
| |
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| Lu(IO3)3 | |
| Molar mass | 699.68 |
| 2.04×10−3 mol·L−1 [1] | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
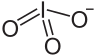
Lutetium iodate is an inorganic compound with the chemical formula Lu(IO3)3. It exists in two anhydrous forms, α-form and β-form, as well as dihydrate and tetrahydrate. It can be produced by the reaction of lutetium nitrate and iodic acid [2] or potassium iodate. [1] It decomposes when heated to generate lutetium oxide. [3]
- ^
a
b Hiroshi Miyamoto, Hiroko Shimura, Kayoko Sasaki (Jul 1985).
"Solubilities of rare earth lodates in aqueous and aqueous alcoholic solvent mixtures". Journal of Solution Chemistry. 14 (7): 485–497.
doi:
10.1007/BF00646980.
ISSN
0095-9782. Retrieved 2020-05-29.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ K. Nassau, J.W. Shiever, B.E. Prescott, A.S. Cooper (Dec 1974).
"Transition metal iodates. V. Preparation and characterization of the smaller lanthanide iodates". Journal of Solid State Chemistry. 11 (4): 314–318.
Bibcode:
1974JSSCh..11..314N.
doi:
10.1016/S0022-4596(74)80036-8.
Archived from the original on 2018-06-28. Retrieved 2020-05-29.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ K. Nassau, J.W. Shiever, B.E. Prescott (Jun 1975).
"Transition metal iodates. VI. Preparation and characterization of the larger lanthanide iodates". Journal of Solid State Chemistry. 14 (2): 122–132.
Bibcode:
1975JSSCh..14..122N.
doi:
10.1016/0022-4596(75)90002-X.
Archived from the original on 2018-07-01. Retrieved 2020-05-29.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)


| |
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| Lu(IO3)3 | |
| Molar mass | 699.68 |
| 2.04×10−3 mol·L−1 [1] | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Lutetium iodate is an inorganic compound with the chemical formula Lu(IO3)3. It exists in two anhydrous forms, α-form and β-form, as well as dihydrate and tetrahydrate. It can be produced by the reaction of lutetium nitrate and iodic acid [2] or potassium iodate. [1] It decomposes when heated to generate lutetium oxide. [3]
- ^
a
b Hiroshi Miyamoto, Hiroko Shimura, Kayoko Sasaki (Jul 1985).
"Solubilities of rare earth lodates in aqueous and aqueous alcoholic solvent mixtures". Journal of Solution Chemistry. 14 (7): 485–497.
doi:
10.1007/BF00646980.
ISSN
0095-9782. Retrieved 2020-05-29.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ K. Nassau, J.W. Shiever, B.E. Prescott, A.S. Cooper (Dec 1974).
"Transition metal iodates. V. Preparation and characterization of the smaller lanthanide iodates". Journal of Solid State Chemistry. 11 (4): 314–318.
Bibcode:
1974JSSCh..11..314N.
doi:
10.1016/S0022-4596(74)80036-8.
Archived from the original on 2018-06-28. Retrieved 2020-05-29.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ K. Nassau, J.W. Shiever, B.E. Prescott (Jun 1975).
"Transition metal iodates. VI. Preparation and characterization of the larger lanthanide iodates". Journal of Solid State Chemistry. 14 (2): 122–132.
Bibcode:
1975JSSCh..14..122N.
doi:
10.1016/0022-4596(75)90002-X.
Archived from the original on 2018-07-01. Retrieved 2020-05-29.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)