
| |
| |
| Names | |
|---|---|
|
IUPAC name
(2S)-3′,5,7-Trihydroxy-4′-methoxyflavan-4-one
| |
|
Systematic IUPAC name
(2S)-5,7-Dihydroxy-2-(3-hydroxy-4-methoxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.007.538 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C16H14O6 | |
| Molar mass | 302.282 g·mol−1 |
| Melting point | 226–228 °C (439–442 °F; 499–501 K) |
| Solubility in other solvents | Sol. EtOH, alkalis |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
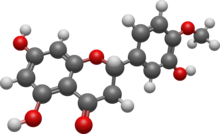
Hesperetin is the 4'-methoxy derivative of eriodictyol, a flavanone. [1] The 7-O-glycoside of hesperetin, hesperidin, is a naturally occurring flavanone-glycoside, the main flavonoid in grapefruits, lemons, and sweet oranges. [1] [2]
Glycosides
Various glycosides of hesperetin are known, including hesperidin (hesperetin-7-O-rutinoside), a water-insoluble flavonoid glycoside with low water solubility, [3] Hesperidin is found in citrus fruits and upon ingestion it releases its aglycone, hesperetin. Neohesperidin is the 7-O- neohesperidoside of hesperetin.
Metabolism
Hesperidin 6-O-α-L-rhamnosyl-β-D-glucosidase is an enzyme that uses hesperidin and H2O to produce hesperetin and rutinose. [1] Upon digestion in the gastrointestinal tract, hesperetin – as for all flavonoids – is rapidly metabolized in intestinal and liver cells, releasing smaller metabolites into the blood and urine for excretion. [1] The biological effects of such metabolites in vivo are unknown.
Laboratory research
In vitro, hesperetin may affect the slow inactivation phase of inward sodium current channels, and therefore could be used as a template to develop drugs against cardiac arrhythmias. [4] Hesperetin also inhibits TRPM3 channels in vitro. [5]
References
- ^ a b c d "Flavonoids". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. 2024. Retrieved 10 May 2024.
- ^ "Hesperetin". PubChem, US National Library of Medicine. 4 May 2024.
- ^ Majumdar S.; Srirangam, R. (2009). "Solubility, stability, physicochemical characteristics and in vitro ocular tissue permeability of hesperidin: a natural bioflavonoid". Pharm. Res. 26 (5): 1217–1225. doi: 10.1007/s11095-008-9729-6. PMC 2664388. PMID 18810327.
- ^ Alvarez‐Collazo, Julio; López‐Requena, Alejandro; Galán, Loipa; Talavera, Ariel; Alvarez, Julio L.; Talavera, Karel (27 March 2019). "The citrus flavanone hesperetin preferentially inhibits slow‐inactivating currents of a long QT syndrome type 3 syndrome Na+ channel mutation". British Journal of Pharmacology. 176 (8): 1090–1105. doi: 10.1111/bph.14577. PMC 6451064. PMID 30650182.
- ^ Straub, Isabelle; Krügel, Ute; Mohr, Florian; Teichert, Jens; Rizun, Oleksandr; Konrad, Maik; Oberwinkler, Johannes; Schaefer, Michael (November 2013). "Flavanones that selectively inhibit TRPM3 attenuate thermal nociception in vivo". Molecular Pharmacology. 84 (5): 736–750. doi: 10.1124/mol.113.086843. ISSN 1521-0111. PMID 24006495.
External links
-
 Media related to
Hesperetin at Wikimedia Commons
Media related to
Hesperetin at Wikimedia Commons

| |

| |
| Names | |
|---|---|
|
IUPAC name
(2S)-3′,5,7-Trihydroxy-4′-methoxyflavan-4-one
| |
|
Systematic IUPAC name
(2S)-5,7-Dihydroxy-2-(3-hydroxy-4-methoxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.007.538 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C16H14O6 | |
| Molar mass | 302.282 g·mol−1 |
| Melting point | 226–228 °C (439–442 °F; 499–501 K) |
| Solubility in other solvents | Sol. EtOH, alkalis |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Hesperetin is the 4'-methoxy derivative of eriodictyol, a flavanone. [1] The 7-O-glycoside of hesperetin, hesperidin, is a naturally occurring flavanone-glycoside, the main flavonoid in grapefruits, lemons, and sweet oranges. [1] [2]
Glycosides
Various glycosides of hesperetin are known, including hesperidin (hesperetin-7-O-rutinoside), a water-insoluble flavonoid glycoside with low water solubility, [3] Hesperidin is found in citrus fruits and upon ingestion it releases its aglycone, hesperetin. Neohesperidin is the 7-O- neohesperidoside of hesperetin.
Metabolism
Hesperidin 6-O-α-L-rhamnosyl-β-D-glucosidase is an enzyme that uses hesperidin and H2O to produce hesperetin and rutinose. [1] Upon digestion in the gastrointestinal tract, hesperetin – as for all flavonoids – is rapidly metabolized in intestinal and liver cells, releasing smaller metabolites into the blood and urine for excretion. [1] The biological effects of such metabolites in vivo are unknown.
Laboratory research
In vitro, hesperetin may affect the slow inactivation phase of inward sodium current channels, and therefore could be used as a template to develop drugs against cardiac arrhythmias. [4] Hesperetin also inhibits TRPM3 channels in vitro. [5]
References
- ^ a b c d "Flavonoids". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. 2024. Retrieved 10 May 2024.
- ^ "Hesperetin". PubChem, US National Library of Medicine. 4 May 2024.
- ^ Majumdar S.; Srirangam, R. (2009). "Solubility, stability, physicochemical characteristics and in vitro ocular tissue permeability of hesperidin: a natural bioflavonoid". Pharm. Res. 26 (5): 1217–1225. doi: 10.1007/s11095-008-9729-6. PMC 2664388. PMID 18810327.
- ^ Alvarez‐Collazo, Julio; López‐Requena, Alejandro; Galán, Loipa; Talavera, Ariel; Alvarez, Julio L.; Talavera, Karel (27 March 2019). "The citrus flavanone hesperetin preferentially inhibits slow‐inactivating currents of a long QT syndrome type 3 syndrome Na+ channel mutation". British Journal of Pharmacology. 176 (8): 1090–1105. doi: 10.1111/bph.14577. PMC 6451064. PMID 30650182.
- ^ Straub, Isabelle; Krügel, Ute; Mohr, Florian; Teichert, Jens; Rizun, Oleksandr; Konrad, Maik; Oberwinkler, Johannes; Schaefer, Michael (November 2013). "Flavanones that selectively inhibit TRPM3 attenuate thermal nociception in vivo". Molecular Pharmacology. 84 (5): 736–750. doi: 10.1124/mol.113.086843. ISSN 1521-0111. PMID 24006495.
External links
-
 Media related to
Hesperetin at Wikimedia Commons
Media related to
Hesperetin at Wikimedia Commons