 | |
| Clinical data | |
|---|---|
| Trade names | Jardiance, others |
| Other names | BI-10773 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a614043 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | By mouth |
| Drug class | Sodium-glucose cotransporter-2 (SGLT2) inhibitor [2] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.122.058 |
| Chemical and physical data | |
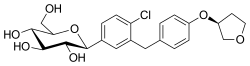
| Formula | C23H27ClO7 |
| Molar mass | 450.91 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Empagliflozin, sold under the brand name Jardiance, among others, is an antidiabetic medication used to improve glucose control in people with type 2 diabetes. [10] [2] [12] It is taken by mouth. [2]
Common side effects include hyperventilation, anorexia, abdominal pain, nausea, vomiting, lethargy, mental status changes, hypotension, acute kidney injury, and vaginal yeast infections. [2] Rarer but more serious side effects include a skin infection of the groin called Fournier's gangrene and a form of diabetic ketoacidosis with normal blood sugar levels. [2] [13] Use during pregnancy or breastfeeding is not recommended. [14] Empagliflozin sometimes causes a transient decline in kidney function, and on rare occasions causes acute kidney injury, so use should be monitored in those with kidney dysfunction. But some trials have indicated that empagliflozin can be used in people with an eGFR as low as 20 mL/min/1.73 m², without increasing adverse kidney outcomes. [15] [16]
The use of empagliflozin has been shown to improve outcomes in people with established cardiovascular disease. [17] [15] There is evidence from high quality studies that empagliflozin can also help to slow the rate of kidney function decline. Irrespective of diabetes status, benefit was observed in those with mild, moderate or severe loss of kidney function. [18] [19] People started on empagliflozin may first see a decrease in kidney function before their glomerular filtration rate stabilises. [20] Greatest benefit was demonstrated in those who had severe loss of kidney function, higher risk of kidney function worsening and background of diabetes. [21]
Empagliflozin is an inhibitor of the sodium glucose co-transporter-2 (SGLT-2), and works by increasing sugar loss in urine. [2]
Empagliflozin was approved for medical use in the United States and in the European Union in 2014. [11] [22] [23] It is on the World Health Organization's List of Essential Medicines. [24] In 2021, it was the 85th most commonly prescribed medication in the United States, with more than 8 million prescriptions. [25] [26] It has received approval as a generic medication from the US Food and Drug Administration (FDA). [27]
Empagliflozin lowers risk of hospitalisation and death in patients with reduced heart function, when added to standard heart failure treatment with or without type 2 diabetes. [28] [29] [30] It is indicated in adults with type 2 diabetes and established cardiovascular disease to reduce the risk of cardiovascular death; and as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. [10] [31] [32]
In June 2023, the US Food and Drug Administration (FDA) expanded the indication, as an addition to diet and exercise, to improve blood sugar control in children 10 years and older with type 2 diabetes. [33]
- History of a severe allergic reaction to empagliflozin [10]
- End-stage kidney disease [10]
- Diabetic ketoacidosis [10]
- Empagliflozin increases the risk of genital fungal infections. The risk is highest in people with a prior history of genital fungal infections. [34]
- Empagliflozin has been thought to be associated with increased risk of urinary tract infections. Reviews of clinical trials have shown there is no significant risk of developing urinary tract infections while taking empagliflozin when compared to placebo or other diabetic medications. [35] [36]
- Empagliflozin reduces systolic and diastolic blood pressure and can increase the risk of low blood pressure, which can cause fainting and/or falls. [34] The risk is higher in older people, people taking diuretics, and people with reduced kidney function. [34]
- Slight increases in Low-density lipoprotein (LDL) cholesterol can be seen with empagliflozin, in the range of 2–4% from baseline. [34]
- Diabetic ketoacidosis, a rare but potentially life-threatening condition, may occur more commonly with empagliflozin and other SGLT-2 inhibitors. [37] [38] While diabetic ketoacidosis is usually associated with elevated blood glucose levels, in people taking SGLT-2 inhibitors diabetic ketoacidosis may be seen with uncharacteristically normal blood glucose levels, a phenomenon called euglycemic diabetic ketoacidosis. [37] The absence of elevated blood glucose levels in people on an SGLT-2 inhibitor may make it more difficult to diagnose diabetic ketoacidosis. The risk of empagliflozin-associated euglycemic diabetic ketoacidosis may be higher in the setting of illness, dehydration, surgery, and/or alcohol consumption. [37] It is also seen in type 1 diabetes who take empagliflozin, which notably is an unapproved or "off-label" use of the medication. [38] To lessen the risk of developing ketoacidosis (a serious condition in which the body produces high levels of blood acids called ketones) after surgery, the FDA has approved changes to the prescribing information for SGLT2 inhibitor diabetes medicines to recommend they be stopped temporarily before scheduled surgery. Empagliflozin should each be stopped at least three days before scheduled surgery. [39] Symptoms of diabetic ketoacidosis include nausea, vomiting, abdominal pain, tiredness, and trouble breathing. [39]
- Fournier's gangrene, a rare but serious infection of the groin, occurs more commonly in people taking empagliflozin and other SGLT-2 inhibitors. [2] [13] Symptoms include feverishness, a general sense of malaise, and pain or swelling around the genitals or in the skin behind them. The infection progresses quickly and urgent medical attention is recommended. [13]
- Empagliflozin can increase the risk of low blood sugar when it is used together with a sulfonylurea or insulin. [40] When used by itself or in addition to metformin it does not appear to increase the risk of hypoglycemia. [41]
Empagliflozin is an inhibitor of the sodium glucose co-transporter-2 (SGLT-2), which is found almost exclusively in the proximal tubules of nephronic components in the kidneys. SGLT-2 accounts for about 90 percent of glucose reabsorption into the blood. Blocking SGLT-2 reduces blood glucose by blocking glucose reabsorption in the kidney and thereby excreting glucose (i.e., blood sugar) via the urine. [42] [43] [44] Of all the SGLT-2 Inhibitors currently available, empagliflozin has the highest degree of selectivity for SGLT-2 over SGLT-1, SGLT-4, SGLT-5 and SGLT-6. [45]
It was developed by Boehringer Ingelheim and is co-marketed by Eli Lilly and Company. It is also available as the combinations empagliflozin/linagliptin, empagliflozin/metformin, and empagliflozin/linagliptin/metformin.
