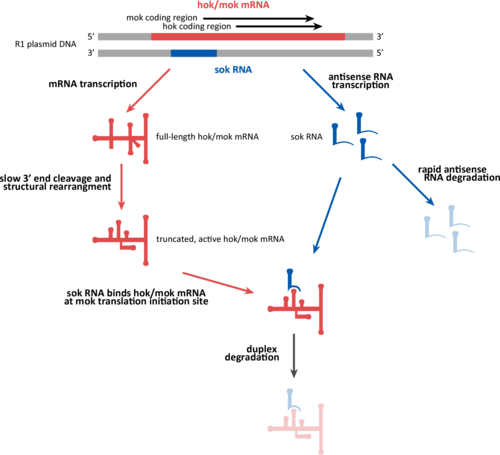
The hok/sok system is a postsegregational killing mechanism employed by the R1 plasmid in Escherichia coli. It was the first type I toxin-antitoxin pair to be identified through characterisation of a plasmid-stabilising locus. [1] It is a type I system because the toxin is neutralised by a complementary RNA, rather than a partnered protein (type II toxin-antitoxin). [2]
The hok/sok system features two transcripts:
- a messenger RNA (mRNA) which encodes the hok and mok proteins (but is referred to as the hok mRNA)
- an antisense RNA known as sok
The hok (host killing) gene of the mRNA encodes a toxic transmembrane protein. The hok and mok (modulation of killing) protein-coding regions in the mRNA are partially overlapping, and their translation is coupled, such that the ribosome first translates the mok reading frame and then slides back to translate the hok gene. [3] The sok (suppression of killing) antisense RNA is complementary to a portion of the mRNA corresponding to the hok 5' untranslated region and the mok translation initiation region (TIR). Upon binding to the hok/mok mRNA, sok inhibits translation of the Mok protein and thus indirectly prevents translation of the Hok protein toxin. [4]
The full-length hok mRNA is stable ( half-life 20 minutes), while the sok RNA is short lived (half-life 30 seconds).
| HOK | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | HOK | ||||||||
| Pfam | PF01848 | ||||||||
| InterPro | IPR000021 | ||||||||
| PROSITE | PDOC00481 | ||||||||
| |||||||||
Hok mRNA is found in two forms:
- stable and inactive full-length mRNA
- active truncated mRNA
The full-length hok transcript is highly structured and immune to both ribosome binding (which initiates Hok toxin translation) and sok antisense binding (which leads to degradation of both transcripts). The full-length hok transcript is slowly processed - 39 nucleotides are cleaved from the 3' end of the molecule. Processing initiates drastic structural rearrangments in the hok transcript, such that the ribosome and sok RNA binding sites become accessible. [5]
When E. coli undergoes cell division, the two daughter cells inherit the stable full-length hok mRNA from the parent cell. While daughter cells also inheret sok RNA antitoxin, due to their short half-life these molecules quickly degrade. [6] Meanwhile, the full-length hok mRNA is slowly processed to the truncated active form.
If a daughter cell has inherited the R1 plasmid, it has inherited the sok gene, which has a strong promoter that brings about high levels of transcription. [7] The newly transcribed Sok RNA then inhibits the translation of Hok protein by binding to the active truncated mRNA. The sok transcript forms a duplex with the leader region of the truncated hok mRNA and this is recognized by RNase III and degraded. The cleavage products are very unstable and soon decay. [8]
Daughter cells without a copy of the R1 plasmid die because they do not have the means to produce more sok antitoxin transcript to inhibit translation of the inherited hok mRNA. The killing system is said to be postsegregational (PSK), [9] since cell death occurs after segregation of the plasmid. [10] [11]
During transcription of the hok mRNA, the ribosome and sok binding sites emerge from the transcriptional machinery before the stable full-length structure can form. Nevertheless, premature translation and degredation are prevented by the formation of transient structures which sequester these binding sites until the molecule is fully synthesized. [12]
The hok gene codes for a 52 amino acid toxic protein which causes cell death by depolarization of the cell membrane. [13] [14] It works in a similar way to holin proteins which are produced by bacteriophages before cell lysis. [2] [15]
hok/sok homologues denoted flmA/B (FlmA is the protein toxin and FlmB RNA the antisense regulator) [16] are carried on the F plasmid which operate in the same way to maintain the stability of the plasmid. [17] The F plasmid contains another homologous toxin-antitoxin system called srnB. [13]
The first type I toxin-antitoxin system to be found in gram-positive bacteria is the RNAI-RNAII system of the pAD1 plasmid in Enterococcus faecalis. Here, RNAI encodes a toxic protein Fst while RNAII is the regulatory sRNA. [18]
In E. coli strain K-12 there are four long direct repeats (ldr) which encode short open reading frames of 35 codons organised in a homologous manner to the hok/sok system. One of the repeats encodes LdrD, a toxic protein which causes cell death. An unstable antisense RNA regulator (Rd1D) blocks the translation of the LdrD transcript. [19] A mok homologue which overlaps each ldr loci has also been found. [6]
IstR RNA works in a similar system in conjunction with the toxic TisB protein. [20]
-
^ Gerdes K, Larsen JE, Molin S (January 1985).
