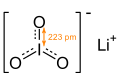
| |

| |
| Names | |
|---|---|
|
IUPAC name
Lithium iodate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.954 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
| UN number | 1479 |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| LiIO3 | |
| Appearance | White hygroscopic crystals |
| Odor | Odorless |
| Density | 4.487 g/cm3 [1] |
| Melting point | 420–450 °C (788–842 °F; 693–723 K) [1] [3] [5] |
| Anhydrous: 89.4 g/100 mL (10 °C) 82.7 g/100 mL (25 °C) 78.4 g/100 mL (40.1 °C) 73 g/100 mL (75.6 °C) [1] Hemihydrate: 80.2 g/100 mL (18 °C) [2] | |
| Solubility | Insoluble in EtOH [3] |
| −47.0·10−6 cm3/mol | |
| Thermal conductivity | 1.27 W/m·K (a-axis) 0.65 W/m·K (c-axis) [1] |
Refractive index (nD)
|
1.8875 (20 °C) 1.6 (RT) n He–Ne: 1.8815 (20 °C) [1] 1.5928 (RT) [4] |
| Structure | |
| Hexagonal, [3] hP10 [6] | |
| P6322, No. 182 [6] | |
| 622 [6] | |
a = 5.46(9) Å, c = 5.15(5) Å
[6] α = 90°, β = 90°, γ = 120°
| |
| Hazards | |
| GHS labelling: | |


 [7]
[7]
| |
| Danger | |
| H272, H315, H319, H335, H360 [7] | |
| P201, P220, P261, P305+P351+P338, P308+P313 [7] | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium iodate (LiIO3) is a negative uniaxial crystal [1] for nonlinear, acousto-optical and piezoelectric applications. It has been utilized for 347 nm ruby lasers. [9] [10]
Properties
Mohs hardness of lithium iodate is 3.5–4. Its linear thermal expansion coefficient at 298 K (25 °C; 77 °F) is 2.8·10−5/°C (a-axis) and 4.8·10−5/°C (c-axis). [1] Its transition to β-form begin at 50 °C (122 °F) and it is irreversible. [5]
References
- ^ a b c d e f g "Rarely Used and Archive Crystals". Nonlinear Optical Crystals: A Complete Survey. 2005. pp. 364–368. doi: 10.1007/0-387-27151-1_8. ISBN 978-0-387-27151-4. Archived from the original on 2014-08-08. Retrieved 2014-08-08.
- ^ Seidell, Atherton; Linke, William F. (1919). Solubilities of Inorganic and Organic Compounds (2nd ed.). New York City: D. Van Nostrand Company. p. 374.
- ^ a b c Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ Polyanskiy, Mikhail. "Refractive index of LiIO3 (Lithium iodate) - Herbst-o". refractiveindex.info. Retrieved 2014-08-08.
- ^ a b Teyssier, Jeremie; Dantec, Ronan Le; Galez, Christine; Mugnier, Yannick; Bouillot, Jacques; Plenet, Jean-Claude (2003-11-20). Andrews, David L; Gaburro, Zeno; Cartwright, Alexander N; Lee, Charles Y. C (eds.). "LiIO3 nanocrystals in SiO2 xerogels, a new material for non-linear optics". Proceedings of SPIE. Nanocrystals, and Organic and Hybrid Nanomaterials. 5222 (26): 26. Bibcode: 2003SPIE.5222...26T. CiteSeerX 10.1.1.605.1743. doi: 10.1117/12.507309. S2CID 136547473.
- ^ a b c d Zachariasen, W.H.; Olof, F.A. BartaLars (1931-06-15). "Crystal Structure of Lithium Iodate". Physical Review Letters. 37 (12): 1626–1630. Bibcode: 1931PhRv...37.1626Z. doi: 10.1103/PhysRev.37.1626.
- ^ a b c Sigma-Aldrich Co., Lithium iodate. Retrieved on 2014-08-08.
- ^ "SDS of Lithium iodate anhydrous" (PDF). pfaltzandbauer.com. Connecticut, USA: Pfaltz & Bauer, Inc. Archived from the original (PDF) on 2014-08-10. Retrieved 2014-08-08.
- ^ Risk, W. P.; Gosnell, T. R.; Nurmikko, A. V. (9 January 2003). Compact Blue-Green Lasers. Cambridge University Press. p. 123. ISBN 978-0-521-52103-1. Retrieved 13 December 2012.
- ^ Nikogosyan, David N. (4 January 2005). Nonlinear Optical Crystals: A Complete Survey. Springer. p. 371. ISBN 978-0-387-22022-2. Retrieved 13 December 2012.
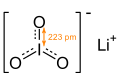
| |

