| MOG | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | MOG, BTN6, BTNL11, MOGIG2, NRCLP7, myelin oligodendrocyte glycoprotein | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 159465; MGI: 97435; HomoloGene: 111009; GeneCards: MOG; OMA: MOG - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Myelin oligodendrocyte glycoprotein (MOG) is a glycoprotein believed to be important in the myelination of nerves in the central nervous system (CNS). In humans this protein is encoded by the MOG gene. [5] [6] [7] It is speculated to serve as a necessary "adhesion molecule" to provide structural integrity to the myelin sheath and is known to develop late on the oligodendrocyte. [8]
Molecular function
While the primary molecular function of MOG is not yet known, its likely role with the myelin sheath is either in sheath "completion and/or maintenance". [7] More specifically, MOG is speculated to be "necessary" as an "adhesion molecule" on the myelin sheath of the CNS to provide the structural integrity of the myelin sheath. [8]"
MOG's cDNA coding region in humans have been shown to be "highly homologous" [9] to rats, mice, and bovine, and hence highly conserved. This suggests "an important biological role for this protein". [7]
Physiology
The gene for MOG, found on chromosome 6 p21.3-p22, [10] was first sequenced in 1995. [3] It is a transmembrane protein expressed on the surface of oligodendrocyte cell and on the outermost surface of myelin sheaths. "MOG is a quantitatively minor type I transmembrane protein, [11] and is found exclusively in the CNS. "A single Ig-domain is exposed to the extracellular space" [11] and consequently allows autoantibodies easy access. and therefore is easily accessible to autoantibodies too. [7] [11] The MOG "primary nuclear transcript … is 15,561 nucleotides in length" [7] and, for humans, it has eight exons which are "separated by seven introns". [7] The introns "contain numerous reptitive [ sic] DNA [7]" sequences, among which are "14 Alu sequences within 3 introns", [7] and have a range varying from 242 to 6484 bp.
Structure
Alternatively spliced human mRNA of the MOG gene form at least nine isoforms. [12]
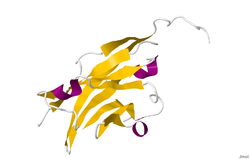
The crystal structure of myelin oligodendrocyte glycoprotein was determined by x-ray diffraction at a resolution of 1.45 Angstroms, using protein from the Norway rat. This protein is 139 residues long, and is a member of the immunoglobulin superfamily. [13] The dssp secondary structure of the protein is 6% helical and 43% beta sheet: there are three short helical segments and ten beta strands. [14] The beta strands are within two antiparallel beta sheets that form an immunoglobulin-like beta-sandwich fold. [15] Several features of the protein structure suggest MOG has a role as an "adhesin in the completion and/or compaction of the myelin sheath." There is a "significant strip" of electronegative charge beginning near the N-terminus and running about half the length of the molecule. Also, MOG was shown to dimerize in solution, and the shape complementarity index is high at the dimer interface, suggesting a "biologically relevant MOG dimer." [16]
Synthesis
Developmentally, MOG is formed "very late on oligodendrocytes and the myelin sheath". [8]
Role in disease
Non-inflammatory demyelinating diseases
Interest in MOG has centered on its role in demyelinating diseases. Some of them are not-inflammatory, such as adrenoleukodystrophy, vanishing white matter disease, and Rubella induced mental retardation. [17]
Anti-MOG associated inflammatory demyelinating diseases
MOG has received much of its laboratory attention in studies dealing with MS. Several studies have shown a role for antibodies against MOG in the pathogenesis of MS, [8] [18] though most of them were written before the discovery of NMO-IgG and the NMO spectrum of diseases.
Anti-MOG status is different depending whether it is measured by ELISA or by microarray ( CBA). The proper way to identify it is by microarray, reacting patient serum with living cells, and detecting the binding IgG via a fluorescent-labeled secondary antibody. [19]
In animal models
Animal models of MS, namely Experimental Autoimmune Encephalomyelitis (EAE) models, have shown that "MOG-specific EAE models (of different animal strains) display/mirror human multiple sclerosis", [8] but basically explains the part involved in the optic neuritis. [20] These models with anti-MOG antibodies have been investigated extensively and are considered the only antibodies with demyelinating capacity [8] but again, EAE pathology is closer to NMO and ADEM than to the confluent demyelination observed in MS.
