| |
| Names | |
|---|---|
| Other names
TL-358
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
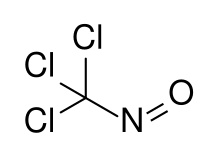
| CCl3NO | |
| Molar mass | 148.37 g·mol−1 |
| Appearance | Deep blue liquid |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Trichloronitrosomethane is a chlorinated nitrosoalkane. It is a deep blue liquid with powerful lachrymatory effects. [1]
Trichloronitrosomethane can be produced with following methods: [1] [2]
- Oxidation of trichloromethylsulfinic acid with nitric acid.
- Reaction of sodium trichloromethylsulfinate with sodium nitrite and sodium nitrate or potassium nitrate in sulfuric acid.
- Pyrolysis of trichloroacethydroxamic acid.
Trichloronitrosomethane is an unstable substance. It slowly decomposes into nitrosyl chloride, nitrogen oxides, and chloropicrin over time. [1]
Trichloronitrosomethane can be reduced to phosgene oxime by hydrogen sulfide. [1]

| |
| Names | |
|---|---|
| Other names
TL-358
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| CCl3NO | |
| Molar mass | 148.37 g·mol−1 |
| Appearance | Deep blue liquid |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Trichloronitrosomethane is a chlorinated nitrosoalkane. It is a deep blue liquid with powerful lachrymatory effects. [1]
Trichloronitrosomethane can be produced with following methods: [1] [2]
- Oxidation of trichloromethylsulfinic acid with nitric acid.
- Reaction of sodium trichloromethylsulfinate with sodium nitrite and sodium nitrate or potassium nitrate in sulfuric acid.
- Pyrolysis of trichloroacethydroxamic acid.
Trichloronitrosomethane is an unstable substance. It slowly decomposes into nitrosyl chloride, nitrogen oxides, and chloropicrin over time. [1]
Trichloronitrosomethane can be reduced to phosgene oxime by hydrogen sulfide. [1]