For cardiovascular death, the FDA based its decision on a postmarketing study it required when it approved empagliflozin in 2014 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. [22] [31] Empagliflozin was studied in a postmarket clinical trial of more than 7,000 participants with type 2 diabetes and cardiovascular disease. [31] In the trial, empagliflozin was shown to reduce the risk of cardiovascular death compared to a placebo when added to standard of care therapies for diabetes and atherosclerotic cardiovascular disease. [31]
For heart failure, the safety and effectiveness of empagliflozin were evaluated by the FDA as an adjunct to standard of care therapy in a randomized, double-blind, international trial comparing 2,997 participants who received empagliflozin, 10 mg, once daily to 2,991 participants who received the placebo. [32] The main efficacy measurement was the time to death from cardiovascular causes or need to be hospitalized for heart failure. [32] Of the individuals who received empagliflozin for an average of about two years, 14% died from cardiovascular causes or were hospitalized for heart failure, compared to 17% of the participants who received the placebo. [32] This benefit was mostly attributable to fewer participants being hospitalized for heart failure. [32]
The FDA granted the application for empagliflozin priority review and granted the approval of Jardiance to Boehringer Ingelheim. [32]
As of May 2013, Boehringer and Lilly had submitted applications for marketing approval to the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA). [46] The drug was approved in the European Union in May 2014, [11] and was approved in the United States in August 2014. [22] [23] [47] The FDA required four postmarketing studies: a cardiovascular outcomes trial, two studies in children, and a toxicity study in animals related to the pediatric trials. [22] [47]
A meta-analysis of short-term randomised controlled trials has shown similar efficacy on glycaemic control between empagliflozin 10mg and 25mg in patients with type 2 diabetes. Whilst there may be a higher reduction in HbA1c with higher doses, this difference is more clinically significant when the patients baseline HbA1c is higher e.g. those with a HbA1c ≥ 8.5%. Therefore patients’ baseline values, such as HbA1c, BMI etc, should be taken into account when prescribing high dose empagliflozin. [48] [49]
Empagliflozin causes moderate reductions in blood pressure and body weight. These effects are likely due to the excretion of glucose in the urine and a slight increase in urinary sodium excretion. [34] [50]
In clinical trials, participants with type 2 diabetes taking empagliflozin with other diabetic medications lost an average of 2% of their baseline body weight. [51] [52] A higher percentage of people taking empagliflozin achieved weight loss greater than 5% from their baseline, which has been associated with improved glucose control. [51] [52] [34] The same extent of weight loss was also observed in a study with heart failure patients taking empagliflozin. [53]
Empagliflozin has been shown to reduce systolic blood pressure by 3 to 5 millimeters of mercury (mmHg) without changes in pulse rate. [51] [52] [34] A greater percentage of people with uncontrolled blood pressure at baseline, achieved controlled blood pressure (i.e. systolic blood pressure <130 mmHg and diastolic blood pressure <80 mmHg) after taking empagliflozin at 24 weeks. [52] The effects on blood pressure and body weight are generally viewed as favorable, as many people with type 2 diabetes have high blood pressure or are overweight or obese. [41] [54]
- ^ "Empagliflozin (Jardiance) Use During Pregnancy". Drugs.com. 30 August 2018. Archived from the original on 5 August 2019. Retrieved 10 February 2020.
- ^ a b c d e f g "Empagliflozin Monograph for Professionals". Drugs.com. AHFS. Archived from the original on 6 April 2019. Retrieved 21 December 2018.
- ^ "AusPAR: Empagliflozin". Therapeutic Goods Administration (TGA), Commonwealth of Australia. 8 November 2017. Retrieved 24 March 2022.
- ^ "JARDIANCE". Boehringer Ingelheim Pty Ltd. Therapeutic Goods Administration (TGA), Commonwealth of Australia. Archived from the original on 18 March 2023.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2014". Therapeutic Goods Administration (TGA), Commonwealth of Australia. 21 June 2022. Archived from the original on 10 April 2023. Retrieved 10 April 2023.
- ^ "Prescription medicines and biologicals: TGA annual summary 2017". Therapeutic Goods Administration (TGA), Commonwealth of Australia. 21 June 2022. Retrieved 31 March 2024.
- ^ "Health Canada New Drug Authorizations: 2015 Highlights". Health Canada. 4 May 2016. Retrieved 7 April 2024.
- ^ "Jardiance 10 mg film-coated tablets – Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 20 September 2020. Retrieved 10 February 2020.
- ^ "Jardiance 25 mg film-coated tablets – Summary of Product Characteristics (SmPC)". (emc). 23 October 2019. Archived from the original on 22 September 2020. Retrieved 10 February 2020.
- ^ a b c d e f "Jardiance- empagliflozin tablet, film coated". DailyMed. 22 January 2020. Archived from the original on 11 February 2020. Retrieved 10 February 2020.
- ^ a b c "Jardiance EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 28 August 2021. Retrieved 10 February 2020.
- ^ Davies MJ, D'Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, et al. (December 2018). "Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD)". Diabetologia. 61 (12): 2461–2498. doi: 10.1007/s00125-018-4729-5. PMID 30288571.