"Stable inheritance of plasmid R1 requires two different loci". J. Bacteriol. 161 (1): 292–8.
doi:
10.1128/jb.161.1.292-298.1985.
PMC
214870.
PMID
2981804. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) - ^
a
b Hayes F (September 2003).
"Toxins-antitoxins: plasmid maintenance, programmed cell death, and cell cycle arrest". Science. 301 (5639): 1496–9.
doi:
10.1126/science.1088157.
PMID
12970556. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Thisted T, Gerdes K (January 1992).
"Mechanism of post-segregational killing by the hok/sok system of plasmid R1. Sok antisense RNA regulates hok gene expression indirectly through the overlapping mok gene". J. Mol. Biol. 223 (1): 41–54.
doi:
10.1016/0022-2836(92)90714-U.
PMID
1370544. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Thisted T, Gerdes K (January 1992).
"Mechanism of post-segregational killing by the hok/sok system of plasmid R1. Sok antisense RNA regulates hok gene expression indirectly through the overlapping mok gene". J. Mol. Biol. 223 (1): 41–54.
doi:
10.1016/0022-2836(92)90714-U.
PMID
1370544. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Franch T, Gultyaev A, Gerdes K (1997). "Programmed cell death by hok/sok of plasmid R1: Processing at the hok mRNA 3'-end triggers structural rearrangements that allow translation and antisense RNA binding". J. Mol. Biol. 273 (1): 38–51.
doi:
10.1006/jmbi.1997.1294.
PMID
9367744.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^
a
b Gerdes K, Wagner EG (April 2007).
"RNA antitoxins". Curr. Opin. Microbiol. 10 (2): 117–24.
doi:
10.1016/j.mib.2007.03.003.
PMID
17376733. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Gerdes K, Thisted T, Martinussen J (November 1990). "Mechanism of post-segregational killing by the hok/sok system of plasmid R1: sok antisense RNA regulates formation of a hok mRNA species correlated with killing of plasmid-free cells". Mol. Microbiol. 4 (11): 1807–18.
doi:
10.1111/j.1365-2958.1990.tb02029.x.
PMID
1707122.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Gerdes K, Nielsen A, Thorsted P, Wagner EG (August 1992).
"Mechanism of killer gene activation. Antisense RNA-dependent RNase III cleavage ensures rapid turn-over of the stable hok, srnB and pndA effector messenger RNAs". J. Mol. Biol. 226 (3): 637–49.
doi:
10.1016/0022-2836(92)90621-P.
PMID
1380562. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Gerdes K, Rasmussen PB, Molin S (May 1986).
"Unique type of plasmid maintenance function: postsegregational killing of plasmid-free cells". Proc. Natl. Acad. Sci. U.S.A. 83 (10): 3116–20.
doi:
10.1073/pnas.83.10.3116.
PMC
323463.
PMID
3517851.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Thisted T, Sørensen NS, Gerdes K (1995). "Mechanism of post-segregational killing: secondary structure analysis of the entire Hok mRNA from plasmid R1 suggests a fold-back structure that prevents translation and antisense RNA binding". J. Mol. Biol. 247 (5): 859–73.
doi:
10.1006/jmbi.1995.0186.