Anti-MOG antibodies have been shown to behave similarly to AQP4 antibodies in animal models, [20] and are considered a biomarker against the MS diagnosis [21] [22]
In seronegative neuromyelitis optica
Anti-MOG autoimmunity has been found to be involved in most AQP4-seronegative NMO [23] [24] and also in optic neuritis and some fulminant forms of ADEM. [25] MOG antibodies in NMOSD are variable depending on the seropositivity status. [26]
In other conditions
The presence of anti-MOG autoantibodies has been associated with the following conditions [27]
- Most cases of aquaporin-4-seronegative neuromyelitis optica: NMO derived from an antiMOG associated encephalomyelitis, [28]
- Some cases of acute disseminated encephalomyelitis, specially the recurrent ones (MDEM) [29] and the fulminant courses [25]
- Some cases of multiple sclerosis [27]
- isolated optic neuritis or transverse myelitis [27]
References
- ^ a b c ENSG00000232641, ENSG00000137345, ENSG00000230885, ENSG00000236561, ENSG00000237834, ENSG00000204655, ENSG00000234623 GRCh38: Ensembl release 89: ENSG00000234096, ENSG00000232641, ENSG00000137345, ENSG00000230885, ENSG00000236561, ENSG00000237834, ENSG00000204655, ENSG00000234623 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000076439 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Pham-Dinh D, Della Gaspera B, Kerlero de Rosbo N, Dautigny A (September 1995). "Structure of the human myelin/oligodendrocyte glycoprotein gene and multiple alternative spliced isoforms". Genomics. 29 (2): 345–52. doi: 10.1006/geno.1995.9995. PMID 8666381.
- ^ Pham-Dinh D, Jones EP, Pitiot G, Della Gaspera B, Daubas P, Mallet J, Le Paslier D, Fischer Lindahl K, Dautigny A (1995). "Physical mapping of the human and mouse MOG gene at the distal end of the MHC class Ib region". Immunogenetics. 42 (5): 386–91. doi: 10.1007/bf00179400. PMID 7590972. S2CID 8310478.
- ^ a b c d e f g h Roth MP, Malfroy L, Offer C, Sevin J, Enault G, Borot N, Pontarotti P, Coppin H (July 1995). "The human myelin oligodendrocyte glycoprotein (MOG) gene: complete nucleotide sequence and structural characterization". Genomics. 28 (2): 241–50. doi: 10.1006/geno.1995.1137. PMID 8530032.
- ^ a b c d e f Berger, T., Innsbruck Medical University Dept. of Neurology interviewed by S. Gillooly, Nov. 24, 2008.
- ^ Pham-Dinh D, Allinquant B, Ruberg M, Della Gaspera B, Nussbaum JL, Dautigny A (December 1994). "Characterization and expression of the cDNA coding for the human myelin/oligodendrocyte glycoprotein". Journal of Neurochemistry. 63 (6): 2353–6. doi: 10.1046/j.1471-4159.1994.63062353.x. PMID 7964757. S2CID 2788720.
- ^ Pham-Dinh D, Mattei MG, Nussbaum JL, Roussel G, Pontarotti P, Roeckel N, Mather IH, Artzt K, Lindahl KF, Dautigny A (September 1993). "Myelin/oligodendrocyte glycoprotein is a member of a subset of the immunoglobulin superfamily encoded within the major histocompatibility complex". Proceedings of the National Academy of Sciences of the United States of America. 90 (17): 7990–4. Bibcode: 1993PNAS...90.7990P. doi: 10.1073/pnas.90.17.7990. PMC 47273. PMID 8367453.
- ^ a b c Berger T, Reindl M (August 2007). "Multiple sclerosis: disease biomarkers as indicated by pathophysiology". Journal of the Neurological Sciences. 259 (1–2): 21–6. doi: 10.1016/j.jns.2006.05.070. PMID 17367811. S2CID 23257594.
- ^ Boyle LH, Traherne JA, Plotnek G, Ward R, Trowsdale J (September 2007). "Splice variation in the cytoplasmic domains of myelin oligodendrocyte glycoprotein affects its cellular localisation and transport". Journal of Neurochemistry. 102 (6): 1853–62. doi: 10.1111/j.1471-4159.2007.04687.x. PMC 2156149. PMID 17573820.