- ^ a b c "FDA warns about rare occurrences of a serious infection of the genital area with SGLT2 inhibitors for diabetes". U.S. Food and Drug Administration (FDA). 9 February 2019. Archived from the original on 13 December 2019. Retrieved 18 March 2019.
- ^ British national formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 691. ISBN 9780857113382.
- ^ a b Zannad F, Butler J, Filippatos GS, Pocock S, Jamal W, Schnee J, et al. (May 2021). "Cardiovascular and Kidney Outcomes with Empagliflozin in Heart Failure". Präzisionsmedizin – Eine Reise in die Zukunft der Diabetologie www.diabeteskongress.de. 16. Georg Thieme Verlag KG. doi: 10.1055/s-0041-1727471.
- ^ Herrington WG, Staplin N, Wanner C, Green JB, Hauske SJ, Emberson JR, et al. (January 2023). "Empagliflozin in Patients with Chronic Kidney Disease". The New England Journal of Medicine. 388 (2): 117–127. doi: 10.1056/NEJMoa2204233. PMC 7614055. PMID 36331190.
- ^ Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. (November 2015). "Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes". The New England Journal of Medicine. 373 (22): 2117–2128. doi: 10.1056/NEJMoa1504720. PMID 26378978.
- ^ Herrington WG, Preiss D, Haynes R, von Eynatten M, Staplin N, Hauske SJ, et al. (August 2020). "Erratum: The potential for improving cardio-renal outcomes by sodium-glucose co-transporter-2 inhibition in people with chronic kidney disease: a rationale for the EMPA-KIDNEY study". Clinical Kidney Journal. 13 (4): 722. doi: 10.1056/NEJMoa2204233. hdl: 20.500.13003/18576. PMC 7467589. PMID 32905262.
- ^ Wanner C, Inzucchi SE, Lachin JM, Fitchett D, von Eynatten M, Mattheus M, et al. (July 2016). "Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes". The New England Journal of Medicine. 375 (4): 323–334. doi: 10.1056/nejmoa1515920. PMID 27299675.
- ^ Wanner C, Inzucchi SE, Lachin JM, Fitchett D, von Eynatten M, Mattheus M, et al. (July 2016). "Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes". The New England Journal of Medicine. 375 (4): 323–334. doi: 10.1056/NEJMoa1515920. PMID 27299675.
- ^ Herrington WG, Preiss D, Haynes R, von Eynatten M, Staplin N, Hauske SJ, et al. (August 2020). "Erratum: The potential for improving cardio-renal outcomes by sodium-glucose co-transporter-2 inhibition in people with chronic kidney disease: a rationale for the EMPA-KIDNEY study". Clinical Kidney Journal. 13 (4): 722. doi: 10.1056/NEJMoa2204233. hdl: 20.500.13003/18576. PMC 7467589. PMID 32905262.
- ^
a
b
c
d
"FDA approves Jardiance to treat type 2 diabetes" (Press release). U.S.
Food and Drug Administration (FDA). 1 August 2014. Archived from
the original on 22 October 2016. Retrieved 5 February 2023.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ a b "Drug Approval Package: Jardiance (empagliflozin) Tablets NDA #204629". U.S. Food and Drug Administration (FDA). 8 September 2014. Archived from the original on 11 February 2020. Retrieved 10 February 2020.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl: 10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Empagliflozin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "2022 First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). 3 March 2023. Archived from the original on 14 January 2024. Retrieved 14 January 2024.
- ^ Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, et al. (October 2020). "Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure". The New England Journal of Medicine. 383 (15): 1413–1424. doi: 10.1056/NEJMoa2022190. hdl: 2066/230126. PMID 32865377.
- ^ Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, et al. (October 2021). "Empagliflozin in Heart Failure with a Preserved Ejection Fraction". The New England Journal of Medicine. 385 (16): 1451–1461. doi: 10.1056/NEJMoa2107038. PMID 34449189.
- ^ Zannad F, Ferreira JP, Pocock SJ, Anker SD, Butler J, Filippatos G, et al. (September 2020). "SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-Reduced and DAPA-HF trials". Lancet. 396 (10254): 819–829. doi: 10.1016/s0140-6736(20)31824-9. PMID 32877652.
- ^
a
b
c
d
"FDA approves Jardiance to reduce cardiovascular death in adults with type 2 diabetes" (Press release). U.S.
Food and Drug Administration (FDA). 6 December 2016.
Archived from the original on 11 February 2020. Retrieved 12 December 2016.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^
a
b
c
d
e
f
"FDA Approves Treatment for Wider Range of Patients with Heart Failure" (Press release). U.S.
Food and Drug Administration (FDA). 24 February 2022.
Archived from the original on 27 February 2022. Retrieved 27 February 2022.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ "FDA Approves New Class of Medicines to Treat Pediatric Type 2 Diabetes". U.S. Food and Drug Administration (FDA) (Press release). 20 June 2023. Archived from the original on 21 June 2023. Retrieved 20 June 2023.
- ^ a b c d e f g "Empagliflozin (Jardiance) National Drug Monograph" (PDF). U.S. Department of Veterans Affairs. October 2015. Archived from the original (PDF) on 18 March 2021.
- ^ Donnan JR, Grandy CA, Chibrikov E, Marra CA, Aubrey-Bassler K, Johnston K, et al. (February 2019). "Comparative safety of the sodium glucose co-transporter 2 (SGLT2) inhibitors: a systematic review and meta-analysis". BMJ Open. 9 (1): e022577. doi: 10.1136/bmjopen-2018-022577. PMC 6361337. PMID 30813108.
