| CoreyŌĆōSeebach reaction | |
|---|---|
| Named after |
Elias James Corey Dieter Seebach |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | corey-seebach-reaction |
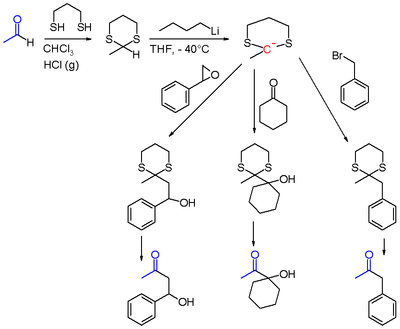
The CoreyŌĆōSeebach reaction, or Seebach Umpolung is a name reaction of organic chemistry that allows for acylation by converting aldehydes into lithiated 1,3-dithianes. The lithiated 1,3-dithianes serves as an acyl anion equivalent, undergoing alkylation with electrophiles. [1] The reaction is named in honor of its discoverers, Elias J. Corey and Dieter Seebach.
Implementation
The aldehyde is first converted into a dithiane, usually with 1,3-propanedithiol. The resulting 1,3-dithiane is then lithiated with the use of butyllithium. The 2-lithio-1,3-dithiane reacts with electrophiles to give a 2-alkyl-1,3-dithiane.
Finally, the 2-alkyl-1,3-dithiane can be converted to a carbonyl by hydrolysis, usually with the use of mercury(II) oxide. Alternatively the 2-alkyl-1,3-dithiane can be reduced to an alkane. [2]
Scope
As a strategy for protecting aldehydes and ketones, dithiane formation is cumbersome because deprotection is inefficient. Typically ketones and aldehydes are protected as their dioxolanes instead of dithianes. The Corey-Seebach reaction is of interest as an acyl anion equivalent, allowing aldehydes to be converted to ketones.
The lithiated 1,3-dithiane can be alkylated with alkyl halides, epoxides, [3] [4] ketones, acyl halides, and iminium salts, which after hydrolysis of dithioacetals can yield ketones, ╬▓-hydroxyketones, ╬▒-hydroxyketones, 1,2-diketones [5] and ╬▒-aminoketones. Notably, ╬▒-hydroxyketones and 1,2-diketones can not be generated through typical reactions of aldehydes such as the aldol reaction. [6] Other possible electrophiles include aldehydes, [7] amides, and esters. [6] [8]
The reaction between lithiated 1,3-dithianes and arenesulfonates offers a similar path to that of alkyl halides, being able to form dithioacetals which can be converted to ketones. [9]
Historic references
- Corey, Elias James; Seebach, Dieter (1 December 1965). "Synthesis of 1,n-Dicarbonyl Derivates Using Carbanions from 1,3-Dithianes". Angewandte Chemie International Edition. 4 (12): 1077ŌĆō1078. doi: 10.1002/anie.196510771.
- Corey, Elias James; Seebach, Dieter (1 December 1966). "Phenylthiomethyllithium and Bis(phenylthio)methyllithium". The Journal of Organic Chemistry. 31 (12): 4097ŌĆō4099. doi: 10.1021/jo01350a052.
- Seebach, Dieter; Corey, Elias James (1 January 1975). "Generation and synthetic applications of 2-lithio-1,3-dithianes". The Journal of Organic Chemistry. 40 (2): 231ŌĆō237. doi: 10.1021/jo00890a018.</ref>
References
- ^ Smith, Amos B.; Adams, Christopher M. (6 May 2004). "Evolution of Dithiane-Based Strategies for the Construction of Architecturally Complex Natural Products". Accounts of Chemical Research. 37 (6): 365ŌĆō377. doi: 10.1021/ar030245r. PMID 15196046.
- ^ Yus, Miguel; N├Ījera, Carmen; Foubelo, Francisco (11 August 2003). "The role of 1,3-dithianes in natural product synthesis". Tetrahedron. 59 (13): 6147ŌĆō6212. doi: 10.1016/S0040-4020(03)00955-4.
- ^ Williams, David R.; Sit, Sing Yuen (1 May 1984). "Total synthesis of (+)-phyllanthocin". Journal of the American Chemical Society. 106 (10): 2949ŌĆō2954. doi: 10.1021/ja00322a035.
- ^ Smith, Amos B.; Xian, Ming (11 January 2006). "Anion Relay Chemistry: An Effective Tactic for Diversity Oriented Synthesis". Journal of the American Chemical Society. 128 (1): 66ŌĆō67. doi: 10.1021/ja057059w. PMID 16390124.
- ^ Corey, Elias James; Seebach, Dieter (1 December 1965). "Synthesis of 1,n-Dicarbonyl Derivates Using Carbanions from 1,3-Dithianes". Angewandte Chemie International Edition. 4 (12): 1077ŌĆō1078. doi: 10.1002/anie.196510771.