- ^ Breithaupt C, Schubart A, Zander H, Skerra A, Huber R, Linington C, Jacob U (August 2003). "Structural insights into the antigenicity of myelin oligodendrocyte glycoprotein". Proceedings of the National Academy of Sciences of the United States of America. 100 (16): 9446–51. Bibcode: 2003PNAS..100.9446B. doi: 10.1073/pnas.1133443100. PMC 170938. PMID 12874380.
- ^ Kabsch W, Sander C (December 1983). "Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features". Biopolymers. 22 (12): 2577–637. doi: 10.1002/bip.360221211. PMID 6667333. S2CID 29185760.
- ^ Murzin AG, Brenner SE, Hubbard T, Chothia C (April 1995). "SCOP: a structural classification of proteins database for the investigation of sequences and structures". Journal of Molecular Biology. 247 (4): 536–40. doi: 10.1016/S0022-2836(05)80134-2. PMID 7723011.
- ^ Clements CS, Reid HH, Beddoe T, Tynan FE, Perugini MA, Johns TG, Bernard CC, Rossjohn J (September 2003). "The crystal structure of myelin oligodendrocyte glycoprotein, a key autoantigen in multiple sclerosis". Proceedings of the National Academy of Sciences of the United States of America. 100 (19): 11059–64. Bibcode: 2003PNAS..10011059C. doi: 10.1073/pnas.1833158100. PMC 196926. PMID 12960396.
- ^ Cong H, Jiang Y, Tien P (November 2011). "Identification of the myelin oligodendrocyte glycoprotein as a cellular receptor for rubella virus". Journal of Virology. 85 (21): 11038–47. doi: 10.1128/JVI.05398-11. PMC 3194935. PMID 21880773.
- ^ Berger T, Rubner P, Schautzer F, Egg R, Ulmer H, Mayringer I, Dilitz E, Deisenhammer F, Reindl M (July 2003). "Antimyelin antibodies as a predictor of clinically definite multiple sclerosis after a first demyelinating event". The New England Journal of Medicine. 349 (2): 139–45. doi: 10.1056/NEJMoa022328. PMID 12853586.
- ^ Ichiro Nakashima, Anti-myelin oligodendrocyte glycoprotein antibody in demyelinating diseases [1]
- ^ a b Kezuka T, Usui Y, Yamakawa N, Matsunaga Y, Matsuda R, Masuda M, Utsumi H, Tanaka K, Goto H (June 2012). "Relationship between NMO-antibody and anti-MOG antibody in optic neuritis". Journal of Neuro-Ophthalmology. 32 (2): 107–10. doi: 10.1097/WNO.0b013e31823c9b6c. PMID 22157536. S2CID 46667141.
- ^ Ketelslegers IA, Van Pelt DE, Bryde S, Neuteboom RF, Catsman-Berrevoets CE, Hamann D, Hintzen RQ (October 2015). "Anti-MOG antibodies plead against MS diagnosis in an Acquired Demyelinating Syndromes cohort". Multiple Sclerosis. 21 (12): 1513–20. doi: 10.1177/1352458514566666. PMID 25662345. S2CID 25321614.
- ^ Kitley J, Woodhall M, Waters P, Leite MI, Devenney E, Craig J, Palace J, Vincent A (September 2012). "Myelin-oligodendrocyte glycoprotein antibodies in adults with a neuromyelitis optica phenotype". Neurology. 79 (12): 1273–7. doi: 10.1212/WNL.0b013e31826aac4e. PMID 22914827. S2CID 855313.
- ^ Pröbstel AK, Rudolf G, Dornmair K, Collongues N, Chanson JB, Sanderson NS, Lindberg RL, Kappos L, de Seze J, Derfuss T (2015). "Anti-MOG antibodies are present in a subgroup of patients with a neuromyelitis optica phenotype". Journal of Neuroinflammation. 12 (1): 46. doi: 10.1186/s12974-015-0256-1. PMC 4359547. PMID 25889963.
