The Barton‚ÄďZard reaction is a route to pyrrole derivatives via the reaction of a nitroalkene with an őĪ-isocyanide under basic conditions. [1] It is named after Derek Barton and Samir Zard who first reported it in 1985. [2]

The mechanism consists of five steps:
- Base catalyzed carbonyl enolization of the őĪ- isocyanide.
- Michael-type addition between the őĪ- isocyanide carbonyl enolate and the nitroalkene.
- 5-endo-dig cyclization (see: Baldwin's rules).
- Base catalyzed elimination of the nitro group.
- Tautomerization leading to aromatisation.
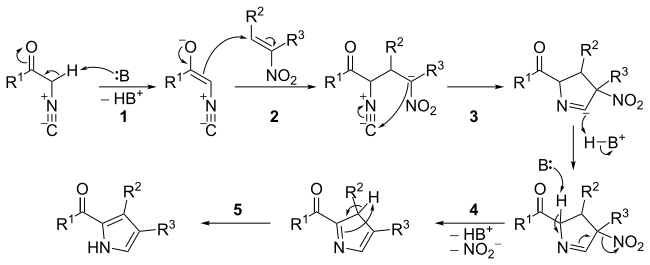
The nitro compound may be aromatic rather than just an alkene. [3] The reaction has been used for the synthesis of polypyrroles, including porphyrins, [4] as well as dipyrromethenes such as BODIPY. [5]
- ^ Jie Jack Li (2013). Heterocyclic Chemistry in Drug Discovery. New York: Wiley. ISBN 9781118354421. pp.43-4
- ^ Barton, Derek H. R.; Zard, Samir Z. (1985). "A new synthesis of pyrroles from nitroalkenes". Journal of the Chemical Society, Chemical Communications (16): 1098. doi: 10.1039/C39850001098.
- ^ Lash, Timothy D.; Novak, Bennett H.; Lin, Yanning (April 1994). "Synthesis of phenanthropyrroles and phenanthrolinopyrroles from isocyanoacetates: An extension of the barton-zard pyrrole condensation". Tetrahedron Letters. 35 (16): 2493‚Äď2494. doi: 10.1016/S0040-4039(00)77152-8.
- ^ Finikova, Olga S.; Cheprakov, Andrei V.; Beletskaya, Irina P.; Carroll, Patrick J.; Vinogradov, Sergei A. (January 2004). "Novel Versatile Synthesis of Substituted Tetrabenzoporphyrins". The Journal of Organic Chemistry. 69 (2): 522‚Äď535. doi: 10.1021/jo0350054. PMID 14725469.
-
^ Ono, Noboru (2008). "Barton-Zard Pyrrole Synthesis and Its Application to Synthesis of Porphyrins, Polypyrroles, and Dipyrromethene Dyes". Heterocycles. 75 (2): 243.
doi:
10.3987/REV-07-622 (inactive 2024-03-07).
{{ cite journal}}: CS1 maint: DOI inactive as of March 2024 ( link)
The Barton‚ÄďZard reaction is a route to pyrrole derivatives via the reaction of a nitroalkene with an őĪ-isocyanide under basic conditions. [1] It is named after Derek Barton and Samir Zard who first reported it in 1985. [2]

The mechanism consists of five steps:
- Base catalyzed carbonyl enolization of the őĪ- isocyanide.
- Michael-type addition between the őĪ- isocyanide carbonyl enolate and the nitroalkene.
- 5-endo-dig cyclization (see: Baldwin's rules).
- Base catalyzed elimination of the nitro group.
- Tautomerization leading to aromatisation.

The nitro compound may be aromatic rather than just an alkene. [3] The reaction has been used for the synthesis of polypyrroles, including porphyrins, [4] as well as dipyrromethenes such as BODIPY. [5]
- ^ Jie Jack Li (2013). Heterocyclic Chemistry in Drug Discovery. New York: Wiley. ISBN 9781118354421. pp.43-4
- ^ Barton, Derek H. R.; Zard, Samir Z. (1985). "A new synthesis of pyrroles from nitroalkenes". Journal of the Chemical Society, Chemical Communications (16): 1098. doi: 10.1039/C39850001098.
- ^ Lash, Timothy D.; Novak, Bennett H.; Lin, Yanning (April 1994). "Synthesis of phenanthropyrroles and phenanthrolinopyrroles from isocyanoacetates: An extension of the barton-zard pyrrole condensation". Tetrahedron Letters. 35 (16): 2493‚Äď2494. doi: 10.1016/S0040-4039(00)77152-8.
- ^ Finikova, Olga S.; Cheprakov, Andrei V.; Beletskaya, Irina P.; Carroll, Patrick J.; Vinogradov, Sergei A. (January 2004). "Novel Versatile Synthesis of Substituted Tetrabenzoporphyrins". The Journal of Organic Chemistry. 69 (2): 522‚Äď535. doi: 10.1021/jo0350054. PMID 14725469.
-
^ Ono, Noboru (2008). "Barton-Zard Pyrrole Synthesis and Its Application to Synthesis of Porphyrins, Polypyrroles, and Dipyrromethene Dyes". Heterocycles. 75 (2): 243.
doi:
10.3987/REV-07-622 (inactive 2024-03-07).
{{ cite journal}}: CS1 maint: DOI inactive as of March 2024 ( link)
Topics in
organic reactions | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||