.17.169.171 ( talk) 09:57, 2 March 2012 (UTC)
What's the speed constant of socrose hydrolyzation reaction ? — Preceding unsigned comment added by JoannaAnn ( talk • contribs) 14:01, 11 April 2012 (UTC)
It seems that all the content of that section is based upon religious issues rather then anything regarding ethics. Religion and ethics are not necessarily the same thing. 76.170.170.58 ( talk) 09:47, 31 March 2013 (UTC)
Someone should do a write-up on the controversies surrounding the presumed melting point of sucrose. As can be seen at http://www.sciencedaily.com/releases/2011/07/110725123549.htm there is an assumption that sugar was always believed to have a melting point, and that this proof of decomposition was startling new information. That would be fascinating enough, yet but to quote the 1853 "Cyclopedia of Useful Arts & Manufactures":
"crystallized cane-sugar and barley-sugar consist of C12H11O11, but if heated to temperatures between 300 and 400 [degrees] they lose two equivalents of water, and become converted to caramel".
The apparent centuries-long *loss* of facts related to sugary non-meltability and its recent rediscovery would make a useful background for students of the scientific method. 76.21.5.244 ( talk) 11:48, 23 August 2013 (UTC)
Hello. I suggest we split this article in an article on "Sucrose" and another article on "Sugar refinement".
The subject of sucrose is much broader than sugar refinement alone and the article as it is, contains too much information on the refinement process and history. While it is important and interesting in itself, I dont think it should fill up so much space in an article on just sucrose. It is worthy of an article all on its own. Such a new page can also contain information on the refinement of other sugars, such as coconut and palm sugar for example. In addition it can also tell more about molasses perhaps. Post your comments below please. RhinoMind ( talk) 19:19, 10 August 2015 (UTC)
- After reading the old merger proposal above, some of the sugar refinement stuff in this article, could perhaps be merged with the Sugar page. Maybe. Just a thought. RhinoMind ( talk) 19:27, 10 August 2015 (UTC)
Note that I just corrected the paragraph about HFCS health concerns. ( diff) While the previous version did not say anything strictly inaccurate - "there are claims" that HFCS is especially nasty, and the industry disputes these claims - it was extremely misleading because it ignored the very large preponderance of scientific and medical opinion, which agrees with the industry that HFCS is not especially nasty. This also brings the paragraph in line with the main high fructose corn syrup article, which correctly describes the consensus (although IMO it still gives too much weight to a few really shitty advocacy studies, but whatever.)
Also, the only "citation" here previously was a half-assed reference to a TV documentary - "'The man who made us fat', TV series narrated by Jacques Peritti, 2012?". Heh. 174.93.32.178 ( talk) 19:32, 27 September 2015 (UTC)
Hello fellow Wikipedians,
I have just modified one external link on Sucrose. Please take a moment to review my edit. If you have any questions, or need the bot to ignore the links, or the page altogether, please visit this simple FaQ for additional information. I made the following changes:
- Corrected formatting/usage for http://www.abcvitaminslife.com/HealthFacts/Article429.aspx
When you have finished reviewing my changes, please set the checked parameter below to true or failed to let others know (documentation at {{
Sourcecheck}}).
This message was posted before February 2018.
After February 2018, "External links modified" talk page sections are no longer generated or monitored by InternetArchiveBot. No special action is required regarding these talk page notices, other than
regular verification using the archive tool instructions below. Editors
have permission to delete these "External links modified" talk page sections if they want to de-clutter talk pages, but see the
RfC before doing mass systematic removals. This message is updated dynamically through the template {{
source check}} (last update: 5 June 2024).
- If you have discovered URLs which were erroneously considered dead by the bot, you can report them with this tool.
- If you found an error with any archives or the URLs themselves, you can fix them with this tool.