- ^ CYNTHIA MCKELVEY, Press Report, What's the Role of Myelin Oligodendrocyte Glycoprotein in NMO? [2]
- ^ a b Di Pauli F, Höftberger R, Reindl M, Beer R, Rhomberg P, Schanda K, Sato D, Fujihara K, Lassmann H, Schmutzhard E, Berger T (December 2015). "Fulminant demyelinating encephalomyelitis: Insights from antibody studies and neuropathology". Neurology: Neuroimmunology & Neuroinflammation. 2 (6): e175. doi: 10.1212/NXI.0000000000000175. PMC 4635550. PMID 26587556.
- ^ Berger T, Reindl M (August 2015). "Antibody biomarkers in CNS demyelinating diseases - a long and winding road". European Journal of Neurology. 22 (8): 1162–8. doi: 10.1111/ene.12759. PMID 26010364. S2CID 39301229.
- ^ a b c Reindl M, Di Pauli F, Rostásy K, Berger T (August 2013). "The spectrum of MOG autoantibody-associated demyelinating diseases". Nature Reviews. Neurology. 9 (8): 455–61. doi: 10.1038/nrneurol.2013.118. PMID 23797245. S2CID 7219279.
- ^ Spadaro M, Gerdes LA, Mayer MC, Ertl-Wagner B, Laurent S, Krumbholz M, Breithaupt C, Högen T, Straube A, Giese A, Hohlfeld R, Lassmann H, Meinl E, Kümpfel T (March 2015). "Histopathology and clinical course of MOG-antibody-associated encephalomyelitis". Annals of Clinical and Translational Neurology. 2 (3): 295–301. doi: 10.1002/acn3.164. PMC 4369279. PMID 25815356.
- ^ Baumann M, Hennes EM, Schanda K, Karenfort M, Kornek B, Seidl R, Diepold K, Lauffer H, Marquardt I, Strautmanis J, Syrbe S, Vieker S, Höftberger R, Reindl M, Rostásy K (2016). "Children with multiphasic disseminated encephalomyelitis and antibodies to the myelin oligodendrocyte glycoprotein (MOG): Extending the spectrum of MOG antibody positive diseases". Multiple Sclerosis (Houndmills, Basingstoke, England). 22 (14): 1821–1829. doi: 10.1177/1352458516631038. PMID 26869530. S2CID 30428892.
External links
| MOG | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | MOG, BTN6, BTNL11, MOGIG2, NRCLP7, myelin oligodendrocyte glycoprotein | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 159465; MGI: 97435; HomoloGene: 111009; GeneCards: MOG; OMA: MOG - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Myelin oligodendrocyte glycoprotein (MOG) is a glycoprotein believed to be important in the myelination of nerves in the central nervous system (CNS). In humans this protein is encoded by the MOG gene. [5] [6] [7] It is speculated to serve as a necessary "adhesion molecule" to provide structural integrity to the myelin sheath and is known to develop late on the oligodendrocyte. [8]
Molecular function
While the primary molecular function of MOG is not yet known, its likely role with the myelin sheath is either in sheath "completion and/or maintenance". [7] More specifically, MOG is speculated to be "necessary" as an "adhesion molecule" on the myelin sheath of the CNS to provide the structural integrity of the myelin sheath. [8]"
MOG's cDNA coding region in humans have been shown to be "highly homologous" [9] to rats, mice, and bovine, and hence highly conserved. This suggests "an important biological role for this protein". [7]
Physiology
The gene for MOG, found on chromosome 6 p21.3-p22, [10] was first sequenced in 1995. [3] It is a transmembrane protein expressed on the surface of oligodendrocyte cell and on the outermost surface of myelin sheaths. "MOG is a quantitatively minor type I transmembrane protein, [11] and is found exclusively in the CNS. "A single Ig-domain is exposed to the extracellular space" [11] and consequently allows autoantibodies easy access. and therefore is easily accessible to autoantibodies too. [7] [11] The MOG "primary nuclear transcript … is 15,561 nucleotides in length" [7] and, for humans, it has eight exons which are "separated by seven introns". [7] The introns "contain numerous reptitive [ sic] DNA [7]" sequences, among which are "14 Alu sequences within 3 introns", [7] and have a range varying from 242 to 6484 bp.
