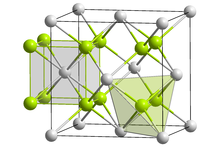
 Crystal structure of potassium polonide
__ K+ __ Po2- | |
| Names | |
|---|---|
|
Preferred IUPAC name
Potassium polonide | |
| Identifiers | |
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| K2Po | |
| Molar mass | 287.18 g/mol |
| Appearance | greyish [1] |
| Related compounds | |
Other
anions
|
Potassium oxide Potassium sulfide Potassium selenide Potassium telluride |
Other
cations
|
Lithium polonide Sodium polonide Rubidium polonide Caesium polonide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium polonide is a chemical compound with the formula K2 Po. It is a polonide, a set of very chemically stable compounds of polonium. [2] [3]
Characteristics
Potassium polonide is thermally more unstable and has stronger electron affinity than potassium telluride (K2Te). [2] [3]
Production
Potassium polonide may be produced from a redox reaction between polonium hydride and potassium metal: [2] [3]
- H2Po + 2 K → K2Po + H2
It may also be produced by heating potassium and polonium together at 300–400 °C. [1] At higher temperature, this reaction may reverse.
Crystal structure
Like sodium polonide, potassium polonide has the antifluorite structure. [2] [3]
References
- ^ a b Bagnall, K. W. (1962). "The Chemistry of Polonium". Advances in Inorganic Chemistry and Radiochemistry. New York: Academic Press. pp. 197–230. ISBN 9780120236046. Retrieved June 17, 2012.
- ^ a b c d Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 899. ISBN 978-0-08-022057-4.
- ^ a b c d Moyer, Harvey V. (1956), "Chemical Properties of Polonium", in Moyer, Harvey V. (ed.), Polonium, Oak Ridge, Tenn.: United States Atomic Energy Commission, pp. 33–96, doi: 10.2172/4367751, TID-5221
 Crystal structure of potassium polonide
__ K+ __ Po2- | |
| Names | |
|---|---|
|
Preferred IUPAC name
Potassium polonide | |
| Identifiers | |
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| K2Po | |
| Molar mass | 287.18 g/mol |
| Appearance | greyish [1] |
| Related compounds | |
Other
anions
|
Potassium oxide Potassium sulfide Potassium selenide Potassium telluride |
Other
cations
|
Lithium polonide Sodium polonide Rubidium polonide Caesium polonide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium polonide is a chemical compound with the formula K2 Po. It is a polonide, a set of very chemically stable compounds of polonium. [2] [3]
Characteristics
Potassium polonide is thermally more unstable and has stronger electron affinity than potassium telluride (K2Te). [2] [3]
Production
Potassium polonide may be produced from a redox reaction between polonium hydride and potassium metal: [2] [3]
- H2Po + 2 K → K2Po + H2
It may also be produced by heating potassium and polonium together at 300–400 °C. [1] At higher temperature, this reaction may reverse.
Crystal structure
Like sodium polonide, potassium polonide has the antifluorite structure. [2] [3]
References
- ^ a b Bagnall, K. W. (1962). "The Chemistry of Polonium". Advances in Inorganic Chemistry and Radiochemistry. New York: Academic Press. pp. 197–230. ISBN 9780120236046. Retrieved June 17, 2012.
- ^ a b c d Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 899. ISBN 978-0-08-022057-4.
- ^ a b c d Moyer, Harvey V. (1956), "Chemical Properties of Polonium", in Moyer, Harvey V. (ed.), Polonium, Oak Ridge, Tenn.: United States Atomic Energy Commission, pp. 33–96, doi: 10.2172/4367751, TID-5221