| |
| Names | |
|---|---|
|
IUPAC name
Lithium iodate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.954 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
| UN number | 1479 |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| LiIO3 | |
| Appearance | White hygroscopic crystals |
| Odor | Odorless |
| Density | 4.487 g/cm3 [1] |
| Melting point | 420–450 °C (788–842 °F; 693–723 K) [1] [3] [5] |
| Anhydrous: 89.4 g/100 mL (10 °C) 82.7 g/100 mL (25 °C) 78.4 g/100 mL (40.1 °C) 73 g/100 mL (75.6 °C) [1] Hemihydrate: 80.2 g/100 mL (18 °C) [2] | |
| Solubility | Insoluble in EtOH [3] |
| −47.0·10−6 cm3/mol | |
| Thermal conductivity | 1.27 W/m·K (a-axis) 0.65 W/m·K (c-axis) [1] |
Refractive index (nD)
|
1.8875 (20 °C) 1.6 (RT) n He–Ne: 1.8815 (20 °C) [1] 1.5928 (RT) [4] |
| Structure | |
| Hexagonal, [3] hP10 [6] | |
| P6322, No. 182 [6] | |
| 622 [6] | |
a = 5.46(9) Å, c = 5.15(5) Å
[6] α = 90°, β = 90°, γ = 120°
| |
| Hazards | |
| GHS labelling: | |


 [7]
[7]
| |
| Danger | |
| H272, H315, H319, H335, H360 [7] | |
| P201, P220, P261, P305+P351+P338, P308+P313 [7] | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium iodate (LiIO3) is a negative uniaxial crystal [1] for nonlinear, acousto-optical and piezoelectric applications. It has been utilized for 347 nm ruby lasers. [9] [10]
Properties
Mohs hardness of lithium iodate is 3.5–4. Its linear thermal expansion coefficient at 298 K (25 °C; 77 °F) is 2.8·10−5/°C (a-axis) and 4.8·10−5/°C (c-axis). [1] Its transition to β-form begin at 50 °C (122 °F) and it is irreversible. [5]
References
- ^ a b c d e f g "Rarely Used and Archive Crystals". Nonlinear Optical Crystals: A Complete Survey. 2005. pp. 364–368. doi: 10.1007/0-387-27151-1_8. ISBN 978-0-387-27151-4. Archived from the original on 2014-08-08. Retrieved 2014-08-08.
- ^ Seidell, Atherton; Linke, William F. (1919). Solubilities of Inorganic and Organic Compounds (2nd ed.). New York City: D. Van Nostrand Company. p. 374.
- ^ a b c Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ Polyanskiy, Mikhail. "Refractive index of LiIO3 (Lithium iodate) - Herbst-o". refractiveindex.info. Retrieved 2014-08-08.
- ^ a b Teyssier, Jeremie; Dantec, Ronan Le; Galez, Christine; Mugnier, Yannick; Bouillot, Jacques; Plenet, Jean-Claude (2003-11-20). Andrews, David L; Gaburro, Zeno; Cartwright, Alexander N; Lee, Charles Y. C (eds.). "LiIO3 nanocrystals in SiO2 xerogels, a new material for non-linear optics". Proceedings of SPIE. Nanocrystals, and Organic and Hybrid Nanomaterials. 5222 (26): 26. Bibcode: 2003SPIE.5222...26T. CiteSeerX 10.1.1.605.1743. doi: 10.1117/12.507309. S2CID 136547473.
- ^ a b c d Zachariasen, W.H.; Olof, F.A. BartaLars (1931-06-15). "Crystal Structure of Lithium Iodate". Physical Review Letters. 37 (12): 1626–1630. Bibcode: 1931PhRv...37.1626Z. doi: 10.1103/PhysRev.37.1626.
- ^ a b c Sigma-Aldrich Co., Lithium iodate. Retrieved on 2014-08-08.
- ^ "SDS of Lithium iodate anhydrous" (PDF). pfaltzandbauer.com. Connecticut, USA: Pfaltz & Bauer, Inc. Archived from the original (PDF) on 2014-08-10. Retrieved 2014-08-08.
- ^ Risk, W. P.; Gosnell, T. R.; Nurmikko, A. V. (9 January 2003). Compact Blue-Green Lasers. Cambridge University Press. p. 123. ISBN 978-0-521-52103-1. Retrieved 13 December 2012.
- ^ Nikogosyan, David N. (4 January 2005). Nonlinear Optical Crystals: A Complete Survey. Springer. p. 371. ISBN 978-0-387-22022-2. Retrieved 13 December 2012.