Structure
Alternatively spliced human mRNA of the MOG gene form at least nine isoforms. [12]
The crystal structure of myelin oligodendrocyte glycoprotein was determined by x-ray diffraction at a resolution of 1.45 Angstroms, using protein from the Norway rat. This protein is 139 residues long, and is a member of the immunoglobulin superfamily. [13] The dssp secondary structure of the protein is 6% helical and 43% beta sheet: there are three short helical segments and ten beta strands. [14] The beta strands are within two antiparallel beta sheets that form an immunoglobulin-like beta-sandwich fold. [15] Several features of the protein structure suggest MOG has a role as an "adhesin in the completion and/or compaction of the myelin sheath." There is a "significant strip" of electronegative charge beginning near the N-terminus and running about half the length of the molecule. Also, MOG was shown to dimerize in solution, and the shape complementarity index is high at the dimer interface, suggesting a "biologically relevant MOG dimer." [16]
Synthesis
Developmentally, MOG is formed "very late on oligodendrocytes and the myelin sheath". [8]
Role in disease
Non-inflammatory demyelinating diseases
Interest in MOG has centered on its role in demyelinating diseases. Some of them are not-inflammatory, such as adrenoleukodystrophy, vanishing white matter disease, and Rubella induced mental retardation. [17]
Anti-MOG associated inflammatory demyelinating diseases
MOG has received much of its laboratory attention in studies dealing with MS. Several studies have shown a role for antibodies against MOG in the pathogenesis of MS, [8] [18] though most of them were written before the discovery of NMO-IgG and the NMO spectrum of diseases.
Anti-MOG status is different depending whether it is measured by ELISA or by microarray ( CBA). The proper way to identify it is by microarray, reacting patient serum with living cells, and detecting the binding IgG via a fluorescent-labeled secondary antibody. [19]
In animal models
Animal models of MS, namely Experimental Autoimmune Encephalomyelitis (EAE) models, have shown that "MOG-specific EAE models (of different animal strains) display/mirror human multiple sclerosis", [8] but basically explains the part involved in the optic neuritis. [20] These models with anti-MOG antibodies have been investigated extensively and are considered the only antibodies with demyelinating capacity [8] but again, EAE pathology is closer to NMO and ADEM than to the confluent demyelination observed in MS.
Anti-MOG antibodies have been shown to behave similarly to AQP4 antibodies in animal models, [20] and are considered a biomarker against the MS diagnosis [21] [22]
In seronegative neuromyelitis optica
Anti-MOG autoimmunity has been found to be involved in most AQP4-seronegative NMO [23] [24] and also in optic neuritis and some fulminant forms of ADEM. [25] MOG antibodies in NMOSD are variable depending on the seropositivity status. [26]
In other conditions
The presence of anti-MOG autoantibodies has been associated with the following conditions [27]
- Most cases of aquaporin-4-seronegative neuromyelitis optica: NMO derived from an antiMOG associated encephalomyelitis, [28]
- Some cases of acute disseminated encephalomyelitis, specially the recurrent ones (MDEM) [29] and the fulminant courses [25]
- Some cases of multiple sclerosis [27]
- isolated optic neuritis or transverse myelitis [27]
References
- ^ a b c ENSG00000232641, ENSG00000137345, ENSG00000230885, ENSG00000236561, ENSG00000237834, ENSG00000204655, ENSG00000234623 GRCh38: Ensembl release 89: ENSG00000234096, ENSG00000232641, ENSG00000137345, ENSG00000230885, ENSG00000236561, ENSG00000237834, ENSG00000204655, ENSG00000234623 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000076439 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Pham-Dinh D, Della Gaspera B, Kerlero de Rosbo N, Dautigny A (September 1995). "Structure of the human myelin/oligodendrocyte glycoprotein gene and multiple alternative spliced isoforms". Genomics. 29 (2): 345–52. doi: 10.1006/geno.1995.9995. PMID 8666381.
- ^ Pham-Dinh D, Jones EP, Pitiot G, Della Gaspera B, Daubas P, Mallet J, Le Paslier D, Fischer Lindahl K, Dautigny A (1995). "Physical mapping of the human and mouse MOG gene at the distal end of the MHC class Ib region". Immunogenetics. 42 (5): 386–91. doi: 10.1007/bf00179400. PMID 7590972. S2CID 8310478.
