
| |

| |

| |
| Names | |
|---|---|
|
IUPAC name
Di-μ-chloro-bis[chloro(pentamethylcyclopentadienyl)rhodium(III)]
| |
| Other names
Dichloro(pentamethylcyclopentadienyl)rhodium(III)
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C20H30Cl4Rh2 | |
| Molar mass | 618.07 g·mol−1 |
| Appearance | red solid |
| dichloromethane, chloroform | |
| Hazards | |
| GHS labelling: | |


| |
| Danger | |
| H302, H312, H315, H319, H332, H334, H335 | |
| P261, P264, P270, P271, P280, P285, P301+P312, P302+P352, P304+P312, P304+P340, P304+P341, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P342+P311, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Pentamethylcyclopentadienyl rhodium dichloride dimer is an organometallic compound with the formula [(C5(CH3)5RhCl2)]2, commonly abbreviated [Cp*RhCl22 This dark red air-stable diamagnetic solid is a reagent in organometallic chemistry. [1]
The compound has idealized C2h symmetry. Each metal centre is pseudo-octahedral.
The compound is prepared by the reaction of rhodium trichloride trihydrate and
pentamethylcyclopentadiene in hot methanol, from which the product precipitates:
[1]
- 2 C5(CH3)5H + 2 RhCl3(H2O)3 → [(C5(CH3)5)RhCl22 + 2 HCl + 6 H2O
It was first prepared by the reaction of hydrated rhodium trichloride with hexamethyl Dewar benzene [2]
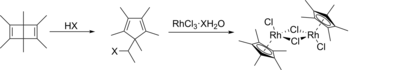
This complex was first prepared from hexamethyl Dewar benzene and RhCl3(H2O)3. [3] [4] [5] The hydrohalic acid necessary for the ring-contraction rearrangement is generated in situ in methanolic solutions of the rhodium salt, and the second step has been carried out separately, confirming this mechanistic description. [6] The reaction occurs with the formation of 1,1-dimethoxyethane, CH3CH(OCH3)2, and hexamethylbenzene is produced by a side reaction. [5] [6]
This rhodium(III) dimer can be reduced with zinc in the presence of CO to produce the rhodium(I) complex [Cp*Rh(CO)2]. [7]
Reductive carbonylation gives [Cp*Rh(CO)2]. [8]
The Rh- μ-Cl bonds are labile and cleave en route to a variety of adducts of the general formula Cp*RhCl2L. Treatment with silver ions in polar coordinating solvents causes precipitation of silver(I) chloride, leaving a solution containing dications of the form [Cp*RhL32+ (L = H2O, MeCN).
The chemistry is similar to that of the analog pentamethylcyclopentadienyl iridium dichloride dimer.
- Kang, Jung W.; Mosley, K.; Maitlis, Peter M. (1968). "Mechanisms of Reactions of Dewar Hexamethylbenzene with Rhodium and Iridium Chlorides". Chem. Commun. (21): 1304–1305. doi: 10.1039/C19680001304.
- Kang, Jung W.; Maitlis, Peter M. (1968). "Conversion of Dewar Hexamethylbenzene to Pentamethylcyclopentadienylrhodium(III) Chloride". J. Am. Chem. Soc. 90 (12): 3259–3261. doi: 10.1021/ja01014a063.
- Criegee, Rudolf; Grüner, H. (1968). "Acid-catalyzed Rearrangements of Hexamethyl-prismane and Hexamethyl-Dewar-benzene". Angew. Chem. Int. Ed. 7 (6): 467–468. doi: 10.1002/anie.196804672.
- Kang, Jung W.; Moseley, K.; Maitlis, Peter M. (1969). "Pentamethylcyclopentadienylrhodium and -iridium halides. I. Synthesis and properties". J. Am. Chem. Soc. 91 (22): 5970–5977. doi: 10.1021/ja01050a008.
- Herrmann, Wolfgang A.; Zybill, Christian (1996). "Bis{(μ-chloro)[chloro(η-pentamethylcyclopentadienyl)rhodium]} — {Rh(μ-Cl)Cl[η-C5(CH3)5]}2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 148–149. ISBN 9783131791610.
- Heck, Richard F. (1974). "Reactions of Dienes Trienes and Tetraenes with Transition Metal Compounds". Organotransition Metal Chemistry: A Mechanistic Approach. Academic Press. pp. 116–117. ISBN 9780323154703.
- ^
a
b White, C.; Yates, A.;
Maitlis, Peter M. (2007). "(η 5 -Pentamethylcyclopentadienyl)Rhodium and -Iridium Compounds". Inorganic Syntheses. Vol. 29. pp. 228–234.
doi:
10.1002/9780470132609.ch53.
ISBN
9780470132609.
{{ cite book}}:|journal=ignored ( help) - ^ Paquette, Leo A.; Krow, Grant R. (1968). "Electrophilic Additions to Hexamethyldewarbenzene". Tetrahedron Lett. 9 (17): 2139–2142. doi: 10.1016/S0040-4039(00)89761-0.
- ^ Paquette, Leo A.; Krow, Grant R. (1968). "Electrophilic Additions to Hexamethyldewarbenzene". Tetrahedron Lett. 9 (17): 2139–2142. doi: 10.1016/S0040-4039(00)89761-0.
- ^ Criegee, Rudolf; Grüner, H. (1968). "Acid-catalyzed Rearrangements of Hexamethyl-prismane and Hexamethyl-Dewar-benzene". Angew. Chem. Int. Ed. 7 (6): 467–468. doi: 10.1002/anie.196804672.
- ^ a b Herrmann, Wolfgang A.; Zybill, Christian (1996). "Bis{(μ-chloro)[chloro(η-pentamethylcyclopentadienyl)rhodium]} — {Rh(μ-Cl)Cl[η-C5(CH3)5]}2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 148–149. ISBN 9783131791610.
- ^ a b Heck, Richard F. (1974). "Reactions of Dienes Trienes and Tetraenes with Transition Metal Compounds". Organotransition Metal Chemistry: A Mechanistic Approach. Academic Press. pp. 116–117. ISBN 9780323154703.
- ^ Herrmann, Wolfgang A.; Zybill, Christian (1996). "Dicarbonyl(η-pentamethylcyclopentadienyl)rhodium — Rh[η-C5(CH3)5](CO)2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 147–148. ISBN 9783131791610.
- ^ Herrmann, Wolfgang A.; Zybill, Christian (1996). "Dicarbonyl(η-pentamethylcyclopentadienyl)rhodium — Rh[η-C5(CH3)5](CO)2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 147–148. ISBN 9783131791610.

