 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C18H23IO2 |
| Molar mass | 398.284 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
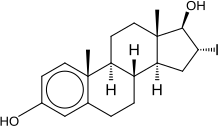
16α-Iodo-E2, or 16α-iodoestradiol, is a synthetic, steroidal, potent estrogen with slight preference for the ERα over the ERβ that is used in scientific research. [1] [2] The KD of 16α-iodo-E2 for the ERα is 0.6 nM and for the ERβ is 0.24 nM, a 4-fold difference in affinity, whereas estradiol is considered to have similar affinity for the two receptor subtypes. [2] Unlike the case of the much weaker estriol (16α-hydroxyestradiol), 16α-iodo-E2 is considered to be equipotent with estradiol in terms of estrogenic activity. [3] Radiolabeled [16α-125I]iodo-E2 has been employed in imaging to study the estrogen receptor. [4]
See also
References
- ^ Manas ES, Unwalla RJ, Xu ZB, Malamas MS, Miller CP, Harris HA, Hsiao C, Akopian T, Hum WT, Malakian K, Wolfrom S, Bapat A, Bhat RA, Stahl ML, Somers WS, Alvarez JC (2004). "Structure-based design of estrogen receptor-beta selective ligands". J. Am. Chem. Soc. 126 (46): 15106–19. doi: 10.1021/ja047633o. PMID 15548008.
- ^ a b Chen GG, Vlantis AC, Zeng Q, van Hasselt CA (2008). "Regulation of cell growth by estrogen signaling and potential targets in thyroid cancer". Curr Cancer Drug Targets. 8 (5): 367–77. doi: 10.2174/156800908785133150. PMID 18690843.
- ^ Hochberg RB, Zielinski JE, Duax WL, Strong P (1986). "The molecular structure of 16 alpha-iodo-17 beta-estradiol, a high affinity ligand for the estrogen receptor". J. Steroid Biochem. 25 (5A): 615–8. doi: 10.1016/0022-4731(86)90002-6. PMID 3795941.
- ^ Cummins CH (1993). "Radiolabeled steroidal estrogens in cancer research". Steroids. 58 (6): 245–59. doi: 10.1016/0039-128x(93)90069-y. PMID 8212070. S2CID 29080385.
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C18H23IO2 |
| Molar mass | 398.284 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
16α-Iodo-E2, or 16α-iodoestradiol, is a synthetic, steroidal, potent estrogen with slight preference for the ERα over the ERβ that is used in scientific research. [1] [2] The KD of 16α-iodo-E2 for the ERα is 0.6 nM and for the ERβ is 0.24 nM, a 4-fold difference in affinity, whereas estradiol is considered to have similar affinity for the two receptor subtypes. [2] Unlike the case of the much weaker estriol (16α-hydroxyestradiol), 16α-iodo-E2 is considered to be equipotent with estradiol in terms of estrogenic activity. [3] Radiolabeled [16α-125I]iodo-E2 has been employed in imaging to study the estrogen receptor. [4]
See also
References
- ^ Manas ES, Unwalla RJ, Xu ZB, Malamas MS, Miller CP, Harris HA, Hsiao C, Akopian T, Hum WT, Malakian K, Wolfrom S, Bapat A, Bhat RA, Stahl ML, Somers WS, Alvarez JC (2004). "Structure-based design of estrogen receptor-beta selective ligands". J. Am. Chem. Soc. 126 (46): 15106–19. doi: 10.1021/ja047633o. PMID 15548008.
- ^ a b Chen GG, Vlantis AC, Zeng Q, van Hasselt CA (2008). "Regulation of cell growth by estrogen signaling and potential targets in thyroid cancer". Curr Cancer Drug Targets. 8 (5): 367–77. doi: 10.2174/156800908785133150. PMID 18690843.
- ^ Hochberg RB, Zielinski JE, Duax WL, Strong P (1986). "The molecular structure of 16 alpha-iodo-17 beta-estradiol, a high affinity ligand for the estrogen receptor". J. Steroid Biochem. 25 (5A): 615–8. doi: 10.1016/0022-4731(86)90002-6. PMID 3795941.
- ^ Cummins CH (1993). "Radiolabeled steroidal estrogens in cancer research". Steroids. 58 (6): 245–59. doi: 10.1016/0039-128x(93)90069-y. PMID 8212070. S2CID 29080385.