- ^ a b c d e f g h Roth MP, Malfroy L, Offer C, Sevin J, Enault G, Borot N, Pontarotti P, Coppin H (July 1995). "The human myelin oligodendrocyte glycoprotein (MOG) gene: complete nucleotide sequence and structural characterization". Genomics. 28 (2): 241–50. doi: 10.1006/geno.1995.1137. PMID 8530032.
- ^ a b c d e f Berger, T., Innsbruck Medical University Dept. of Neurology interviewed by S. Gillooly, Nov. 24, 2008.
- ^ Pham-Dinh D, Allinquant B, Ruberg M, Della Gaspera B, Nussbaum JL, Dautigny A (December 1994). "Characterization and expression of the cDNA coding for the human myelin/oligodendrocyte glycoprotein". Journal of Neurochemistry. 63 (6): 2353–6. doi: 10.1046/j.1471-4159.1994.63062353.x. PMID 7964757. S2CID 2788720.
- ^ Pham-Dinh D, Mattei MG, Nussbaum JL, Roussel G, Pontarotti P, Roeckel N, Mather IH, Artzt K, Lindahl KF, Dautigny A (September 1993). "Myelin/oligodendrocyte glycoprotein is a member of a subset of the immunoglobulin superfamily encoded within the major histocompatibility complex". Proceedings of the National Academy of Sciences of the United States of America. 90 (17): 7990–4. Bibcode: 1993PNAS...90.7990P. doi: 10.1073/pnas.90.17.7990. PMC 47273. PMID 8367453.
- ^ a b c Berger T, Reindl M (August 2007). "Multiple sclerosis: disease biomarkers as indicated by pathophysiology". Journal of the Neurological Sciences. 259 (1–2): 21–6. doi: 10.1016/j.jns.2006.05.070. PMID 17367811. S2CID 23257594.
- ^ Boyle LH, Traherne JA, Plotnek G, Ward R, Trowsdale J (September 2007). "Splice variation in the cytoplasmic domains of myelin oligodendrocyte glycoprotein affects its cellular localisation and transport". Journal of Neurochemistry. 102 (6): 1853–62. doi: 10.1111/j.1471-4159.2007.04687.x. PMC 2156149. PMID 17573820.
- ^ Breithaupt C, Schubart A, Zander H, Skerra A, Huber R, Linington C, Jacob U (August 2003). "Structural insights into the antigenicity of myelin oligodendrocyte glycoprotein". Proceedings of the National Academy of Sciences of the United States of America. 100 (16): 9446–51. Bibcode: 2003PNAS..100.9446B. doi: 10.1073/pnas.1133443100. PMC 170938. PMID 12874380.
- ^ Kabsch W, Sander C (December 1983). "Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features". Biopolymers. 22 (12): 2577–637. doi: 10.1002/bip.360221211. PMID 6667333. S2CID 29185760.
- ^ Murzin AG, Brenner SE, Hubbard T, Chothia C (April 1995). "SCOP: a structural classification of proteins database for the investigation of sequences and structures". Journal of Molecular Biology. 247 (4): 536–40. doi: 10.1016/S0022-2836(05)80134-2. PMID 7723011.
- ^ Clements CS, Reid HH, Beddoe T, Tynan FE, Perugini MA, Johns TG, Bernard CC, Rossjohn J (September 2003). "The crystal structure of myelin oligodendrocyte glycoprotein, a key autoantigen in multiple sclerosis". Proceedings of the National Academy of Sciences of the United States of America. 100 (19): 11059–64. Bibcode: 2003PNAS..10011059C. doi: 10.1073/pnas.1833158100. PMC 196926. PMID 12960396.
- ^ Cong H, Jiang Y, Tien P (November 2011). "Identification of the myelin oligodendrocyte glycoprotein as a cellular receptor for rubella virus". Journal of Virology. 85 (21): 11038–47. doi: 10.1128/JVI.05398-11. PMC 3194935. PMID 21880773.
- ^ Berger T, Rubner P, Schautzer F, Egg R, Ulmer H, Mayringer I, Dilitz E, Deisenhammer F, Reindl M (July 2003). "Antimyelin antibodies as a predictor of clinically definite multiple sclerosis after a first demyelinating event". The New England Journal of Medicine. 349 (2): 139–45. doi: 10.1056/NEJMoa022328. PMID 12853586.