- ^ Kinduryte Schorling O, Clark D, Zwiener I, Kaspers S, Lee J, Iliev H (August 2020). "Pooled Safety and Tolerability Analysis of Empagliflozin in Patients with Type 2 Diabetes Mellitus". Advances in Therapy. 37 (8): 3463–3484. doi: 10.1007/s12325-020-01329-7. PMC 7370973. PMID 32372290.
- ^ a b c Rosenstock J, Ferrannini E (September 2015). "Euglycemic Diabetic Ketoacidosis: A Predictable, Detectable, and Preventable Safety Concern With SGLT2 Inhibitors". Diabetes Care. 38 (9): 1638–1642. doi: 10.2337/dc15-1380. PMID 26294774.
- ^ a b Handelsman Y, Henry RR, Bloomgarden ZT, Dagogo-Jack S, DeFronzo RA, Einhorn D, et al. (June 2016). "American Association of Clinical Endocrinologists and American College of Endocrinology Position Statement on the Association of Sglt-2 Inhibitors and Diabetic Ketoacidosis". Endocrine Practice. 22 (6): 753–762. doi: 10.4158/EP161292.PS. PMID 27082665.
- ^
a
b
"FDA revises labels of SGLT2 inhibitors for diabetes to include warning". U.S.
Food and Drug Administration (FDA). 19 March 2020.
Archived from the original on 7 June 2020. Retrieved 6 June 2020.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ "Empagliflozin in combination therapy for treating type 2 diabetes". NICE. 25 March 2015. Archived from the original on 21 December 2018. Retrieved 21 December 2018.
- ^ a b Anderson JE, Wright EE, Shaefer CF (February 2017). "Empagliflozin: Role in Treatment Options for Patients with Type 2 Diabetes Mellitus". Diabetes Therapy. 8 (1): 33–53. doi: 10.1007/s13300-016-0211-x. PMC 5306110. PMID 27837465.
- ^ Abdul-Ghani MA, DeFronzo RA (September 2008). "Inhibition of renal glucose reabsorption: a novel strategy for achieving glucose control in type 2 diabetes mellitus". Endocrine Practice. 14 (6): 782–790. doi: 10.4158/ep.14.6.782. PMID 18996802.
- ^ Nair S, Wilding JP (January 2010). "Sodium glucose cotransporter 2 inhibitors as a new treatment for diabetes mellitus". The Journal of Clinical Endocrinology and Metabolism. 95 (1): 34–42. doi: 10.1210/jc.2009-0473. PMID 19892839.
- ^ Bays H (March 2009). "From victim to ally: the kidney as an emerging target for the treatment of diabetes mellitus". Current Medical Research and Opinion. 25 (3): 671–681. doi: 10.1185/03007990802710422. PMID 19232040. S2CID 73341491.
- ^ Tentolouris A, Vlachakis P, Tzeravini E, Eleftheriadou I, Tentolouris N (August 2019). "SGLT2 Inhibitors: A Review of Their Antidiabetic and Cardioprotective Effects". International Journal of Environmental Research and Public Health. 16 (16): 2965. doi: 10.3390/ijerph16162965. PMC 6720282. PMID 31426529.
- ^ Tucker ME (7 May 2013). "First details of empagliflozin trials follow US and EU filings". Medscape Medical News. Archived from the original on 18 June 2013. Retrieved 18 January 2014.
- ^ a b Mechatie E (1 August 2014). "FDA approves empagliflozin for adults with type 2 diabetes". Clinical Endocrinology News Digital Network. Archived from the original on 28 August 2021. Retrieved 23 August 2014.
- ^ Liakos A, Karagiannis T, Athanasiadou E, Sarigianni M, Mainou M, Papatheodorou K, et al. (October 2014). "Efficacy and safety of empagliflozin for type 2 diabetes: a systematic review and meta-analysis". Diabetes, Obesity & Metabolism. 16 (10): 984–993. doi: 10.1111/dom.12307. PMID 24766495.
- ^ Inzucchi SE, Davies MJ, Khunti K, Trivedi P, George JT, Zwiener I, et al. (February 2021). "Empagliflozin treatment effects across categories of baseline HbA1c, body weight and blood pressure as an add-on to metformin in patients with type 2 diabetes". Diabetes, Obesity & Metabolism. 23 (2): 425–433. doi: 10.1111/dom.14234. PMC 7839733. PMID 33084149.
- ^ Verma S, McMurray JJ (October 2018). "SGLT2 inhibitors and mechanisms of cardiovascular benefit: a state-of-the-art review". Diabetologia. 61 (10): 2108–2117. doi: 10.1007/s00125-018-4670-7. PMID 30132036.
- ^ a b c Kovacs CS, Seshiah V, Swallow R, Jones R, Rattunde H, Woerle HJ, et al. (February 2014). "Empagliflozin improves glycaemic and weight control as add-on therapy to pioglitazone or pioglitazone plus metformin in patients with type 2 diabetes: a 24-week, randomized, placebo-controlled trial". Diabetes, Obesity & Metabolism. 16 (2): 147–158. doi: 10.1111/dom.12188. PMID 23906415.
- ^ a b c d Häring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Broedl UC, et al. (June 2014). "Empagliflozin as add-on to metformin in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial". Diabetes Care. 37 (6): 1650–1659. doi: 10.2337/dc13-2105. PMID 24722494.
- ^ Anker SD, Khan MS, Butler J, Ofstad AP, Peil B, Pfarr E, et al. (January 2023). "Weight change and clinical outcomes in heart failure with reduced ejection fraction: insights from EMPEROR-Reduced". European Journal of Heart Failure. 25 (1): 117–127. doi: 10.1002/ejhf.2728. PMC 10098519. PMID 36325584.