PMID
7536849.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Gerdes K; Bech FW; Jørgensen ST; et al. (August 1986).
"Mechanism of postsegregational killing by the hok gene product of the parB system of plasmid R1 and its homology with the relF gene product of the E. coli relB operon". EMBO J. 5 (8): 2023–9.
doi:
10.1002/j.1460-2075.1986.tb04459.x.
PMC
1167073.
PMID
3019679.
{{ cite journal}}: Unknown parameter|author-separator=ignored ( help)CS1 maint: date and year ( link) -
^ Gultyaev A, Franch T, Gerdes K (1997). "Programmed cell death by hok/sok of plasmid R1: Coupled nucleotide covariations reveal a phylogenetically conserved folding pathway in the hok family of mRNAs". J. Mol. Biol. 273 (1): 26–37.
doi:
10.1006/jmbi.1997.1295.
PMID
9367743.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^
a
b Gerdes K, Poulsen LK, Thisted T, Nielsen AK, Martinussen J, Andreasen PH (November 1990). "The hok killer gene family in gram-negative bacteria". New Biol. 2 (11): 946–56.
PMID
2101633.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Pecota DC, Osapay G, Selsted ME, Wood TK (2003). "Antimicrobial properties of the Escherichia coli R1 plasmid host killing peptide". J. Biotechnol. 100 (1): 1–12.
doi:
10.1016/S0168-1656(02)00240-7.
PMID
12413781.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Wang IN, Smith DL, Young R (2000).
"Holins: the protein clocks of bacteriophage infections". Annu. Rev. Microbiol. 54: 799–825.
doi:
10.1146/annurev.micro.54.1.799.
PMID
11018145. Retrieved 2010-08-19.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Loh SM, Cram DS, Skurray RA (June 1988). "Nucleotide sequence and transcriptional analysis of a third function (Flm) involved in F-plasmid maintenance". Gene. 66 (2): 259–68.
doi:
10.1016/0378-1119(88)90362-9.
PMID
3049248.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Pedersen K, Gerdes K (June 1999).
"Multiple hok genes on the chromosome of Escherichia coli". Mol. Microbiol. 32 (5): 1090–102.
doi:
10.1046/j.1365-2958.1999.01431.x.
PMID
10361310. Retrieved 2010-08-09.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Greenfield TJ, Ehli E, Kirshenmann T, Franch T, Gerdes K, Weaver KE (August 2000).
"The antisense RNA of the par locus of pAD1 regulates the expression of a 33-amino-acid toxic peptide by an unusual mechanism". Mol. Microbiol. 37 (3): 652–60.
doi:
10.1046/j.1365-2958.2000.02035.x.
PMID
10931358. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Kawano M, Oshima T, Kasai H, Mori H (July 2002).
"Molecular characterization of long direct repeat (LDR) sequences expressing a stable mRNA encoding for a 35-amino-acid cell-killing peptide and a cis-encoded small antisense RNA in Escherichia coli". Mol. Microbiol. 45 (2): 333–49.
doi:
10.1046/j.1365-2958.2002.03042.x.
PMID
12123448. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Darfeuille F, Unoson C, Vogel J, Wagner EG (May 2007).
"An antisense RNA inhibits translation by competing with standby ribosomes". Mol. Cell. 26 (3): 381–92.
doi:
10.1016/j.molcel.2007.04.003.
PMID
17499044. Retrieved 2010-08-11.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link)
- Gerdes K, Gultyaev AP, Franch T, Pedersen K, Mikkelsen ND (1997).
"Antisense RNA-regulated programmed cell death". Annu. Rev. Genet. 31: 1–31.
doi:
10.1146/annurev.genet.31.1.1.
PMID
9442888. Retrieved 2010-08-09.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - Poulsen LK, Larsen NW, Molin S, Andersson P (November 1989). "A family of genes encoding a cell-killing function may be conserved in all gram-negative bacteria". Mol. Microbiol. 3 (11): 1463–72.
doi:
10.1111/j.1365-2958.1989.tb00131.x.
PMID
2693900.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) - Nagel JH, Gultyaev AP, Gerdes K, Pleij CW (November 1999).