- ^ Ichiro Nakashima, Anti-myelin oligodendrocyte glycoprotein antibody in demyelinating diseases [1]
- ^ a b Kezuka T, Usui Y, Yamakawa N, Matsunaga Y, Matsuda R, Masuda M, Utsumi H, Tanaka K, Goto H (June 2012). "Relationship between NMO-antibody and anti-MOG antibody in optic neuritis". Journal of Neuro-Ophthalmology. 32 (2): 107–10. doi: 10.1097/WNO.0b013e31823c9b6c. PMID 22157536. S2CID 46667141.
- ^ Ketelslegers IA, Van Pelt DE, Bryde S, Neuteboom RF, Catsman-Berrevoets CE, Hamann D, Hintzen RQ (October 2015). "Anti-MOG antibodies plead against MS diagnosis in an Acquired Demyelinating Syndromes cohort". Multiple Sclerosis. 21 (12): 1513–20. doi: 10.1177/1352458514566666. PMID 25662345. S2CID 25321614.
- ^ Kitley J, Woodhall M, Waters P, Leite MI, Devenney E, Craig J, Palace J, Vincent A (September 2012). "Myelin-oligodendrocyte glycoprotein antibodies in adults with a neuromyelitis optica phenotype". Neurology. 79 (12): 1273–7. doi: 10.1212/WNL.0b013e31826aac4e. PMID 22914827. S2CID 855313.
- ^ Pröbstel AK, Rudolf G, Dornmair K, Collongues N, Chanson JB, Sanderson NS, Lindberg RL, Kappos L, de Seze J, Derfuss T (2015). "Anti-MOG antibodies are present in a subgroup of patients with a neuromyelitis optica phenotype". Journal of Neuroinflammation. 12 (1): 46. doi: 10.1186/s12974-015-0256-1. PMC 4359547. PMID 25889963.
- ^ CYNTHIA MCKELVEY, Press Report, What's the Role of Myelin Oligodendrocyte Glycoprotein in NMO? [2]
- ^ a b Di Pauli F, Höftberger R, Reindl M, Beer R, Rhomberg P, Schanda K, Sato D, Fujihara K, Lassmann H, Schmutzhard E, Berger T (December 2015). "Fulminant demyelinating encephalomyelitis: Insights from antibody studies and neuropathology". Neurology: Neuroimmunology & Neuroinflammation. 2 (6): e175. doi: 10.1212/NXI.0000000000000175. PMC 4635550. PMID 26587556.
- ^ Berger T, Reindl M (August 2015). "Antibody biomarkers in CNS demyelinating diseases - a long and winding road". European Journal of Neurology. 22 (8): 1162–8. doi: 10.1111/ene.12759. PMID 26010364. S2CID 39301229.
- ^ a b c Reindl M, Di Pauli F, Rostásy K, Berger T (August 2013). "The spectrum of MOG autoantibody-associated demyelinating diseases". Nature Reviews. Neurology. 9 (8): 455–61. doi: 10.1038/nrneurol.2013.118. PMID 23797245. S2CID 7219279.
- ^ Spadaro M, Gerdes LA, Mayer MC, Ertl-Wagner B, Laurent S, Krumbholz M, Breithaupt C, Högen T, Straube A, Giese A, Hohlfeld R, Lassmann H, Meinl E, Kümpfel T (March 2015). "Histopathology and clinical course of MOG-antibody-associated encephalomyelitis". Annals of Clinical and Translational Neurology. 2 (3): 295–301. doi: 10.1002/acn3.164. PMC 4369279. PMID 25815356.
- ^ Baumann M, Hennes EM, Schanda K, Karenfort M, Kornek B, Seidl R, Diepold K, Lauffer H, Marquardt I, Strautmanis J, Syrbe S, Vieker S, Höftberger R, Reindl M, Rostásy K (2016). "Children with multiphasic disseminated encephalomyelitis and antibodies to the myelin oligodendrocyte glycoprotein (MOG): Extending the spectrum of MOG antibody positive diseases". Multiple Sclerosis (Houndmills, Basingstoke, England). 22 (14): 1821–1829. doi: 10.1177/1352458516631038. PMID 26869530. S2CID 30428892.