- ^ "Obesity and overweight fact sheet" (PDF). World Health Organization. Archived (PDF) from the original on 23 January 2019. Retrieved 23 March 2019.
 | |
| Clinical data | |
|---|---|
| Trade names | Jardiance, others |
| Other names | BI-10773 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a614043 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | By mouth |
| Drug class | Sodium-glucose cotransporter-2 (SGLT2) inhibitor [2] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.122.058 |
| Chemical and physical data | |
| Formula | C23H27ClO7 |
| Molar mass | 450.91 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Empagliflozin, sold under the brand name Jardiance, among others, is an antidiabetic medication used to improve glucose control in people with type 2 diabetes. [10] [2] [12] It is taken by mouth. [2]
Common side effects include hyperventilation, anorexia, abdominal pain, nausea, vomiting, lethargy, mental status changes, hypotension, acute kidney injury, and vaginal yeast infections. [2] Rarer but more serious side effects include a skin infection of the groin called Fournier's gangrene and a form of diabetic ketoacidosis with normal blood sugar levels. [2] [13] Use during pregnancy or breastfeeding is not recommended. [14] Empagliflozin sometimes causes a transient decline in kidney function, and on rare occasions causes acute kidney injury, so use should be monitored in those with kidney dysfunction. But some trials have indicated that empagliflozin can be used in people with an eGFR as low as 20 mL/min/1.73 m², without increasing adverse kidney outcomes. [15] [16]
The use of empagliflozin has been shown to improve outcomes in people with established cardiovascular disease. [17] [15] There is evidence from high quality studies that empagliflozin can also help to slow the rate of kidney function decline. Irrespective of diabetes status, benefit was observed in those with mild, moderate or severe loss of kidney function. [18] [19] People started on empagliflozin may first see a decrease in kidney function before their glomerular filtration rate stabilises. [20] Greatest benefit was demonstrated in those who had severe loss of kidney function, higher risk of kidney function worsening and background of diabetes. [21]
Empagliflozin is an inhibitor of the sodium glucose co-transporter-2 (SGLT-2), and works by increasing sugar loss in urine. [2]
Empagliflozin was approved for medical use in the United States and in the European Union in 2014. [11] [22] [23] It is on the World Health Organization's List of Essential Medicines. [24] In 2021, it was the 85th most commonly prescribed medication in the United States, with more than 8 million prescriptions. [25] [26] It has received approval as a generic medication from the US Food and Drug Administration (FDA). [27]
Empagliflozin lowers risk of hospitalisation and death in patients with reduced heart function, when added to standard heart failure treatment with or without type 2 diabetes. [28] [29] [30] It is indicated in adults with type 2 diabetes and established cardiovascular disease to reduce the risk of cardiovascular death; and as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. [10] [31] [32]
In June 2023, the US Food and Drug Administration (FDA) expanded the indication, as an addition to diet and exercise, to improve blood sugar control in children 10 years and older with type 2 diabetes. [33]
- History of a severe allergic reaction to empagliflozin [10]
- End-stage kidney disease [10]
- Diabetic ketoacidosis [10]
- Empagliflozin increases the risk of genital fungal infections. The risk is highest in people with a prior history of genital fungal infections. [34]
- Empagliflozin has been thought to be associated with increased risk of urinary tract infections. Reviews of clinical trials have shown there is no significant risk of developing urinary tract infections while taking empagliflozin when compared to placebo or other diabetic medications. [35] [36]
- Empagliflozin reduces systolic and diastolic blood pressure and can increase the risk of low blood pressure, which can cause fainting and/or falls. [34] The risk is higher in older people, people taking diuretics, and people with reduced kidney function. [34]
- Slight increases in Low-density lipoprotein (LDL) cholesterol can be seen with empagliflozin, in the range of 2–4% from baseline. [34]
- Diabetic ketoacidosis, a rare but potentially life-threatening condition, may occur more commonly with empagliflozin and other SGLT-2 inhibitors. [37] [38] While diabetic ketoacidosis is usually associated with elevated blood glucose levels, in people taking SGLT-2 inhibitors diabetic ketoacidosis may be seen with uncharacteristically normal blood glucose levels, a phenomenon called euglycemic diabetic ketoacidosis. [37] The absence of elevated blood glucose levels in people on an SGLT-2 inhibitor may make it more difficult to diagnose diabetic ketoacidosis. The risk of empagliflozin-associated euglycemic diabetic ketoacidosis may be higher in the setting of illness, dehydration, surgery, and/or alcohol consumption. [37] It is also seen in type 1 diabetes who take empagliflozin, which notably is an unapproved or "off-label" use of the medication. [38] To lessen the risk of developing ketoacidosis (a serious condition in which the body produces high levels of blood acids called ketones) after surgery, the FDA has approved changes to the prescribing information for SGLT2 inhibitor diabetes medicines to recommend they be stopped temporarily before scheduled surgery. Empagliflozin should each be stopped at least three days before scheduled surgery. [39] Symptoms of diabetic ketoacidosis include nausea, vomiting, abdominal pain, tiredness, and trouble breathing. [39]
- Fournier's gangrene, a rare but serious infection of the groin, occurs more commonly in people taking empagliflozin and other SGLT-2 inhibitors. [2] [13] Symptoms include feverishness, a general sense of malaise, and pain or swelling around the genitals or in the skin behind them. The infection progresses quickly and urgent medical attention is recommended. [13]
- Empagliflozin can increase the risk of low blood sugar when it is used together with a sulfonylurea or insulin. [40] When used by itself or in addition to metformin it does not appear to increase the risk of hypoglycemia. [41]
Empagliflozin is an inhibitor of the sodium glucose co-transporter-2 (SGLT-2), which is found almost exclusively in the proximal tubules of nephronic components in the kidneys. SGLT-2 accounts for about 90 percent of glucose reabsorption into the blood. Blocking SGLT-2 reduces blood glucose by blocking glucose reabsorption in the kidney and thereby excreting glucose (i.e., blood sugar) via the urine. [42] [43] [44] Of all the SGLT-2 Inhibitors currently available, empagliflozin has the highest degree of selectivity for SGLT-2 over SGLT-1, SGLT-4, SGLT-5 and SGLT-6. [45]
It was developed by Boehringer Ingelheim and is co-marketed by Eli Lilly and Company. It is also available as the combinations empagliflozin/linagliptin, empagliflozin/metformin, and empagliflozin/linagliptin/metformin.