"Metastable structures and refolding kinetics in hok mRNA of plasmid R1". RNA. 5 (11): 1408–18.
doi:
10.1017/S1355838299990805.
PMC
1369862.
PMID
10580469. Retrieved 2010-08-09.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link)
{{DEFAULTSORT:Hok/Sok System}} [[Category:Bacteriology]] [[Category:Escherichia coli]] [[Category:RNA antitoxins]]
The hok/sok system is a postsegregational killing mechanism employed by the R1 plasmid in Escherichia coli. It was the first type I toxin-antitoxin pair to be identified through characterisation of a plasmid-stabilising locus. [1] It is a type I system because the toxin is neutralised by a complementary RNA, rather than a partnered protein (type II toxin-antitoxin). [2]

The hok/sok system features two transcripts:
- a messenger RNA (mRNA) which encodes the hok and mok proteins (but is referred to as the hok mRNA)
- an antisense RNA known as sok
The hok (host killing) gene of the mRNA encodes a toxic transmembrane protein. The hok and mok (modulation of killing) protein-coding regions in the mRNA are partially overlapping, and their translation is coupled, such that the ribosome first translates the mok reading frame and then slides back to translate the hok gene. [3] The sok (suppression of killing) antisense RNA is complementary to a portion of the mRNA corresponding to the hok 5' untranslated region and the mok translation initiation region (TIR). Upon binding to the hok/mok mRNA, sok inhibits translation of the Mok protein and thus indirectly prevents translation of the Hok protein toxin. [4]
The full-length hok mRNA is stable ( half-life 20 minutes), while the sok RNA is short lived (half-life 30 seconds).
| HOK | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | HOK | ||||||||
| Pfam | PF01848 | ||||||||
| InterPro | IPR000021 | ||||||||
| PROSITE | PDOC00481 | ||||||||
| |||||||||
Hok mRNA is found in two forms:
- stable and inactive full-length mRNA
- active truncated mRNA
The full-length hok transcript is highly structured and immune to both ribosome binding (which initiates Hok toxin translation) and sok antisense binding (which leads to degradation of both transcripts). The full-length hok transcript is slowly processed - 39 nucleotides are cleaved from the 3' end of the molecule. Processing initiates drastic structural rearrangments in the hok transcript, such that the ribosome and sok RNA binding sites become accessible. [5]
When E. coli undergoes cell division, the two daughter cells inherit the stable full-length hok mRNA from the parent cell. While daughter cells also inheret sok RNA antitoxin, due to their short half-life these molecules quickly degrade. [6] Meanwhile, the full-length hok mRNA is slowly processed to the truncated active form.
If a daughter cell has inherited the R1 plasmid, it has inherited the sok gene, which has a strong promoter that brings about high levels of transcription. [7] The newly transcribed Sok RNA then inhibits the translation of Hok protein by binding to the active truncated mRNA. The sok transcript forms a duplex with the leader region of the truncated hok mRNA and this is recognized by RNase III and degraded. The cleavage products are very unstable and soon decay. [8]
Daughter cells without a copy of the R1 plasmid die because they do not have the means to produce more sok antitoxin transcript to inhibit translation of the inherited hok mRNA. The killing system is said to be postsegregational (PSK), [9] since cell death occurs after segregation of the plasmid. [10] [11]
During transcription of the hok mRNA, the ribosome and sok binding sites emerge from the transcriptional machinery before the stable full-length structure can form. Nevertheless, premature translation and degredation are prevented by the formation of transient structures which sequester these binding sites until the molecule is fully synthesized. [12]
The hok gene codes for a 52 amino acid toxic protein which causes cell death by depolarization of the cell membrane. [13] [14] It works in a similar way to holin proteins which are produced by bacteriophages before cell lysis. [2] [15]
hok/sok homologues denoted flmA/B (FlmA is the protein toxin and FlmB RNA the antisense regulator) [16] are carried on the F plasmid which operate in the same way to maintain the stability of the plasmid. [17] The F plasmid contains another homologous toxin-antitoxin system called srnB. [13]
The first type I toxin-antitoxin system to be found in gram-positive bacteria is the RNAI-RNAII system of the pAD1 plasmid in Enterococcus faecalis. Here, RNAI encodes a toxic protein Fst while RNAII is the regulatory sRNA. [18]
In E. coli strain K-12 there are four long direct repeats (ldr) which encode short open reading frames of 35 codons organised in a homologous manner to the hok/sok system. One of the repeats encodes LdrD, a toxic protein which causes cell death. An unstable antisense RNA regulator (Rd1D) blocks the translation of the LdrD transcript. [19] A mok homologue which overlaps each ldr loci has also been found. [6]
IstR RNA works in a similar system in conjunction with the toxic TisB protein. [20]
-
^ Gerdes K, Larsen JE, Molin S (January 1985).
