The Carroll rearrangement is a rearrangement reaction in organic chemistry and involves the transformation of a β- keto allyl ester into a α-allyl-β-ketocarboxylic acid. [1] This organic reaction is accompanied by decarboxylation and the final product is a γ,δ-allylketone. The Carroll rearrangement is an adaptation of the Claisen rearrangement and effectively a decarboxylative allylation.
Reaction mechanism
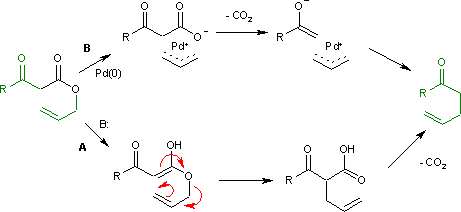
The Carroll rearrangement (1940) in the presence of base and with high reaction temperature (path A) takes place through an intermediate enol which then rearranges in a sigmatropic Claisen rearrangement. The follow-up is a decarboxylation. With palladium(0) as a catalyst, the reaction (Tsuji, 1980) is much milder (path B) with an intermediate allyl cation / carboxylic acid anion organometallic complex. [2]
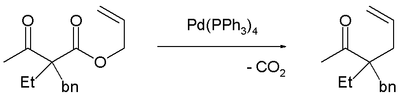
Decarboxylation precedes allylation as evidenced by this reaction catalyzed by tetrakis(triphenylphosphine)palladium(0): [3]
Asymmetric decarboxylative allylation
By introducing suitable chiral ligands, the reaction becomes enantioselective. [4]
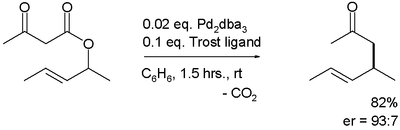
The first reported asymmetric rearrangement is catalyzed by tris(dibenzylideneacetone)dipalladium(0) and the Trost ligand: [3]
A similar reaction [5] uses additional naphthol.
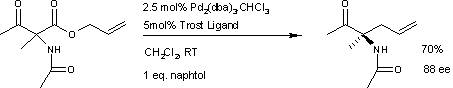
This reaction delivers the main enantiomer with 88% enantiomeric excess. It remains to be seen if this reaction will have a wide scope because the acetamido group appears to be a prerequisite.
The same catalyst but a different ligand is employed in this enantioconvergent reaction: [6]
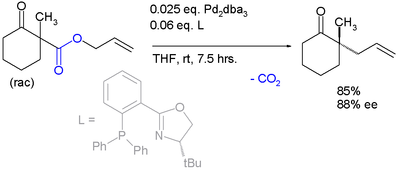
The scope is extended to asymmetric α-alkylation of ketones masked as their enol carbonate esters: [7]

References
- ^ Carroll, M. F. "131. Addition of α,β-unsaturated alcohols to the active methylene group. Part I. The action of ethyl acetoacetate on linalool and geraniol". Journal of the Chemical Society 1940, 704–706. doi: 10.1039/JR9400000704
- ^ Palladium-catalyzed rearrangement of allylic esters of acetoacetic acid to give γ,δ-unsaturated methyl ketones Tetrahedron Letters, Volume 21, Issue 33, 1980, Pages 3199-3202 Isao Shimizu, Toshiro Yamada and Jiro Tsuji doi: 10.1016/S0040-4039(00)77444-2
- ^ a b Asymmetric Allylic Alkylation of Ketone Enolates: An Asymmetric Claisen Surrogate Erin C. Burger and Jon A. Tunge Org. Lett.; 2004; 6(22) pp 4113 - 4115; (Letter) doi: 10.1021/ol048149t
- ^ Enantioselective Palladium-Catalyzed Decarboxylative Allylic Alkylations Shu-Li You and Li-Xin Dai Angew. Chem. Int. Ed. 2006, 45, 5246 – 5248 doi: 10.1002/anie.200601889
- ^ Kuwano, R.; Ishida N.; Murakami, M. "Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex". Chemical Communications 2005, (31), 3951–3952. doi: 10.1039/b505105c
- ^ Deracemization of Quaternary Stereocenters by Pd-Catalyzed Enantioconvergent Decarboxylative Allylation of Racemic b-Ketoesters Justin T. Mohr, Douglas C. Behenna, Andrew M. Harned, and Brian M. Stoltz Angew. Chem. Int. Ed. 2005, 44, 6924 –6927 doi: 10.1002/anie.200502018
- ^ Palladium-Catalyzed Asymmetric Allylic α-Alkylation of Acyclic Ketones Barry M. Trost and Jiayi Xu J. Am. Chem. Soc.; 2005; 127(49) pp 17180 - 17181; (Communication) doi: 10.1021/ja055968f
The Carroll rearrangement is a rearrangement reaction in organic chemistry and involves the transformation of a β- keto allyl ester into a α-allyl-β-ketocarboxylic acid. [1] This organic reaction is accompanied by decarboxylation and the final product is a γ,δ-allylketone. The Carroll rearrangement is an adaptation of the Claisen rearrangement and effectively a decarboxylative allylation.
Reaction mechanism
The Carroll rearrangement (1940) in the presence of base and with high reaction temperature (path A) takes place through an intermediate enol which then rearranges in a sigmatropic Claisen rearrangement. The follow-up is a decarboxylation. With palladium(0) as a catalyst, the reaction (Tsuji, 1980) is much milder (path B) with an intermediate allyl cation / carboxylic acid anion organometallic complex. [2]