For cardiovascular death, the FDA based its decision on a postmarketing study it required when it approved empagliflozin in 2014 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. [22] [31] Empagliflozin was studied in a postmarket clinical trial of more than 7,000 participants with type 2 diabetes and cardiovascular disease. [31] In the trial, empagliflozin was shown to reduce the risk of cardiovascular death compared to a placebo when added to standard of care therapies for diabetes and atherosclerotic cardiovascular disease. [31]
For heart failure, the safety and effectiveness of empagliflozin were evaluated by the FDA as an adjunct to standard of care therapy in a randomized, double-blind, international trial comparing 2,997 participants who received empagliflozin, 10 mg, once daily to 2,991 participants who received the placebo. [32] The main efficacy measurement was the time to death from cardiovascular causes or need to be hospitalized for heart failure. [32] Of the individuals who received empagliflozin for an average of about two years, 14% died from cardiovascular causes or were hospitalized for heart failure, compared to 17% of the participants who received the placebo. [32] This benefit was mostly attributable to fewer participants being hospitalized for heart failure. [32]
The FDA granted the application for empagliflozin priority review and granted the approval of Jardiance to Boehringer Ingelheim. [32]
As of May 2013, Boehringer and Lilly had submitted applications for marketing approval to the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA). [46] The drug was approved in the European Union in May 2014, [11] and was approved in the United States in August 2014. [22] [23] [47] The FDA required four postmarketing studies: a cardiovascular outcomes trial, two studies in children, and a toxicity study in animals related to the pediatric trials. [22] [47]
A meta-analysis of short-term randomised controlled trials has shown similar efficacy on glycaemic control between empagliflozin 10mg and 25mg in patients with type 2 diabetes. Whilst there may be a higher reduction in HbA1c with higher doses, this difference is more clinically significant when the patients baseline HbA1c is higher e.g. those with a HbA1c ≥ 8.5%. Therefore patients’ baseline values, such as HbA1c, BMI etc, should be taken into account when prescribing high dose empagliflozin. [48] [49]
Empagliflozin causes moderate reductions in blood pressure and body weight. These effects are likely due to the excretion of glucose in the urine and a slight increase in urinary sodium excretion. [34] [50]
In clinical trials, participants with type 2 diabetes taking empagliflozin with other diabetic medications lost an average of 2% of their baseline body weight. [51] [52] A higher percentage of people taking empagliflozin achieved weight loss greater than 5% from their baseline, which has been associated with improved glucose control. [51] [52] [34] The same extent of weight loss was also observed in a study with heart failure patients taking empagliflozin. [53]
Empagliflozin has been shown to reduce systolic blood pressure by 3 to 5 millimeters of mercury (mmHg) without changes in pulse rate. [51] [52] [34] A greater percentage of people with uncontrolled blood pressure at baseline, achieved controlled blood pressure (i.e. systolic blood pressure <130 mmHg and diastolic blood pressure <80 mmHg) after taking empagliflozin at 24 weeks. [52] The effects on blood pressure and body weight are generally viewed as favorable, as many people with type 2 diabetes have high blood pressure or are overweight or obese. [41] [54]
- ^ "Empagliflozin (Jardiance) Use During Pregnancy". Drugs.com. 30 August 2018. Archived from the original on 5 August 2019. Retrieved 10 February 2020.
- ^ a b c d e f g "Empagliflozin Monograph for Professionals". Drugs.com. AHFS. Archived from the original on 6 April 2019. Retrieved 21 December 2018.
- ^ "AusPAR: Empagliflozin". Therapeutic Goods Administration (TGA), Commonwealth of Australia. 8 November 2017. Retrieved 24 March 2022.
- ^ "JARDIANCE". Boehringer Ingelheim Pty Ltd. Therapeutic Goods Administration (TGA), Commonwealth of Australia. Archived from the original on 18 March 2023.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2014". Therapeutic Goods Administration (TGA), Commonwealth of Australia. 21 June 2022. Archived from the original on 10 April 2023. Retrieved 10 April 2023.
- ^ "Prescription medicines and biologicals: TGA annual summary 2017". Therapeutic Goods Administration (TGA), Commonwealth of Australia. 21 June 2022. Retrieved 31 March 2024.
- ^ "Health Canada New Drug Authorizations: 2015 Highlights". Health Canada. 4 May 2016. Retrieved 7 April 2024.
- ^ "Jardiance 10 mg film-coated tablets – Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 20 September 2020. Retrieved 10 February 2020.
- ^ "Jardiance 25 mg film-coated tablets – Summary of Product Characteristics (SmPC)". (emc). 23 October 2019. Archived from the original on 22 September 2020. Retrieved 10 February 2020.
- ^ a b c d e f "Jardiance- empagliflozin tablet, film coated". DailyMed. 22 January 2020. Archived from the original on 11 February 2020. Retrieved 10 February 2020.
- ^ a b c "Jardiance EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 28 August 2021. Retrieved 10 February 2020.
- ^ Davies MJ, D'Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, et al. (December 2018). "Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD)". Diabetologia. 61 (12): 2461–2498. doi: 10.1007/s00125-018-4729-5. PMID 30288571.