"Stable inheritance of plasmid R1 requires two different loci". J. Bacteriol. 161 (1): 292–8.
doi:
10.1128/jb.161.1.292-298.1985.
PMC
214870.
PMID
2981804. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) - ^
a
b Hayes F (September 2003).
"Toxins-antitoxins: plasmid maintenance, programmed cell death, and cell cycle arrest". Science. 301 (5639): 1496–9.
doi:
10.1126/science.1088157.
PMID
12970556. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Thisted T, Gerdes K (January 1992).
"Mechanism of post-segregational killing by the hok/sok system of plasmid R1. Sok antisense RNA regulates hok gene expression indirectly through the overlapping mok gene". J. Mol. Biol. 223 (1): 41–54.
doi:
10.1016/0022-2836(92)90714-U.
PMID
1370544. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Thisted T, Gerdes K (January 1992).
"Mechanism of post-segregational killing by the hok/sok system of plasmid R1. Sok antisense RNA regulates hok gene expression indirectly through the overlapping mok gene". J. Mol. Biol. 223 (1): 41–54.
doi:
10.1016/0022-2836(92)90714-U.
PMID
1370544. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Franch T, Gultyaev A, Gerdes K (1997). "Programmed cell death by hok/sok of plasmid R1: Processing at the hok mRNA 3'-end triggers structural rearrangements that allow translation and antisense RNA binding". J. Mol. Biol. 273 (1): 38–51.
doi:
10.1006/jmbi.1997.1294.
PMID
9367744.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^
a
b Gerdes K, Wagner EG (April 2007).
"RNA antitoxins". Curr. Opin. Microbiol. 10 (2): 117–24.
doi:
10.1016/j.mib.2007.03.003.
PMID
17376733. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Gerdes K, Thisted T, Martinussen J (November 1990). "Mechanism of post-segregational killing by the hok/sok system of plasmid R1: sok antisense RNA regulates formation of a hok mRNA species correlated with killing of plasmid-free cells". Mol. Microbiol. 4 (11): 1807–18.
doi:
10.1111/j.1365-2958.1990.tb02029.x.
PMID
1707122.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Gerdes K, Nielsen A, Thorsted P, Wagner EG (August 1992).
"Mechanism of killer gene activation. Antisense RNA-dependent RNase III cleavage ensures rapid turn-over of the stable hok, srnB and pndA effector messenger RNAs". J. Mol. Biol. 226 (3): 637–49.
doi:
10.1016/0022-2836(92)90621-P.
PMID
1380562. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Gerdes K, Rasmussen PB, Molin S (May 1986).
"Unique type of plasmid maintenance function: postsegregational killing of plasmid-free cells". Proc. Natl. Acad. Sci. U.S.A. 83 (10): 3116–20.
doi:
10.1073/pnas.83.10.3116.
PMC
323463.
PMID
3517851.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Thisted T, Sørensen NS, Gerdes K (1995). "Mechanism of post-segregational killing: secondary structure analysis of the entire Hok mRNA from plasmid R1 suggests a fold-back structure that prevents translation and antisense RNA binding". J. Mol. Biol. 247 (5): 859–73.
doi:
10.1006/jmbi.1995.0186.
PMID
7536849.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Gerdes K; Bech FW; Jørgensen ST; et al. (August 1986).
"Mechanism of postsegregational killing by the hok gene product of the parB system of plasmid R1 and its homology with the relF gene product of the E. coli relB operon". EMBO J. 5 (8): 2023–9.
doi:
10.1002/j.1460-2075.1986.tb04459.x.
PMC
1167073.
PMID
3019679.
