| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.031.666 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Rh2O3 | |
| Molar mass | 253.8092 g/mol |
| Appearance | dark grey odorless powder |
| Density | 8.20 g/cm3 |
| Melting point | 1,100 °C (2,010 °F; 1,370 K) (decomposes) |
| insoluble | |
| Solubility | insoluble in aqua regia |
| +104.0·10−6 cm3/mol | |
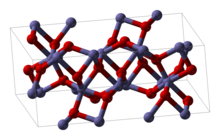
| Structure [1] | |
| hexagonal ( corundum) | |
| R3c | |
a = 512.7 pm (hexagonal setting), c = 1385.3 pm (hexagonal setting)
| |
| Hazards | |
| GHS labelling: [2] | |


| |
| Danger | |
| H302+H332, H315, H319, H335 | |
| P280, P301+P330+P331, P302+P352, P304+P340, P312, P332+P313, P337+P313 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Rhodium(III) oxide (or Rhodium sesquioxide) is the inorganic compound with the formula Rh2 O3. It is a gray solid that is insoluble in ordinary solvents.
Structure
Rh2O3 has been found in two major forms. The hexagonal form adopts the corundum structure. It transforms into an orthorhombic structure when heated above 750 °C. [1]
Production
Rhodium oxide can be produced via several routes:
- Treating RhCl3 with oxygen at high temperatures. [3]
- Rh metal powder is fused with potassium hydrogen sulfate. Adding sodium hydroxide results in hydrated rhodium oxide, which upon heating converts to Rh2O3. [4]
- Rhodium oxide thin films can be produced by exposing Rh layer to oxygen plasma. [5]
- Nanoparticles can be produced by the hydrothermal synthesis. [6]
Physical properties
Rhodium oxide films behave as a fast two-color electrochromic system: Reversible yellow ↔ dark green or yellow ↔ brown-purple color changes are obtained in KOH solutions by applying voltage ~1 V. [7]
Rhodium oxide films are transparent and conductive, like indium tin oxide (ITO) - the common transparent electrode, but Rh2O3 has 0.2 eV lower work function than ITO. Consequently, deposition of rhodium oxide on ITO improves the carrier injection from ITO thereby improving the electrical properties of organic light-emitting diodes. [5]
Catalytic properties
Rhodium oxides are catalysts for hydroformylation of alkenes, [8] N2O production from NO, [9] and the hydrogenation of CO. [10]
See also
References
- ^ a b Coey, J. M. D. (1970-11-01). "The crystal structure of Rh2O3". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry. 26 (11). International Union of Crystallography (IUCr): 1876–1877. doi: 10.1107/s0567740870005022. ISSN 0567-7408.
- ^ GHS: Alfa Aesar 011814 SDS (Feb 2021)
- ^ H. L. Grube (1963). "The Platinum Metals". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. NY: Academic Press. p. 1588.
- ^ Wold, Aaron; Arnott, Ronald J.; Croft, William J. (1963). "The Reaction of Rare Earth Oxides with a High Temperature Form of Rhodium(III) Oxide". Inorganic Chemistry. 2 (5). American Chemical Society (ACS): 972–974. doi: 10.1021/ic50009a023. ISSN 0020-1669.
- ^ a b Kim, Soo Young; Baik, Jeong Min; Yu, Hak Ki; Kim, Kwang Young; Tak, Yoon-Heung; Lee, Jong-Lam (2005-08-15). "Rhodium-oxide-coated indium tin oxide for enhancement of hole injection in organic light emitting diodes". Applied Physics Letters. 87 (7). AIP Publishing: 072105. Bibcode: 2005ApPhL..87g2105K. doi: 10.1063/1.2012534. ISSN 0003-6951.
- ^ Mulukutla, Ravichandra S.; Asakura, Kiyotaka; Kogure, Toshihiro; Namba, Seitaro; Iwasawa, Yasuhiro (1999). "Synthesis and characterization of rhodium oxide nanoparticles in mesoporous MCM-41". Physical Chemistry Chemical Physics. 1 (8). Royal Society of Chemistry (RSC): 2027–2032. Bibcode: 1999PCCP....1.2027M. doi: 10.1039/a900588i. ISSN 1463-9076.
- ^ Gottesfeld, S. (1980). "The Anodic Rhodium Oxide Film: A Two-Color Electrochromic System". Journal of the Electrochemical Society. 127 (2). The Electrochemical Society: 272. doi: 10.1149/1.2129654. ISSN 0013-4651.
- ^ Pino, P.; Botteghi, C. (1977). "Aldehydes from olefins: cyclohexanecarboxaldehyde". Organic Syntheses. 57: 11. doi: 10.15227/orgsyn.057.0011.
- ^ Mulukutla, Ravichandra S; Shido, Takafumi; Asakura, Kiyotaka; Kogure, Toshihiro; Iwasawa, Yasuhiro (2002). "Characterization of rhodium oxide nanoparticles in MCM-41 and their catalytic performances for NO–CO reactions in excess O2". Applied Catalysis A: General. 228 (1–2). Elsevier BV: 305–314. doi: 10.1016/s0926-860x(01)00992-9. ISSN 0926-860X.
- ^ Watson, P; Somorjai, G. A. (1981). "The hydrogenation of carbon monoxide over rhodium oxide surfaces". Journal of Catalysis. 72 (2). Elsevier BV: 347–363. doi: 10.1016/0021-9517(81)90018-x. ISSN 0021-9517.

| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.031.666 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Rh2O3 | |
| Molar mass | 253.8092 g/mol |
| Appearance | dark grey odorless powder |
| Density | 8.20 g/cm3 |
| Melting point | 1,100 °C (2,010 °F; 1,370 K) (decomposes) |
| insoluble | |
| Solubility | insoluble in aqua regia |
| +104.0·10−6 cm3/mol | |
| Structure [1] | |
| hexagonal ( corundum) | |
| R3c | |
a = 512.7 pm (hexagonal setting), c = 1385.3 pm (hexagonal setting)
| |
| Hazards | |
| GHS labelling: [2] | |


| |
| Danger | |
| H302+H332, H315, H319, H335 | |
| P280, P301+P330+P331, P302+P352, P304+P340, P312, P332+P313, P337+P313 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Rhodium(III) oxide (or Rhodium sesquioxide) is the inorganic compound with the formula Rh2 O3. It is a gray solid that is insoluble in ordinary solvents.
Structure
Rh2O3 has been found in two major forms. The hexagonal form adopts the corundum structure. It transforms into an orthorhombic structure when heated above 750 °C. [1]
Production
Rhodium oxide can be produced via several routes:
- Treating RhCl3 with oxygen at high temperatures. [3]
- Rh metal powder is fused with potassium hydrogen sulfate. Adding sodium hydroxide results in hydrated rhodium oxide, which upon heating converts to Rh2O3. [4]
- Rhodium oxide thin films can be produced by exposing Rh layer to oxygen plasma. [5]
- Nanoparticles can be produced by the hydrothermal synthesis. [6]
Physical properties
Rhodium oxide films behave as a fast two-color electrochromic system: Reversible yellow ↔ dark green or yellow ↔ brown-purple color changes are obtained in KOH solutions by applying voltage ~1 V. [7]
Rhodium oxide films are transparent and conductive, like indium tin oxide (ITO) - the common transparent electrode, but Rh2O3 has 0.2 eV lower work function than ITO. Consequently, deposition of rhodium oxide on ITO improves the carrier injection from ITO thereby improving the electrical properties of organic light-emitting diodes. [5]
Catalytic properties
Rhodium oxides are catalysts for hydroformylation of alkenes, [8] N2O production from NO, [9] and the hydrogenation of CO. [10]
See also
References
- ^ a b Coey, J. M. D. (1970-11-01). "The crystal structure of Rh2O3". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry. 26 (11). International Union of Crystallography (IUCr): 1876–1877. doi: 10.1107/s0567740870005022. ISSN 0567-7408.
- ^ GHS: Alfa Aesar 011814 SDS (Feb 2021)
- ^ H. L. Grube (1963). "The Platinum Metals". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. NY: Academic Press. p. 1588.
- ^ Wold, Aaron; Arnott, Ronald J.; Croft, William J. (1963). "The Reaction of Rare Earth Oxides with a High Temperature Form of Rhodium(III) Oxide". Inorganic Chemistry. 2 (5). American Chemical Society (ACS): 972–974. doi: 10.1021/ic50009a023. ISSN 0020-1669.
- ^ a b Kim, Soo Young; Baik, Jeong Min; Yu, Hak Ki; Kim, Kwang Young; Tak, Yoon-Heung; Lee, Jong-Lam (2005-08-15). "Rhodium-oxide-coated indium tin oxide for enhancement of hole injection in organic light emitting diodes". Applied Physics Letters. 87 (7). AIP Publishing: 072105. Bibcode: 2005ApPhL..87g2105K. doi: 10.1063/1.2012534. ISSN 0003-6951.
- ^ Mulukutla, Ravichandra S.; Asakura, Kiyotaka; Kogure, Toshihiro; Namba, Seitaro; Iwasawa, Yasuhiro (1999). "Synthesis and characterization of rhodium oxide nanoparticles in mesoporous MCM-41". Physical Chemistry Chemical Physics. 1 (8). Royal Society of Chemistry (RSC): 2027–2032. Bibcode: 1999PCCP....1.2027M. doi: 10.1039/a900588i. ISSN 1463-9076.
- ^ Gottesfeld, S. (1980). "The Anodic Rhodium Oxide Film: A Two-Color Electrochromic System". Journal of the Electrochemical Society. 127 (2). The Electrochemical Society: 272. doi: 10.1149/1.2129654. ISSN 0013-4651.
- ^ Pino, P.; Botteghi, C. (1977). "Aldehydes from olefins: cyclohexanecarboxaldehyde". Organic Syntheses. 57: 11. doi: 10.15227/orgsyn.057.0011.
- ^ Mulukutla, Ravichandra S; Shido, Takafumi; Asakura, Kiyotaka; Kogure, Toshihiro; Iwasawa, Yasuhiro (2002). "Characterization of rhodium oxide nanoparticles in MCM-41 and their catalytic performances for NO–CO reactions in excess O2". Applied Catalysis A: General. 228 (1–2). Elsevier BV: 305–314. doi: 10.1016/s0926-860x(01)00992-9. ISSN 0926-860X.
- ^ Watson, P; Somorjai, G. A. (1981). "The hydrogenation of carbon monoxide over rhodium oxide surfaces". Journal of Catalysis. 72 (2). Elsevier BV: 347–363. doi: 10.1016/0021-9517(81)90018-x. ISSN 0021-9517.