- ^ a b c "FDA warns about rare occurrences of a serious infection of the genital area with SGLT2 inhibitors for diabetes". U.S. Food and Drug Administration (FDA). 9 February 2019. Archived from the original on 13 December 2019. Retrieved 18 March 2019.
- ^ British national formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 691. ISBN 9780857113382.
- ^ a b Zannad F, Butler J, Filippatos GS, Pocock S, Jamal W, Schnee J, et al. (May 2021). "Cardiovascular and Kidney Outcomes with Empagliflozin in Heart Failure". Präzisionsmedizin – Eine Reise in die Zukunft der Diabetologie www.diabeteskongress.de. 16. Georg Thieme Verlag KG. doi: 10.1055/s-0041-1727471.
- ^ Herrington WG, Staplin N, Wanner C, Green JB, Hauske SJ, Emberson JR, et al. (January 2023). "Empagliflozin in Patients with Chronic Kidney Disease". The New England Journal of Medicine. 388 (2): 117–127. doi: 10.1056/NEJMoa2204233. PMC 7614055. PMID 36331190.
- ^ Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. (November 2015). "Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes". The New England Journal of Medicine. 373 (22): 2117–2128. doi: 10.1056/NEJMoa1504720. PMID 26378978.
- ^ Herrington WG, Preiss D, Haynes R, von Eynatten M, Staplin N, Hauske SJ, et al. (August 2020). "Erratum: The potential for improving cardio-renal outcomes by sodium-glucose co-transporter-2 inhibition in people with chronic kidney disease: a rationale for the EMPA-KIDNEY study". Clinical Kidney Journal. 13 (4): 722. doi: 10.1056/NEJMoa2204233. hdl: 20.500.13003/18576. PMC 7467589. PMID 32905262.
- ^ Wanner C, Inzucchi SE, Lachin JM, Fitchett D, von Eynatten M, Mattheus M, et al. (July 2016). "Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes". The New England Journal of Medicine. 375 (4): 323–334. doi: 10.1056/nejmoa1515920. PMID 27299675.
- ^ Wanner C, Inzucchi SE, Lachin JM, Fitchett D, von Eynatten M, Mattheus M, et al. (July 2016). "Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes". The New England Journal of Medicine. 375 (4): 323–334. doi: 10.1056/NEJMoa1515920. PMID 27299675.
- ^ Herrington WG, Preiss D, Haynes R, von Eynatten M, Staplin N, Hauske SJ, et al. (August 2020). "Erratum: The potential for improving cardio-renal outcomes by sodium-glucose co-transporter-2 inhibition in people with chronic kidney disease: a rationale for the EMPA-KIDNEY study". Clinical Kidney Journal. 13 (4): 722. doi: 10.1056/NEJMoa2204233. hdl: 20.500.13003/18576. PMC 7467589. PMID 32905262.
- ^
a
b
c
d
"FDA approves Jardiance to treat type 2 diabetes" (Press release). U.S.
Food and Drug Administration (FDA). 1 August 2014. Archived from
the original on 22 October 2016. Retrieved 5 February 2023.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ a b "Drug Approval Package: Jardiance (empagliflozin) Tablets NDA #204629". U.S. Food and Drug Administration (FDA). 8 September 2014. Archived from the original on 11 February 2020. Retrieved 10 February 2020.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl: 10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Empagliflozin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "2022 First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). 3 March 2023. Archived from the original on 14 January 2024. Retrieved 14 January 2024.
- ^ Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, et al. (October 2020). "Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure". The New England Journal of Medicine. 383 (15): 1413–1424. doi: 10.1056/NEJMoa2022190. hdl: 2066/230126. PMID 32865377.
- ^ Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, et al. (October 2021). "Empagliflozin in Heart Failure with a Preserved Ejection Fraction". The New England Journal of Medicine. 385 (16): 1451–1461. doi: 10.1056/NEJMoa2107038. PMID 34449189.
- ^ Zannad F, Ferreira JP, Pocock SJ, Anker SD, Butler J, Filippatos G, et al. (September 2020). "SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-Reduced and DAPA-HF trials". Lancet. 396 (10254): 819–829. doi: 10.1016/s0140-6736(20)31824-9. PMID 32877652.
- ^
a
b
c
d
"FDA approves Jardiance to reduce cardiovascular death in adults with type 2 diabetes" (Press release). U.S.
Food and Drug Administration (FDA). 6 December 2016.
Archived from the original on 11 February 2020. Retrieved 12 December 2016.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^
a
b
c
d
e
f
"FDA Approves Treatment for Wider Range of Patients with Heart Failure" (Press release). U.S.
Food and Drug Administration (FDA). 24 February 2022.
Archived from the original on 27 February 2022. Retrieved 27 February 2022.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ "FDA Approves New Class of Medicines to Treat Pediatric Type 2 Diabetes". U.S. Food and Drug Administration (FDA) (Press release). 20 June 2023. Archived from the original on 21 June 2023. Retrieved 20 June 2023.
- ^ a b c d e f g "Empagliflozin (Jardiance) National Drug Monograph" (PDF). U.S. Department of Veterans Affairs. October 2015. Archived from the original (PDF) on 18 March 2021.
- ^ Donnan JR, Grandy CA, Chibrikov E, Marra CA, Aubrey-Bassler K, Johnston K, et al. (February 2019). "Comparative safety of the sodium glucose co-transporter 2 (SGLT2) inhibitors: a systematic review and meta-analysis". BMJ Open. 9 (1): e022577. doi: 10.1136/bmjopen-2018-022577. PMC 6361337. PMID 30813108.
