| |
| Names | |
|---|---|
|
IUPAC name
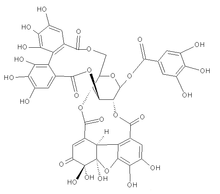
(1R,7R,8S,26R,28S,29R,38R)-1,13,14,15,18,19,20,34,35,39,39-Undecahydroxy-2,5,10,23,31-pentaoxo-6,9,24,27,30,40-hexaoxaoctacyclo[34.3.1.04,38.07,26.08,29.011,16.017,22.032,37]tetraconta-3,1 1,13,15,17,19,21,32,34,36-decaen-28-yl 3,4,5-trihydroxybenzoate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| |
| Properties | |
| C41H28O27 | |
| Molar mass | 952.64 g/mol |
| Density | 2.26 g/mL |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Geraniin is a dehydro ellagitannin found in geraniums. [1] It is found for instance in Geranium thunbergii, which is one of the most popular folk medicines and also an official antidiarrheic drug in Japan. [2] It can also be found in the rind of Nephelium lappaceum (rambutan). [3]
It mediates apoptosis by cleavage of focal adhesion kinase through up-regulation of Fas ligand expression in human melanoma cells. [1]
Geraniin has also been shown to possess immunomodularity properties, as it inhibits tumor necrosis factor-alpha, and NF-κB in ovarian cancer cells. [4]
Geraniin was studied for its anticancer activity and shown to target apoptosis via inactivation of PI3K/Akt/mTOR signaling pathway involving NF-κB when treated against HT-29 human colorectal adenocarcinoma cells. [5]
It is formed with one hexahydroxydiphenic acid unit, one modified hexahydroxydiphenic acid unit ( dehydrohexahydroxydiphenic acid or DHHDP) and one gallic acid unit linked to a glucose molecule. It is forming an equilibrium mixture of six-membered hemi-ketal and five-membered hemi-ketal forms.[ citation needed]
Chebulagic acid is formed from geraniin through a glutathione-mediated conversion. [6]
- ^ a b Lee, Jang-Chang; Tsai, Chih-Yen; Kao, Jung-Yie; Kao, Ming-Ching; Tsai, Shih-Chang; Chang, Chih-Shiang; Huang, Li-Jiau; Kuo, Sheng-Chu; et al. (2008). "Geraniin-mediated apoptosis by cleavage of focal adhesion kinase through up-regulation of Fas ligand expression in human melanoma cells". Molecular Nutrition & Food Research. 52 (6): 655–63. doi: 10.1002/mnfr.200700381. PMID 18435487.
- ^ Luger, P.; Weber, M.; Kashino, S.; Amakura, Y.; Yoshida, T.; Okuda, T.; Beurskens, G.; Dauter, Z. (1998). "Structure of the Tannin Geraniin Based on Conventional X-ray Data at 295 K and on Synchrotron Data at 293 and 120 K". Acta Crystallographica Section B. 54 (5): 687. Bibcode: 1998AcCrB..54..687L. doi: 10.1107/S0108768198000081.
- ^ Rapid isolation of geraniin from Nephelium lappaceum rind waste and its anti-hyperglycemic activity. Uma D. Palanisamy, Lai Teng Ling, Thamilvaani Manaharan, and David Appleton, Food Chemistry, 1 July 2011, Volume 127, Issue 1, Pages 21–27, doi: 10.1016/j.foodchem.2010.12.070
- ^ Wang X, et al. Geraniin suppresses ovarian cancer growth through inhibition of NF-κB activation and downregulation of Mcl-1 expression. J Biochem Mol Toxicol. 2017 Sep;31(9)
- ^ Chan, Chim Kei; Tang, Liu Ying; Goh, Bey Hing; Abdul Kadir, Habsah (2019). "Targeting apoptosis via inactivation of PI3K/Akt/MTOR signaling pathway involving NF-κB by geraniin in HT-29 human colorectal adenocarcinoma cells". Progress in Drug Discovery & Biomedical Science. 2. doi: 10.36877/pddbs.a0000030.