{{ cite journal}}: Unknown parameter|author-separator=ignored ( help)CS1 maint: date and year ( link) -
^ Gultyaev A, Franch T, Gerdes K (1997). "Programmed cell death by hok/sok of plasmid R1: Coupled nucleotide covariations reveal a phylogenetically conserved folding pathway in the hok family of mRNAs". J. Mol. Biol. 273 (1): 26–37.
doi:
10.1006/jmbi.1997.1295.
PMID
9367743.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^
a
b Gerdes K, Poulsen LK, Thisted T, Nielsen AK, Martinussen J, Andreasen PH (November 1990). "The hok killer gene family in gram-negative bacteria". New Biol. 2 (11): 946–56.
PMID
2101633.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Pecota DC, Osapay G, Selsted ME, Wood TK (2003). "Antimicrobial properties of the Escherichia coli R1 plasmid host killing peptide". J. Biotechnol. 100 (1): 1–12.
doi:
10.1016/S0168-1656(02)00240-7.
PMID
12413781.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Wang IN, Smith DL, Young R (2000).
"Holins: the protein clocks of bacteriophage infections". Annu. Rev. Microbiol. 54: 799–825.
doi:
10.1146/annurev.micro.54.1.799.
PMID
11018145. Retrieved 2010-08-19.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Loh SM, Cram DS, Skurray RA (June 1988). "Nucleotide sequence and transcriptional analysis of a third function (Flm) involved in F-plasmid maintenance". Gene. 66 (2): 259–68.
doi:
10.1016/0378-1119(88)90362-9.
PMID
3049248.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Pedersen K, Gerdes K (June 1999).
"Multiple hok genes on the chromosome of Escherichia coli". Mol. Microbiol. 32 (5): 1090–102.
doi:
10.1046/j.1365-2958.1999.01431.x.
PMID
10361310. Retrieved 2010-08-09.
{{ cite journal}}: CS1 maint: date and year ( link) -
^ Greenfield TJ, Ehli E, Kirshenmann T, Franch T, Gerdes K, Weaver KE (August 2000).
"The antisense RNA of the par locus of pAD1 regulates the expression of a 33-amino-acid toxic peptide by an unusual mechanism". Mol. Microbiol. 37 (3): 652–60.
doi:
10.1046/j.1365-2958.2000.02035.x.
PMID
10931358. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Kawano M, Oshima T, Kasai H, Mori H (July 2002).
"Molecular characterization of long direct repeat (LDR) sequences expressing a stable mRNA encoding for a 35-amino-acid cell-killing peptide and a cis-encoded small antisense RNA in Escherichia coli". Mol. Microbiol. 45 (2): 333–49.
doi:
10.1046/j.1365-2958.2002.03042.x.
PMID
12123448. Retrieved 2010-08-10.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Darfeuille F, Unoson C, Vogel J, Wagner EG (May 2007).
"An antisense RNA inhibits translation by competing with standby ribosomes". Mol. Cell. 26 (3): 381–92.
doi:
10.1016/j.molcel.2007.04.003.
PMID
17499044. Retrieved 2010-08-11.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link)
- Gerdes K, Gultyaev AP, Franch T, Pedersen K, Mikkelsen ND (1997).
"Antisense RNA-regulated programmed cell death". Annu. Rev. Genet. 31: 1–31.
doi:
10.1146/annurev.genet.31.1.1.
PMID
9442888. Retrieved 2010-08-09.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - Poulsen LK, Larsen NW, Molin S, Andersson P (November 1989). "A family of genes encoding a cell-killing function may be conserved in all gram-negative bacteria". Mol. Microbiol. 3 (11): 1463–72.
doi:
10.1111/j.1365-2958.1989.tb00131.x.
PMID
2693900.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) - Nagel JH, Gultyaev AP, Gerdes K, Pleij CW (November 1999).
"Metastable structures and refolding kinetics in hok mRNA of plasmid R1". RNA. 5 (11): 1408–18.
doi:
10.1017/S1355838299990805.
PMC
1369862.
PMID
10580469. Retrieved 2010-08-09.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link)
{{DEFAULTSORT:Hok/Sok System}} [[Category:Bacteriology]] [[Category:Escherichia coli]] [[Category:RNA antitoxins]]