| |

| |

| |
| Names | |
|---|---|
|
IUPAC name
Di-μ-chloro-bis[chloro(pentamethylcyclopentadienyl)rhodium(III)]
| |
| Other names
Dichloro(pentamethylcyclopentadienyl)rhodium(III)
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C20H30Cl4Rh2 | |
| Molar mass | 618.07 g·mol−1 |
| Appearance | red solid |
| dichloromethane, chloroform | |
| Hazards | |
| GHS labelling: | |


| |
| Danger | |
| H302, H312, H315, H319, H332, H334, H335 | |
| P261, P264, P270, P271, P280, P285, P301+P312, P302+P352, P304+P312, P304+P340, P304+P341, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P342+P311, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Pentamethylcyclopentadienyl rhodium dichloride dimer is an organometallic compound with the formula [(C5(CH3)5RhCl2)]2, commonly abbreviated [Cp*RhCl22 This dark red air-stable diamagnetic solid is a reagent in organometallic chemistry. [1]
The compound has idealized C2h symmetry. Each metal centre is pseudo-octahedral.
The compound is prepared by the reaction of rhodium trichloride trihydrate and
pentamethylcyclopentadiene in hot methanol, from which the product precipitates:
[1]
- 2 C5(CH3)5H + 2 RhCl3(H2O)3 → [(C5(CH3)5)RhCl22 + 2 HCl + 6 H2O
It was first prepared by the reaction of hydrated rhodium trichloride with hexamethyl Dewar benzene [2]