- ^ Glutathione-mediated conversion of the ellagitannin geraniin into chebulagic acid. Tanaka T, Kouno I and Nonaka G.I, Chemical and pharmaceutical bulletin, 1996, volume 44, no 1, pages 34-40, INIST 3003361

| |
| Names | |
|---|---|
|
IUPAC name
(1R,7R,8S,26R,28S,29R,38R)-1,13,14,15,18,19,20,34,35,39,39-Undecahydroxy-2,5,10,23,31-pentaoxo-6,9,24,27,30,40-hexaoxaoctacyclo[34.3.1.04,38.07,26.08,29.011,16.017,22.032,37]tetraconta-3,1 1,13,15,17,19,21,32,34,36-decaen-28-yl 3,4,5-trihydroxybenzoate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| |
| Properties | |
| C41H28O27 | |
| Molar mass | 952.64 g/mol |
| Density | 2.26 g/mL |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Geraniin is a dehydro ellagitannin found in geraniums. [1] It is found for instance in Geranium thunbergii, which is one of the most popular folk medicines and also an official antidiarrheic drug in Japan. [2] It can also be found in the rind of Nephelium lappaceum (rambutan). [3]
It mediates apoptosis by cleavage of focal adhesion kinase through up-regulation of Fas ligand expression in human melanoma cells. [1]
Geraniin has also been shown to possess immunomodularity properties, as it inhibits tumor necrosis factor-alpha, and NF-κB in ovarian cancer cells. [4]
Geraniin was studied for its anticancer activity and shown to target apoptosis via inactivation of PI3K/Akt/mTOR signaling pathway involving NF-κB when treated against HT-29 human colorectal adenocarcinoma cells. [5]
It is formed with one hexahydroxydiphenic acid unit, one modified hexahydroxydiphenic acid unit ( dehydrohexahydroxydiphenic acid or DHHDP) and one gallic acid unit linked to a glucose molecule. It is forming an equilibrium mixture of six-membered hemi-ketal and five-membered hemi-ketal forms.[ citation needed]
Chebulagic acid is formed from geraniin through a glutathione-mediated conversion. [6]
- ^ a b Lee, Jang-Chang; Tsai, Chih-Yen; Kao, Jung-Yie; Kao, Ming-Ching; Tsai, Shih-Chang; Chang, Chih-Shiang; Huang, Li-Jiau; Kuo, Sheng-Chu; et al. (2008). "Geraniin-mediated apoptosis by cleavage of focal adhesion kinase through up-regulation of Fas ligand expression in human melanoma cells". Molecular Nutrition & Food Research. 52 (6): 655–63. doi: 10.1002/mnfr.200700381. PMID 18435487.
- ^ Luger, P.; Weber, M.; Kashino, S.; Amakura, Y.; Yoshida, T.; Okuda, T.; Beurskens, G.; Dauter, Z. (1998). "Structure of the Tannin Geraniin Based on Conventional X-ray Data at 295 K and on Synchrotron Data at 293 and 120 K". Acta Crystallographica Section B. 54 (5): 687. Bibcode: 1998AcCrB..54..687L. doi: 10.1107/S0108768198000081.
- ^ Rapid isolation of geraniin from Nephelium lappaceum rind waste and its anti-hyperglycemic activity. Uma D. Palanisamy, Lai Teng Ling, Thamilvaani Manaharan, and David Appleton, Food Chemistry, 1 July 2011, Volume 127, Issue 1, Pages 21–27, doi: 10.1016/j.foodchem.2010.12.070
- ^ Wang X, et al. Geraniin suppresses ovarian cancer growth through inhibition of NF-κB activation and downregulation of Mcl-1 expression. J Biochem Mol Toxicol. 2017 Sep;31(9)
- ^ Chan, Chim Kei; Tang, Liu Ying; Goh, Bey Hing; Abdul Kadir, Habsah (2019). "Targeting apoptosis via inactivation of PI3K/Akt/MTOR signaling pathway involving NF-κB by geraniin in HT-29 human colorectal adenocarcinoma cells". Progress in Drug Discovery & Biomedical Science. 2. doi: 10.36877/pddbs.a0000030.
- ^ Glutathione-mediated conversion of the ellagitannin geraniin into chebulagic acid. Tanaka T, Kouno I and Nonaka G.I, Chemical and pharmaceutical bulletin, 1996, volume 44, no 1, pages 34-40, INIST 3003361