- ^ Kinduryte Schorling O, Clark D, Zwiener I, Kaspers S, Lee J, Iliev H (August 2020). "Pooled Safety and Tolerability Analysis of Empagliflozin in Patients with Type 2 Diabetes Mellitus". Advances in Therapy. 37 (8): 3463–3484. doi: 10.1007/s12325-020-01329-7. PMC 7370973. PMID 32372290.
- ^ a b c Rosenstock J, Ferrannini E (September 2015). "Euglycemic Diabetic Ketoacidosis: A Predictable, Detectable, and Preventable Safety Concern With SGLT2 Inhibitors". Diabetes Care. 38 (9): 1638–1642. doi: 10.2337/dc15-1380. PMID 26294774.
- ^ a b Handelsman Y, Henry RR, Bloomgarden ZT, Dagogo-Jack S, DeFronzo RA, Einhorn D, et al. (June 2016). "American Association of Clinical Endocrinologists and American College of Endocrinology Position Statement on the Association of Sglt-2 Inhibitors and Diabetic Ketoacidosis". Endocrine Practice. 22 (6): 753–762. doi: 10.4158/EP161292.PS. PMID 27082665.
- ^
a
b
"FDA revises labels of SGLT2 inhibitors for diabetes to include warning". U.S.
Food and Drug Administration (FDA). 19 March 2020.
Archived from the original on 7 June 2020. Retrieved 6 June 2020.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ "Empagliflozin in combination therapy for treating type 2 diabetes". NICE. 25 March 2015. Archived from the original on 21 December 2018. Retrieved 21 December 2018.
- ^ a b Anderson JE, Wright EE, Shaefer CF (February 2017). "Empagliflozin: Role in Treatment Options for Patients with Type 2 Diabetes Mellitus". Diabetes Therapy. 8 (1): 33–53. doi: 10.1007/s13300-016-0211-x. PMC 5306110. PMID 27837465.
- ^ Abdul-Ghani MA, DeFronzo RA (September 2008). "Inhibition of renal glucose reabsorption: a novel strategy for achieving glucose control in type 2 diabetes mellitus". Endocrine Practice. 14 (6): 782–790. doi: 10.4158/ep.14.6.782. PMID 18996802.
- ^ Nair S, Wilding JP (January 2010). "Sodium glucose cotransporter 2 inhibitors as a new treatment for diabetes mellitus". The Journal of Clinical Endocrinology and Metabolism. 95 (1): 34–42. doi: 10.1210/jc.2009-0473. PMID 19892839.
- ^ Bays H (March 2009). "From victim to ally: the kidney as an emerging target for the treatment of diabetes mellitus". Current Medical Research and Opinion. 25 (3): 671–681. doi: 10.1185/03007990802710422. PMID 19232040. S2CID 73341491.
- ^ Tentolouris A, Vlachakis P, Tzeravini E, Eleftheriadou I, Tentolouris N (August 2019). "SGLT2 Inhibitors: A Review of Their Antidiabetic and Cardioprotective Effects". International Journal of Environmental Research and Public Health. 16 (16): 2965. doi: 10.3390/ijerph16162965. PMC 6720282. PMID 31426529.
- ^ Tucker ME (7 May 2013). "First details of empagliflozin trials follow US and EU filings". Medscape Medical News. Archived from the original on 18 June 2013. Retrieved 18 January 2014.
- ^ a b Mechatie E (1 August 2014). "FDA approves empagliflozin for adults with type 2 diabetes". Clinical Endocrinology News Digital Network. Archived from the original on 28 August 2021. Retrieved 23 August 2014.
- ^ Liakos A, Karagiannis T, Athanasiadou E, Sarigianni M, Mainou M, Papatheodorou K, et al. (October 2014). "Efficacy and safety of empagliflozin for type 2 diabetes: a systematic review and meta-analysis". Diabetes, Obesity & Metabolism. 16 (10): 984–993. doi: 10.1111/dom.12307. PMID 24766495.
- ^ Inzucchi SE, Davies MJ, Khunti K, Trivedi P, George JT, Zwiener I, et al. (February 2021). "Empagliflozin treatment effects across categories of baseline HbA1c, body weight and blood pressure as an add-on to metformin in patients with type 2 diabetes". Diabetes, Obesity & Metabolism. 23 (2): 425–433. doi: 10.1111/dom.14234. PMC 7839733. PMID 33084149.
- ^ Verma S, McMurray JJ (October 2018). "SGLT2 inhibitors and mechanisms of cardiovascular benefit: a state-of-the-art review". Diabetologia. 61 (10): 2108–2117. doi: 10.1007/s00125-018-4670-7. PMID 30132036.
- ^ a b c Kovacs CS, Seshiah V, Swallow R, Jones R, Rattunde H, Woerle HJ, et al. (February 2014). "Empagliflozin improves glycaemic and weight control as add-on therapy to pioglitazone or pioglitazone plus metformin in patients with type 2 diabetes: a 24-week, randomized, placebo-controlled trial". Diabetes, Obesity & Metabolism. 16 (2): 147–158. doi: 10.1111/dom.12188. PMID 23906415.
- ^ a b c d Häring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Broedl UC, et al. (June 2014). "Empagliflozin as add-on to metformin in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial". Diabetes Care. 37 (6): 1650–1659. doi: 10.2337/dc13-2105. PMID 24722494.
- ^ Anker SD, Khan MS, Butler J, Ofstad AP, Peil B, Pfarr E, et al. (January 2023). "Weight change and clinical outcomes in heart failure with reduced ejection fraction: insights from EMPEROR-Reduced". European Journal of Heart Failure. 25 (1): 117–127. doi: 10.1002/ejhf.2728. PMC 10098519. PMID 36325584.
- ^ "Obesity and overweight fact sheet" (PDF). World Health Organization. Archived (PDF) from the original on 23 January 2019. Retrieved 23 March 2019.