This complex was first prepared from hexamethyl Dewar benzene and RhCl3(H2O)3. [3] [4] [5] The hydrohalic acid necessary for the ring-contraction rearrangement is generated in situ in methanolic solutions of the rhodium salt, and the second step has been carried out separately, confirming this mechanistic description. [6] The reaction occurs with the formation of 1,1-dimethoxyethane, CH3CH(OCH3)2, and hexamethylbenzene is produced by a side reaction. [5] [6]
This rhodium(III) dimer can be reduced with zinc in the presence of CO to produce the rhodium(I) complex [Cp*Rh(CO)2]. [7]
Reductive carbonylation gives [Cp*Rh(CO)2]. [8]
The Rh- μ-Cl bonds are labile and cleave en route to a variety of adducts of the general formula Cp*RhCl2L. Treatment with silver ions in polar coordinating solvents causes precipitation of silver(I) chloride, leaving a solution containing dications of the form [Cp*RhL32+ (L = H2O, MeCN).
The chemistry is similar to that of the analog pentamethylcyclopentadienyl iridium dichloride dimer.
- Kang, Jung W.; Mosley, K.; Maitlis, Peter M. (1968). "Mechanisms of Reactions of Dewar Hexamethylbenzene with Rhodium and Iridium Chlorides". Chem. Commun. (21): 1304–1305. doi: 10.1039/C19680001304.
- Kang, Jung W.; Maitlis, Peter M. (1968). "Conversion of Dewar Hexamethylbenzene to Pentamethylcyclopentadienylrhodium(III) Chloride". J. Am. Chem. Soc. 90 (12): 3259–3261. doi: 10.1021/ja01014a063.
- Criegee, Rudolf; Grüner, H. (1968). "Acid-catalyzed Rearrangements of Hexamethyl-prismane and Hexamethyl-Dewar-benzene". Angew. Chem. Int. Ed. 7 (6): 467–468. doi: 10.1002/anie.196804672.
- Kang, Jung W.; Moseley, K.; Maitlis, Peter M. (1969). "Pentamethylcyclopentadienylrhodium and -iridium halides. I. Synthesis and properties". J. Am. Chem. Soc. 91 (22): 5970–5977. doi: 10.1021/ja01050a008.
- Herrmann, Wolfgang A.; Zybill, Christian (1996). "Bis{(μ-chloro)[chloro(η-pentamethylcyclopentadienyl)rhodium]} — {Rh(μ-Cl)Cl[η-C5(CH3)5]}2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 148–149. ISBN 9783131791610.
- Heck, Richard F. (1974). "Reactions of Dienes Trienes and Tetraenes with Transition Metal Compounds". Organotransition Metal Chemistry: A Mechanistic Approach. Academic Press. pp. 116–117. ISBN 9780323154703.
- ^
a
b White, C.; Yates, A.;
Maitlis, Peter M. (2007). "(η 5 -Pentamethylcyclopentadienyl)Rhodium and -Iridium Compounds". Inorganic Syntheses. Vol. 29. pp. 228–234.
doi:
10.1002/9780470132609.ch53.
ISBN
9780470132609.
{{ cite book}}:|journal=ignored ( help) - ^ Paquette, Leo A.; Krow, Grant R. (1968). "Electrophilic Additions to Hexamethyldewarbenzene". Tetrahedron Lett. 9 (17): 2139–2142. doi: 10.1016/S0040-4039(00)89761-0.
- ^ Paquette, Leo A.; Krow, Grant R. (1968). "Electrophilic Additions to Hexamethyldewarbenzene". Tetrahedron Lett. 9 (17): 2139–2142. doi: 10.1016/S0040-4039(00)89761-0.
- ^ Criegee, Rudolf; Grüner, H. (1968). "Acid-catalyzed Rearrangements of Hexamethyl-prismane and Hexamethyl-Dewar-benzene". Angew. Chem. Int. Ed. 7 (6): 467–468. doi: 10.1002/anie.196804672.
- ^ a b Herrmann, Wolfgang A.; Zybill, Christian (1996). "Bis{(μ-chloro)[chloro(η-pentamethylcyclopentadienyl)rhodium]} — {Rh(μ-Cl)Cl[η-C5(CH3)5]}2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 148–149. ISBN 9783131791610.
- ^ a b Heck, Richard F. (1974). "Reactions of Dienes Trienes and Tetraenes with Transition Metal Compounds". Organotransition Metal Chemistry: A Mechanistic Approach. Academic Press. pp. 116–117. ISBN 9780323154703.
- ^ Herrmann, Wolfgang A.; Zybill, Christian (1996). "Dicarbonyl(η-pentamethylcyclopentadienyl)rhodium — Rh[η-C5(CH3)5](CO)2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 147–148. ISBN 9783131791610.
- ^ Herrmann, Wolfgang A.; Zybill, Christian (1996). "Dicarbonyl(η-pentamethylcyclopentadienyl)rhodium — Rh[η-C5(CH3)5](CO)2". In Herrmann, Wolfgang A.; Salzer, Albrecht (eds.). Synthetic Methods of Organometallic and Inorganic Chemistry – Volume 1: Literature, Laboratory Techniques, and Common Starting Materials. Georg Thieme Verlag. pp. 147–148. ISBN 9783131791610.