Decarboxylation precedes allylation as evidenced by this reaction catalyzed by tetrakis(triphenylphosphine)palladium(0): [3]

Asymmetric decarboxylative allylation
By introducing suitable chiral ligands, the reaction becomes enantioselective. [4]
The first reported asymmetric rearrangement is catalyzed by tris(dibenzylideneacetone)dipalladium(0) and the Trost ligand: [3]

A similar reaction [5] uses additional naphthol.
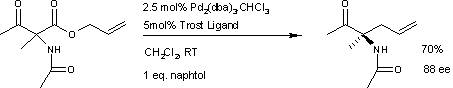
This reaction delivers the main enantiomer with 88% enantiomeric excess. It remains to be seen if this reaction will have a wide scope because the acetamido group appears to be a prerequisite.
The same catalyst but a different ligand is employed in this enantioconvergent reaction: [6]

The scope is extended to asymmetric α-alkylation of ketones masked as their enol carbonate esters: [7]

References
- ^ Carroll, M. F. "131. Addition of α,β-unsaturated alcohols to the active methylene group. Part I. The action of ethyl acetoacetate on linalool and geraniol". Journal of the Chemical Society 1940, 704–706. doi: 10.1039/JR9400000704
- ^ Palladium-catalyzed rearrangement of allylic esters of acetoacetic acid to give γ,δ-unsaturated methyl ketones Tetrahedron Letters, Volume 21, Issue 33, 1980, Pages 3199-3202 Isao Shimizu, Toshiro Yamada and Jiro Tsuji doi: 10.1016/S0040-4039(00)77444-2
- ^ a b Asymmetric Allylic Alkylation of Ketone Enolates: An Asymmetric Claisen Surrogate Erin C. Burger and Jon A. Tunge Org. Lett.; 2004; 6(22) pp 4113 - 4115; (Letter) doi: 10.1021/ol048149t
- ^ Enantioselective Palladium-Catalyzed Decarboxylative Allylic Alkylations Shu-Li You and Li-Xin Dai Angew. Chem. Int. Ed. 2006, 45, 5246 – 5248 doi: 10.1002/anie.200601889
- ^ Kuwano, R.; Ishida N.; Murakami, M. "Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex". Chemical Communications 2005, (31), 3951–3952. doi: 10.1039/b505105c
- ^ Deracemization of Quaternary Stereocenters by Pd-Catalyzed Enantioconvergent Decarboxylative Allylation of Racemic b-Ketoesters Justin T. Mohr, Douglas C. Behenna, Andrew M. Harned, and Brian M. Stoltz Angew. Chem. Int. Ed. 2005, 44, 6924 –6927 doi: 10.1002/anie.200502018
- ^ Palladium-Catalyzed Asymmetric Allylic α-Alkylation of Acyclic Ketones Barry M. Trost and Jiayi Xu J. Am. Chem. Soc.; 2005; 127(49) pp 17180 - 17181; (Communication) doi: 10.1021/ja055968f