| |

| |
| Names | |
|---|---|
|
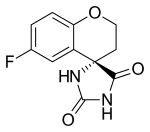
Preferred IUPAC name
(4S)-6-Fluoro-2,3-dihydrospiro[[1]benzopyran-4,4′-imidazolidine]-2,5-dione | |
| Other names
(S)-6-Fluorospiro(chroman-4,4'-imidazolidine)-2',5'-dione; CP 45634
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.210.173 |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties [1] | |
| C11H9FN2O3 | |
| Molar mass | 236.202 g·mol−1 |
| Appearance | White to off-white powder |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Sorbinil ( INN) is an aldose reductase inhibitor being investigated for treatment of diabetic complications including neuropathy and retinopathy. [3] Aldose reductase is an enzyme present in lens and brain that removes excess glucose by converting it to sorbitol. Sorbitol accumulation can lead to the development of cataracts in the lens and neuropathy in peripheral nerves. Sorbinil has been shown to inhibit aldose reductase in human brain [4] and placenta [5] and calf [6] and rat lens. [5] Sorbinil reduced sorbitol accumulation in rat lens and sciatic nerve of diabetic rats orally administered 0.25 mg/kg sorbinil. [5]
References
- ^ Sorbinil at Sigma-Aldrich
- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary names (Rec. INN): List 25" (PDF). World Health Organization. 1985. Retrieved 11 November 2016.
- ^ MacCari, R; Del Corso, A; Giglio, M; Moschini, R; Mura, U; Ottanà, R (2011). "In vitro evaluation of 5-arylidene-2-thioxo-4-thiazolidinones active as aldose reductase inhibitors". Bioorganic & Medicinal Chemistry Letters. 21 (1): 200–3. doi: 10.1016/j.bmcl.2010.11.041. PMID 21129963.
- ^ O'Brien MM; et al. (1982). "Inhibition of human brain aldose reductase and hexonate dehydrogenase by alrestatin and sorbinil". J Neurochem. 39 (3): 810–4. doi: 10.1111/j.1471-4159.1982.tb07964.x. PMID 6808090. S2CID 22368580.
- ^ a b c Kinoshita JH; et al. (1979). "Aldose reductase in diabetic complications of the eye". Metabolism. 28 (suppl 1): 462–9. doi: 10.1016/0026-0495(79)90057-x. PMID 45423.
- ^ Peterson MJ; et al. (1979). "CP45,634: A novel aldose reductase inhibitor that inhibits polyol pathway activity in diabetic and galactosemic rats". Metabolism. 28 (Suppl 1): 456–61. doi: 10.1016/0026-0495(79)90056-8. PMID 122297.

| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(4S)-6-Fluoro-2,3-dihydrospiro[[1]benzopyran-4,4′-imidazolidine]-2,5-dione | |
| Other names
(S)-6-Fluorospiro(chroman-4,4'-imidazolidine)-2',5'-dione; CP 45634
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.210.173 |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties [1] | |
| C11H9FN2O3 | |
| Molar mass | 236.202 g·mol−1 |
| Appearance | White to off-white powder |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Sorbinil ( INN) is an aldose reductase inhibitor being investigated for treatment of diabetic complications including neuropathy and retinopathy. [3] Aldose reductase is an enzyme present in lens and brain that removes excess glucose by converting it to sorbitol. Sorbitol accumulation can lead to the development of cataracts in the lens and neuropathy in peripheral nerves. Sorbinil has been shown to inhibit aldose reductase in human brain [4] and placenta [5] and calf [6] and rat lens. [5] Sorbinil reduced sorbitol accumulation in rat lens and sciatic nerve of diabetic rats orally administered 0.25 mg/kg sorbinil. [5]
References
- ^ Sorbinil at Sigma-Aldrich
- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary names (Rec. INN): List 25" (PDF). World Health Organization. 1985. Retrieved 11 November 2016.
- ^ MacCari, R; Del Corso, A; Giglio, M; Moschini, R; Mura, U; Ottanà, R (2011). "In vitro evaluation of 5-arylidene-2-thioxo-4-thiazolidinones active as aldose reductase inhibitors". Bioorganic & Medicinal Chemistry Letters. 21 (1): 200–3. doi: 10.1016/j.bmcl.2010.11.041. PMID 21129963.
- ^ O'Brien MM; et al. (1982). "Inhibition of human brain aldose reductase and hexonate dehydrogenase by alrestatin and sorbinil". J Neurochem. 39 (3): 810–4. doi: 10.1111/j.1471-4159.1982.tb07964.x. PMID 6808090. S2CID 22368580.
- ^ a b c Kinoshita JH; et al. (1979). "Aldose reductase in diabetic complications of the eye". Metabolism. 28 (suppl 1): 462–9. doi: 10.1016/0026-0495(79)90057-x. PMID 45423.
- ^ Peterson MJ; et al. (1979). "CP45,634: A novel aldose reductase inhibitor that inhibits polyol pathway activity in diabetic and galactosemic rats". Metabolism. 28 (Suppl 1): 456–61. doi: 10.1016/0026-0495(79)90056-8. PMID 122297.