- ^ a b Page, Philip C. Bulman; van Niel, Monique B.; Pordger, Jeremy C. (29 November 1988). "Synthetic uses of the 1,3-dithiane grouping from 1977 to 1988". Tetrahedron. 45 (24): 7643ŌĆō7677. doi: 10.1016/S0040-4020(01)85784-7.
- ^ Ley, Steven V.; Abad-Somovilla, Antonio; Anderson, James C.; Ayats, Carles; B├żnteli, Rolf; Beckmann, Edith; Boyer, Alistair; Brasca, Maria G.; Brice, Abigail; Broughton, Howard B.; Burke, Brenda J.; Cleator, Ed; Craig, Donald; Denholm, Alastair A.; Denton, Ross M.; Durand-Reville, Thomas; Gobbi, Luca B.; G├Čbel, Michael; Gray, Brian Lawrence; Grossmann, Robert B.; Gutteridge, Clair E.; Hahn, Norbert; Harding, Sarah L.; Jennens, David C.; Jennens, Lynn; Lovell, Peter J.; Lovell, Helen J.; de la Puente, Mary L.; Kolb, Martmuth C.; Koot, Win-Jan; Maslen, Sarah L.; McCusker, Catherine; Mattes, Amos; Pape, Andrew R.; Pinto, Andrea; Santafianos, Dinos; Scott, James S.; Smith, Stephen C.; Somers, Andrew Q.; Spilling, Christopher D.; Stelzer, Frank; Toogood, Peter L.; Turner, Richard M; Veitch, Gemma E.; Wood, Anthony; Zumbrunn, Cornelia (1 January 2018). "The synthesis of azadirachtin: a potent insect antifeedant". Chemistry: A European Journal. 14 (34): 10683ŌĆō10704. doi: 10.1002/chem.200801103. PMID 18821532.
- ^ Chen, Yue-Lei; Leguijt, Robin; Redlich, Hartmut; Fr├Čhlich, Roland (30 April 2006). "Trimethylene Dithioacetals of Carbohydrates, Part 6: C-C Coupling Reactions of Dilithiated N-Acetyl-D-glucosamine Trimethylene Dithioacetal Derivatives". Synthesis. 2006 (24): 4212ŌĆō4218. doi: 10.1055/s-2006-950359.
- ^ Seebach, Dieter; Wilka, Eva-Maria (1976). "Alkylation of 2-Lithio-1,3-dithianes with Arenesulfonates of Primary Alcohols". Synthesis. 1976 (7): 476ŌĆō477. doi: 10.1055/s-1976-24092.
| CoreyŌĆōSeebach reaction | |
|---|---|
| Named after |
Elias James Corey Dieter Seebach |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | corey-seebach-reaction |
The CoreyŌĆōSeebach reaction, or Seebach Umpolung is a name reaction of organic chemistry that allows for acylation by converting aldehydes into lithiated 1,3-dithianes. The lithiated 1,3-dithianes serves as an acyl anion equivalent, undergoing alkylation with electrophiles. [1] The reaction is named in honor of its discoverers, Elias J. Corey and Dieter Seebach.
Implementation
The aldehyde is first converted into a dithiane, usually with 1,3-propanedithiol. The resulting 1,3-dithiane is then lithiated with the use of butyllithium. The 2-lithio-1,3-dithiane reacts with electrophiles to give a 2-alkyl-1,3-dithiane.
Finally, the 2-alkyl-1,3-dithiane can be converted to a carbonyl by hydrolysis, usually with the use of mercury(II) oxide. Alternatively the 2-alkyl-1,3-dithiane can be reduced to an alkane. [2]
Scope
As a strategy for protecting aldehydes and ketones, dithiane formation is cumbersome because deprotection is inefficient. Typically ketones and aldehydes are protected as their dioxolanes instead of dithianes. The Corey-Seebach reaction is of interest as an acyl anion equivalent, allowing aldehydes to be converted to ketones.
The lithiated 1,3-dithiane can be alkylated with alkyl halides, epoxides, [3] [4] ketones, acyl halides, and iminium salts, which after hydrolysis of dithioacetals can yield ketones, ╬▓-hydroxyketones, ╬▒-hydroxyketones, 1,2-diketones [5] and ╬▒-aminoketones. Notably, ╬▒-hydroxyketones and 1,2-diketones can not be generated through typical reactions of aldehydes such as the aldol reaction. [6] Other possible electrophiles include aldehydes, [7] amides, and esters. [6] [8]
The reaction between lithiated 1,3-dithianes and arenesulfonates offers a similar path to that of alkyl halides, being able to form dithioacetals which can be converted to ketones. [9]
Historic references
- Corey, Elias James; Seebach, Dieter (1 December 1965). "Synthesis of 1,n-Dicarbonyl Derivates Using Carbanions from 1,3-Dithianes". Angewandte Chemie International Edition. 4 (12): 1077ŌĆō1078. doi: 10.1002/anie.196510771.
