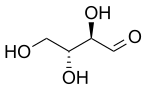
 D-Erythrose
| |
 L-Erythrose
| |
| Names | |
|---|---|
|
IUPAC names
(2R,3R)-2,3,4-Trihydroxybutanal (D)
(2S,3S)-2,3,4-Trihydroxybutanal (L) | |
| Identifiers | |
3D model (
JSmol)
|
|
| 5805561 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.643 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C4H8O4 | |
| Molar mass | 120.104 g·mol−1 |
| Appearance | Light yellow syrup |
| highly soluble | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Erythrose is a tetrose saccharide with the chemical formula C4H8O4. It has one aldehyde group, and is thus part of the aldose family. The natural isomer is D-erythrose; it is a diastereomer of D -threose. [2]
Erythrose was first isolated in 1849 from rhubarb by the French pharmacist Louis Feux Joseph Garot (1798-1869), [3] and was named as such because of its red hue in the presence of alkali metals (ἐρυθρός, "red"). [4] [5]
Erythrose 4-phosphate is an intermediate in the pentose phosphate pathway [6] and the Calvin cycle. [7]
Oxidative bacteria can be made to use erythrose as its sole energy source. [8]
See also
References
- ^ Merck Index, 11th Edition, 3637
- ^ "4.5: Diastereomers". Chemistry LibreTexts. 2015-04-01. Retrieved 2021-11-17.
- ^ Obituary of Garot (1869) Journal de pharmacie et de chimie, 4th series, 9 : 472-473.
- ^ Garot (1850) "De la matière colorante rouge des rhubarbes exotiques et indigènes et de son application (comme matière colorante) aux arts et à la pharmacie" (On the red coloring material of exotic and indigenous rhubarb and on its application (as a coloring material) in the arts and in pharmacy), Journal de Pharmacie et de Chimie, 3rd series, 17 : 5-19. Erythrose is named on p. 10: "Celui que je propose, sans y attacher toutefois la moindre importance, est celui d'érythrose, du verbe grec 'ερυθραινω, rougir (1)." (The one [i.e., name] that I propose, without attaching any importance to it, is that of erythrose, from the Greek verb ερυθραινω, to redden (1).)
-
^ Wells, David Ames; Cross, Charles Robert; Bliss, George; Trowbridge, John; Nichols, William Ripley; Kneeland, Samuel (1851).
Annual of Scientific Discovery. Boston: Gould, Kendall, and Lincoln. p.
211. Retrieved 11 December 2014.
erythrose discovery.
- ^ Kruger, Nicholas J; von Schaewen, Antje (June 2003). "The oxidative pentose phosphate pathway: structure and organisation". Current Opinion in Plant Biology. 6 (3): 236–246. doi: 10.1016/S1369-5266(03)00039-6. PMID 12753973.
- ^ Schwender, Jörg; Goffman, Fernando; Ohlrogge, John B.; Shachar-Hill, Yair (9 December 2004). "Rubisco without the Calvin cycle improves the carbon efficiency of developing green seeds". Nature. 432 (7018): 779–782. Bibcode: 2004Natur.432..779S. doi: 10.1038/nature03145. PMID 15592419. S2CID 4401215.
- ^ Hiatt, Howard H; Horecker, B L (13 October 1955). "D-erythrose metabolism in a strain of Alcaligenes faecalis". Journal of Bacteriology. 71 (6): 649–654. doi: 10.1128/jb.71.6.649-654.1956. PMC 314578. PMID 13345750. Retrieved 11 December 2014.
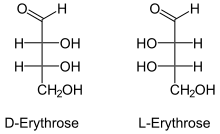
 D-Erythrose
| |
 L-Erythrose
| |
| Names | |
|---|---|
|
IUPAC names
(2R,3R)-2,3,4-Trihydroxybutanal (D)
(2S,3S)-2,3,4-Trihydroxybutanal (L) | |
| Identifiers | |
3D model (
JSmol)
|
|
| 5805561 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.643 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C4H8O4 | |
| Molar mass | 120.104 g·mol−1 |
| Appearance | Light yellow syrup |
| highly soluble | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Erythrose is a tetrose saccharide with the chemical formula C4H8O4. It has one aldehyde group, and is thus part of the aldose family. The natural isomer is D-erythrose; it is a diastereomer of D -threose. [2]

Erythrose was first isolated in 1849 from rhubarb by the French pharmacist Louis Feux Joseph Garot (1798-1869), [3] and was named as such because of its red hue in the presence of alkali metals (ἐρυθρός, "red"). [4] [5]
Erythrose 4-phosphate is an intermediate in the pentose phosphate pathway [6] and the Calvin cycle. [7]
Oxidative bacteria can be made to use erythrose as its sole energy source. [8]
See also
References
- ^ Merck Index, 11th Edition, 3637
- ^ "4.5: Diastereomers". Chemistry LibreTexts. 2015-04-01. Retrieved 2021-11-17.
- ^ Obituary of Garot (1869) Journal de pharmacie et de chimie, 4th series, 9 : 472-473.
- ^ Garot (1850) "De la matière colorante rouge des rhubarbes exotiques et indigènes et de son application (comme matière colorante) aux arts et à la pharmacie" (On the red coloring material of exotic and indigenous rhubarb and on its application (as a coloring material) in the arts and in pharmacy), Journal de Pharmacie et de Chimie, 3rd series, 17 : 5-19. Erythrose is named on p. 10: "Celui que je propose, sans y attacher toutefois la moindre importance, est celui d'érythrose, du verbe grec 'ερυθραινω, rougir (1)." (The one [i.e., name] that I propose, without attaching any importance to it, is that of erythrose, from the Greek verb ερυθραινω, to redden (1).)
-
^ Wells, David Ames; Cross, Charles Robert; Bliss, George; Trowbridge, John; Nichols, William Ripley; Kneeland, Samuel (1851).
Annual of Scientific Discovery. Boston: Gould, Kendall, and Lincoln. p.
211. Retrieved 11 December 2014.
erythrose discovery.
- ^ Kruger, Nicholas J; von Schaewen, Antje (June 2003). "The oxidative pentose phosphate pathway: structure and organisation". Current Opinion in Plant Biology. 6 (3): 236–246. doi: 10.1016/S1369-5266(03)00039-6. PMID 12753973.
- ^ Schwender, Jörg; Goffman, Fernando; Ohlrogge, John B.; Shachar-Hill, Yair (9 December 2004). "Rubisco without the Calvin cycle improves the carbon efficiency of developing green seeds". Nature. 432 (7018): 779–782. Bibcode: 2004Natur.432..779S. doi: 10.1038/nature03145. PMID 15592419. S2CID 4401215.
- ^ Hiatt, Howard H; Horecker, B L (13 October 1955). "D-erythrose metabolism in a strain of Alcaligenes faecalis". Journal of Bacteriology. 71 (6): 649–654. doi: 10.1128/jb.71.6.649-654.1956. PMC 314578. PMID 13345750. Retrieved 11 December 2014.
