 | |
| Clinical data | |
|---|---|
| Trade names | Emrok |
| Other names | (S)-(-)-Nadifloxacin |
|
Routes of administration | Parenteral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
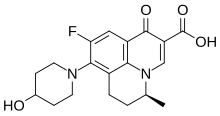
| Formula | C19H21FN2O4 |
| Molar mass | 360.385 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Levonadifloxacin (trade name Emrok) is an antibiotic drug of the fluoroquinolone class. [1] [2] Chemically, it is the (S)- enantiomer of the racemic drug nadifloxacin.
It is approved in India for the treatment of skin and soft tissue infections of Gram-positive bacteria. [3] It is also being studied for potential use against resistant strains of bacteria including Streptococcus pneumoniae, Streptococcus pyogenes, Haemophilus influenzae, and Moraxella catarrhalis. [4]
Levonadifloxacin has poor oral bioavailability. A prodrug of levonadifloxacin with high oral bioavailability, alalevonadifloxacin, has been developed to mitigate this problem. [5]
- ^ Patel A, Sangle GV, Trivedi J, Shengule SA, Thorve D, Patil M, et al. (April 2020). "Levonadifloxacin, a Novel Benzoquinolizine Fluoroquinolone, Modulates Lipopolysaccharide-Induced Inflammatory Responses in Human Whole-Blood Assay and Murine Acute Lung Injury Model". Antimicrobial Agents and Chemotherapy. 64 (5). doi: 10.1128/aac.00084-20. PMC 7179645. PMID 32152077.
- ^ Bakthavatchalam YD, Shankar A, Muniyasamy R, Peter JV, Marcus Z, Triplicane Dwarakanathan H, et al. (August 2020). "Levonadifloxacin, a recently approved benzoquinolizine fluoroquinolone, exhibits potent in vitro activity against contemporary Staphylococcus aureus isolates and Bengal Bay clone isolates collected from a large Indian tertiary care hospital". The Journal of Antimicrobial Chemotherapy. 75 (8): 2156–2159. doi: 10.1093/jac/dkaa142. PMID 32361727.
- ^ "Levonadifloxacin". AdisInsight. Springer Nature Switzerland AG.
- ^ Bhagwat SS, Nandanwar M, Kansagara A, Patel A, Takalkar S, Chavan R, et al. (2019). "Levonadifloxacin, a Novel Broad-Spectrum Anti-MRSA Benzoquinolizine Quinolone Agent: Review of Current Evidence". Drug Design, Development and Therapy. 13: 4351–4365. doi: 10.2147/DDDT.S229882. PMC 6935279. PMID 31920285.
- ^ Bhawsar S, Kale R, Deshpande P, Yeole R, Bhagwat S, Patel M (December 2021). "Design and synthesis of an oral prodrug alalevonadifloxacin for the treatment of MRSA infection". Bioorganic & Medicinal Chemistry Letters. 54: 128432. doi: 10.1016/j.bmcl.2021.128432. PMID 34757217.
 | |
| Clinical data | |
|---|---|
| Trade names | Emrok |
| Other names | (S)-(-)-Nadifloxacin |
|
Routes of administration | Parenteral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C19H21FN2O4 |
| Molar mass | 360.385 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Levonadifloxacin (trade name Emrok) is an antibiotic drug of the fluoroquinolone class. [1] [2] Chemically, it is the (S)- enantiomer of the racemic drug nadifloxacin.
It is approved in India for the treatment of skin and soft tissue infections of Gram-positive bacteria. [3] It is also being studied for potential use against resistant strains of bacteria including Streptococcus pneumoniae, Streptococcus pyogenes, Haemophilus influenzae, and Moraxella catarrhalis. [4]
Levonadifloxacin has poor oral bioavailability. A prodrug of levonadifloxacin with high oral bioavailability, alalevonadifloxacin, has been developed to mitigate this problem. [5]
- ^ Patel A, Sangle GV, Trivedi J, Shengule SA, Thorve D, Patil M, et al. (April 2020). "Levonadifloxacin, a Novel Benzoquinolizine Fluoroquinolone, Modulates Lipopolysaccharide-Induced Inflammatory Responses in Human Whole-Blood Assay and Murine Acute Lung Injury Model". Antimicrobial Agents and Chemotherapy. 64 (5). doi: 10.1128/aac.00084-20. PMC 7179645. PMID 32152077.
- ^ Bakthavatchalam YD, Shankar A, Muniyasamy R, Peter JV, Marcus Z, Triplicane Dwarakanathan H, et al. (August 2020). "Levonadifloxacin, a recently approved benzoquinolizine fluoroquinolone, exhibits potent in vitro activity against contemporary Staphylococcus aureus isolates and Bengal Bay clone isolates collected from a large Indian tertiary care hospital". The Journal of Antimicrobial Chemotherapy. 75 (8): 2156–2159. doi: 10.1093/jac/dkaa142. PMID 32361727.
- ^ "Levonadifloxacin". AdisInsight. Springer Nature Switzerland AG.
- ^ Bhagwat SS, Nandanwar M, Kansagara A, Patel A, Takalkar S, Chavan R, et al. (2019). "Levonadifloxacin, a Novel Broad-Spectrum Anti-MRSA Benzoquinolizine Quinolone Agent: Review of Current Evidence". Drug Design, Development and Therapy. 13: 4351–4365. doi: 10.2147/DDDT.S229882. PMC 6935279. PMID 31920285.
- ^ Bhawsar S, Kale R, Deshpande P, Yeole R, Bhagwat S, Patel M (December 2021). "Design and synthesis of an oral prodrug alalevonadifloxacin for the treatment of MRSA infection". Bioorganic & Medicinal Chemistry Letters. 54: 128432. doi: 10.1016/j.bmcl.2021.128432. PMID 34757217.