Cheers.— cyberbot II Talk to my owner:Online 12:36, 4 April 2016 (UTC)
The comment(s) below were originally left at Talk:Sucrose/Comments, and are posted here for posterity. Following several discussions in past years, these subpages are now deprecated. The comments may be irrelevant or outdated; if so, please feel free to remove this section.
| This article is in need of urgent upgrading. I am no expert but have a layman's interest in scientific advances in the study of glucose/sucrose/fructose in the human diet. Even I can see that the comments on the adverse health effects of the fructose component of sucrose are way off the mark. Recent studies have identified Fructose as a major culprit in causing insulin resistance, diabeties, obesity, artery/heart disease etc. The Wikipedia article on Fructose contains much of the required information; it just needs to be worked into the Sucrose article by someone who knows what they are doing. 121.45.110.194 ( talk) 09:40, 28 January 2008 (UTC) |
Last edited at 09:40, 28 January 2008 (UTC). Substituted at 07:14, 30 April 2016 (UTC)
On the nutrient label of any edible product, why is sugar the only nutrient ingredient without a daily nutrient value? Thank you M.cook — Preceding unsigned comment added by 65.94.195.17 ( talk) 18:06, 20 June 2016 (UTC)
Can someone publish the magnetic susceptibility of sucrose in the chembox section #2 physical properties? I'm having a hard time finding such simple data. If someone has a magnetic susceptibility balance it would be just a short calculation that could benefit everyone! Standard magnetic susceptibility units are either cm3/mol or cm3/g. Remember to fill in the correct units or place the data here and I can place it in the physical properties box for us. TerpeneOtto ( talk) 04:47, 7 December 2016 (UTC)
Someone asked me why sugar is so soluble in water. I had previously told them that ionic bonds generally resulted in things that were soluble in water, whereas covalent bonds generally resulted in things that were relatively insoluble in water. But they brought up sugar, and I guessed that its solubility had s.t. to do with the oxygens. I came here looking for an answer, but the problem is not discussed. Could someone who knows more about such things than I do add a paragraph on that? Looking elsewhere, it appears that it has to do with the OH groups on sugar, which are somewhat polar (as are the OH bonds in water). But a fuller explanation would be...well, fuller. Mcswell ( talk) 02:24, 1 May 2018 (UTC)
"digestion releases about 4 small calories per gram"
Doesn't a gram contain 4 regular kcal? Perhaps a historic unit like "small" should be avoided. -- J7n ( talk) 05:03, 24 February 2021 (UTC)
I see that the source is : "Prices from 1960 to 2010 are ICE spot sugar prices. Prices from 2011 onward are ICE contract 11 nearby futures prices. Data compiled by the USDA.". It would be useful if the graph could be annotated to say whether the prices are for 1 January, 30 June, maximum for the year, average for the year, or what. Exbrum ( talk) 10:55, 2 April 2021 (UTC)
The image currently* describing sucrose on the Main page shows a (#1C to #5C) linkage of glucose to fructose respectively. But it should show (#1C to #2C) linkage of glucose to fructose respectively.
- -> At time of writing that is 19 November 2021; 12:45 IST 2402:3A80:1818:654B:0:6:9D67:9B01 ( talk) 19:16, 18 November 2021 (UTC)
At the end of the summary article*, the chemical formula is malformed when seen in Android 10. Perhaps the author omitted trailing "end sub" digit delimiters in angle brackets. *Typically what shows up when there's an embedded ref. in another article Nikevich 22:38, 12 January 2022 (UTC)
- B-Class vital articles
- Wikipedia level-5 vital articles
- Wikipedia vital articles in Biology and health sciences
- B-Class level-5 vital articles
- Wikipedia level-5 vital articles in Biology and health sciences
- B-Class vital articles in Biology and health sciences
- B-Class chemicals articles
- High-importance chemicals articles
- B-Class Food and drink articles
- Mid-importance Food and drink articles
- WikiProject Food and drink articles
- B-Class Molecular Biology articles
- Unknown-importance Molecular Biology articles
- B-Class MCB articles
- Low-importance MCB articles
- WikiProject Molecular and Cellular Biology articles
- All WikiProject Molecular Biology pages