- Corey, Elias James; Seebach, Dieter (1 December 1966). "Phenylthiomethyllithium and Bis(phenylthio)methyllithium". The Journal of Organic Chemistry. 31 (12): 4097ŌĆō4099. doi: 10.1021/jo01350a052.
- Seebach, Dieter; Corey, Elias James (1 January 1975). "Generation and synthetic applications of 2-lithio-1,3-dithianes". The Journal of Organic Chemistry. 40 (2): 231ŌĆō237. doi: 10.1021/jo00890a018.</ref>
References
- ^ Smith, Amos B.; Adams, Christopher M. (6 May 2004). "Evolution of Dithiane-Based Strategies for the Construction of Architecturally Complex Natural Products". Accounts of Chemical Research. 37 (6): 365ŌĆō377. doi: 10.1021/ar030245r. PMID 15196046.
- ^ Yus, Miguel; N├Ījera, Carmen; Foubelo, Francisco (11 August 2003). "The role of 1,3-dithianes in natural product synthesis". Tetrahedron. 59 (13): 6147ŌĆō6212. doi: 10.1016/S0040-4020(03)00955-4.
- ^ Williams, David R.; Sit, Sing Yuen (1 May 1984). "Total synthesis of (+)-phyllanthocin". Journal of the American Chemical Society. 106 (10): 2949ŌĆō2954. doi: 10.1021/ja00322a035.
- ^ Smith, Amos B.; Xian, Ming (11 January 2006). "Anion Relay Chemistry: An Effective Tactic for Diversity Oriented Synthesis". Journal of the American Chemical Society. 128 (1): 66ŌĆō67. doi: 10.1021/ja057059w. PMID 16390124.
- ^ Corey, Elias James; Seebach, Dieter (1 December 1965). "Synthesis of 1,n-Dicarbonyl Derivates Using Carbanions from 1,3-Dithianes". Angewandte Chemie International Edition. 4 (12): 1077ŌĆō1078. doi: 10.1002/anie.196510771.
- ^ a b Page, Philip C. Bulman; van Niel, Monique B.; Pordger, Jeremy C. (29 November 1988). "Synthetic uses of the 1,3-dithiane grouping from 1977 to 1988". Tetrahedron. 45 (24): 7643ŌĆō7677. doi: 10.1016/S0040-4020(01)85784-7.
- ^ Ley, Steven V.; Abad-Somovilla, Antonio; Anderson, James C.; Ayats, Carles; B├żnteli, Rolf; Beckmann, Edith; Boyer, Alistair; Brasca, Maria G.; Brice, Abigail; Broughton, Howard B.; Burke, Brenda J.; Cleator, Ed; Craig, Donald; Denholm, Alastair A.; Denton, Ross M.; Durand-Reville, Thomas; Gobbi, Luca B.; G├Čbel, Michael; Gray, Brian Lawrence; Grossmann, Robert B.; Gutteridge, Clair E.; Hahn, Norbert; Harding, Sarah L.; Jennens, David C.; Jennens, Lynn; Lovell, Peter J.; Lovell, Helen J.; de la Puente, Mary L.; Kolb, Martmuth C.; Koot, Win-Jan; Maslen, Sarah L.; McCusker, Catherine; Mattes, Amos; Pape, Andrew R.; Pinto, Andrea; Santafianos, Dinos; Scott, James S.; Smith, Stephen C.; Somers, Andrew Q.; Spilling, Christopher D.; Stelzer, Frank; Toogood, Peter L.; Turner, Richard M; Veitch, Gemma E.; Wood, Anthony; Zumbrunn, Cornelia (1 January 2018). "The synthesis of azadirachtin: a potent insect antifeedant". Chemistry: A European Journal. 14 (34): 10683ŌĆō10704. doi: 10.1002/chem.200801103. PMID 18821532.
- ^ Chen, Yue-Lei; Leguijt, Robin; Redlich, Hartmut; Fr├Čhlich, Roland (30 April 2006). "Trimethylene Dithioacetals of Carbohydrates, Part 6: C-C Coupling Reactions of Dilithiated N-Acetyl-D-glucosamine Trimethylene Dithioacetal Derivatives". Synthesis. 2006 (24): 4212ŌĆō4218. doi: 10.1055/s-2006-950359.
- ^ Seebach, Dieter; Wilka, Eva-Maria (1976). "Alkylation of 2-Lithio-1,3-dithianes with Arenesulfonates of Primary Alcohols". Synthesis. 1976 (7): 476ŌĆō477. doi: 10.1055/s-1976-24092.